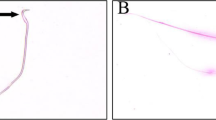
Intramuscular injections of Relatox in therapeutic and toxic doses to young outbred laboratory rats for 14 days caused no changes in the peripheral blood and bone marrow parameters, serum biochemical parameters, and morphology of the major viscera. In the toxic dose, the drug caused local irritation (inflammation, atrophy, and sclerosis in muscle tissue). Regeneration processes started in muscle tissue 7 days after Relatox withdrawal.
Similar content being viewed by others
References
A. Kurenkov, S. Nikitin, A. Artemenko, and O. Orlova, Vrach, No. 7, 8-12 (2009).
S. A. Likhachev, Yu. N. Rushkevich, T. N. Chernukha, and E. V. Veevnik, Zh. Nevrol., 9, No. 3, 27-31 (2009).
Microscopic Methods [in Russian], Eds. D. S. Sarkisov and Yu. L. Petrov, Moscow (1996).
O. R. Orlova, Ross. Med. Zh., 14, No. 23, 1700-1707 (2006).
E. V. Plotnikova and V. D. Elkin, Eksp. Klin. Dermatokosmetol., No. 6, 3-6 (2013).
Manual of Preclinical Studies of Drugs [in Russian], Ed. A. N. Mironov, Part 1, Moscow (2012).
V. N. Salkov, Det. Podrostkov. Reabilitats., No. 2, 33-36 (2004).
Supplement: Spasticity: Etiology, Evaluation, Management, and the Role of Botulinum Toxin type A. A Training Syllabus Developed by the Spasticity Study Group, Muscle Nerve, 20, No. S6, 146-168 (1997).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 160, No. 7, pp. 63-66, July, 2015
Rights and permissions
About this article
Cite this article
Churin, A.A., Fomina, T.I., Ermolaeva, L.A. et al. Study of Subchronic Toxicity of Relatox on Sexually Immature Animals. Bull Exp Biol Med 160, 53–56 (2015). https://doi.org/10.1007/s10517-015-3096-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-015-3096-1