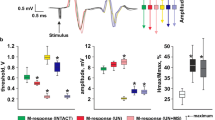
Functional state of the neuromotor system after administration of a nonspecific NO synthase blocker L-NAME was studied on the model of experimental contusion of the spinal cord. Electron paramagnetic resonance measurements of NO production in the damaged segment of the spinal cord were performed for estimation of the dynamics of intensity of NO production during traumatic disease of the spinal cord and selection of optimal period for L-NAME administration. The status of the neuromotor system was evaluated by stimulation electromyography. Treatment with L-NAME during the acute period of traumatic injury to the spinal cord sharply reduced the intensity of evoked motor responses and more pronounced increase in excitability of peripheral motor structures. The results suggest that NO system is a factor of regulation of the stress-induced and adaptive responses of the body at the early stage of spinal cord injury.
Similar content being viewed by others
References
Andrianov VV, Yafarova GG, Tumakaev RF. NO content in rat heart increased in 3 days after spinal cord injury: EPR spectroscopy data. Al’manakh Sovr. Nauki Orazovaniya. 2013(3): 12-13. Russian.
Basnakian AG, Baskov AV, Sokolov NN, Borshenko IA. Apoptosis after traumatic spinal cort injuri: prospects of the pharmacological correction reveiw. Vopr. Med. Khimii. 2000; 46(5):431-443. Russian.
Gainutdinov KhL, Faisullina RI, Andrianov VV, Gilmutdinova RI, Iyudin VS, Jafarova GG, Sitdikov FG. Nitric oxide level in the rat tissues increases after 30-day hypokinesia: studies by electron paramagnetic resonance (EPR) spectroscopy. Bull. Exp. Biol. Med. 2013;154(5):635-637.
Dmitrenko NP, Holian A. Role of interaction of metabolism paths of formaldehyde and nitrogen oxide in the mechanism of their toxic effect. 2. Toxic effect of nitrogen oxide. Ukr. Biochem. J. 2005;77(5):5-23.
Lomonosova YuN, Nemirovskaya TL, Shvets VI, Kalamkarov GR, Bugrova AE, Shevchenko TF, Kartashkina NL, Lysenko EA. Protective effect of L-arginine administration on proteins of unloaded m. soleus. Biochemistry (Mosc.). 2011;76(5):571-580.
Malyshev IYu, Manukhina EB. Stress, adaptation, and nitric oxide. Biochemistry (Mosc.). 1998;63(7):840-853.
Reutov VP. Cycle of NO in mammals. Uspekhi Biol. Khimii. 1995;35:189-228. Russian.
Sitdikova GF, Zefirov AL. Gaseous messengers in the nervous system. Ross. Fiziol. Zh. 2006;92(7):872-882. Russian.
Bredt DS. Nitric oxide signaling in brain: potentiating the gain with YC-1. Mol. Pharmacol. 2003;63(6):1206-1208.
Calabrese V, Cornelius C, Rizzarelli E, Owen JB, Dinkova-Kostova AT, Butterfield DA. Nitric oxide in cell survival: a janus molecule. Antioxid. Redox Signal. 2009;11(11):2717-2739.
Evangelista AM, Rao VS, Filo AR, Marozkina NV, Doctor A, Jones DR, Gaston B, Guilford WH. Direct regulation of striated muscle myosins by nitric oxide and endogenous nitrosothiols. PLoS One. 2010;5(6):e11209.
Hamada Y, Ikata T, Katoh S, Tsuchiya K, Niwa M, Tsutsumishita Y, Fukuzawa K. Roles of nitric oxide in compression injury of rat spinal cord. Free Radic Biol. Med. 1996;20(1):1-9.
Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007;87(1):315-424.
Gotoh T, Mori M. Nitric oxide and endoplasmic reticulum stress. Arterioscler. Thromb. Vasc. Biol. 2006;26(7):1439-1446.
Vanin AF, Huisman A, van Faassen EE. Iron dithiocarbamate as spin trap for nitric oxide detection: pitfalls and successes. Methods Enzymol. 2002;359:27-42.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 162, No. 9, pp. 295-299, September, 2016
Rights and permissions
About this article
Cite this article
Yafarova, G.G., Andrianov, V.V., Yagudin, R.K. et al. Effects of NO Synthase Blocker L-NAME on Functional State of the Neuromotor System during Traumatic Disease of the Spinal Cord. Bull Exp Biol Med 162, 316–319 (2017). https://doi.org/10.1007/s10517-017-3604-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-017-3604-6