Abstract
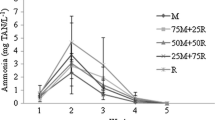
The aim of this study was to evaluate the effect of using different vegetable brans as organic carbon source in synbiotic system fertilization on the nitrification process, plankton composition, and growth of Penaeus vannamei in the nursery phase, also comparing it with the biofloc system. An extended nursery rearing was carried out for 53 days, at a density of 2000 shrimp m−3 (initial weight: 0.03 ± 0.01 g). The following treatments were established, with five repetitions: CW, clear water (control); BFT, biofloc system; RB, synbiotic system fertilized with rice bran; SB, synbiotic system fertilized with soybean bran; and WB, synbiotic system fertilized with wheat bran. The synbiotic fertilization protocol used a commercial blend of Bacillus subtilis and Bacillus licheniformis, molasses, sodium bicarbonate as buffer, and water. The fertilizers were processed by an anaerobic (24 h) and an aerobic (24 h) phase. BFT treatment used molasses as organic carbon source. At the end of the trial, final weight was higher in CW, BFT, and RB treatments than in WB. In RB, SB, and WB treatments, TAN was controlled between days 10 and 14 and NO2−-N was controlled from day 40 of the trial, resembling a newly started system. At the end of the trial, a higher abundance of coccoid and bacillus was observed in the RB treatment, while a higher abundance of vibrio bacteria was observed in WB. Rice bran proved to be the best alternative for the synbiotic fertilization, as it presented a final weight (3.27 g) similar to BFT and CW treatments, and higher than WB (2.61 g). Also, the use of rice bran produced a high load of microorganisms, which can improve shrimp growth.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Abakari G, Luo G, Kombat EO (2021) Dynamics of nitrogenous compounds and their control in biofloc technology (BFT) systems: a review. Aquac Fish 6:441–447. https://doi.org/10.1016/j.aaf.2020.05.005
Abdel-Tawwab M, Khalil RH, Nour AM, Elkhayat BK, Khalifa E, Abdel-Latif HMR (2020) Effects of Bacillus subtilis-fermented rice bran on water quality. Performance. Antioxidants/oxidants. And immunity biomarkers of white leg shrimp (Litopenaeus vannamei) reared at different salinities with zero water exchange. J Appl Aquac. https://doi.org/10.1080/10454438.2020.1844110
Abreu PC, Ballester ELC, Odebrecht C, Wasielesky W, Cavalli RO, Granéli W, Anesio AM (2007) Importance of biofilm as food source for shrimp (Farfantepenaeus paulensis) evaluated by stable isotopes (δ13C and δ15N). J Exp Mar Biol Ecol 347(1–2):88–96. https://doi.org/10.1016/j.jembe.2007.03.012
Alday-Sanz V (2010) The shrimp Book. Nottingham University, Nottingham
Aminot A, Chaussepied M (1983) Manuel Des analyses chimiques en milieu marin. CNEXO, Brest
Andrade RJV, dos Santos EP, Costa GKA, Campos CVFS, da Silva SMBC, Gálvez AO, Brito LO (2021) Effect of different frequencies of the addition of Brachionus plicatilis on the performance of Litopenaeus vannamei in a nursery biofloc system with rice bran (anaerobic and aerobic) as an organic carbon source. Aquaculture 540:736669. https://doi.org/10.1016/j.aquaculture.2021.736669
Anson NM, Hemery YM, Bast A, Haenen GRMM (2012) Optimizing the bioactive potential of wheat bran by processing. Food Funct 3(4):362. https://doi.org/10.1039/C2FO10241B
APHA (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association. Washington, DC
Avnimelech Y (2012) Biofloc technology – a practical guide book, 2nd edn. The World Aquaculture Society. Baton Rouge.
Boone L (1931) A collection of anomuran and macruran Crustacea from the Bay of Panama and the fresh waters of the Canal Zone. Bull Am Museum Nat History 63:137–189
Boyd CE, Tucker CS (1998) Pond aquaculture water quality management. Kluwer Academic
Decamp O, Warren A, Sanchez R (1999) The role of ciliated protozoa in subsurface flow wetlands and their potential as bioindicators. 40:91–98. https://doi.org/10.1016/S0273-1223(99)00444-8
Deng M, Chen J, Gou J, Hou J, Li D, He X (2018) The effect of different carbon sources on water quality. Microbial community and structure of biofloc systems. Aquaculture 482:103–110. https://doi.org/10.1016/j.aquaculture.2017.09.030
Dinno A (2017) Dunn.test: Dunn’s test of multiple comparisons using rank sums. R package version 1.3.5. https://CRAN.R-project.org/package=dunn.test
Eaton DE, Clesceri LS, Greenberg AE (1995) Standard methods for the examination of water and wastewater, 19th edn. Publication Office, American Public Health Association, Washington D.C
Ebeling JM, Timmons MB, Bisogni JJ (2006) Engineering Analysis of the Stoichiometry of Photoautotrophic. Autotrophic. And Heterotrophic Removal of Ammonia – Nitrogen in Aquaculture Systems. 257, 346–358. https://doi.org/10.1016/j.aquaculture.2006.03.019
Ekasari J, Hanif Azhar M, Surawidjaja EH, Nuryati S, De Schryver P, Bossier P (2014) Immune response and disease resistance of shrimp fed biofloc grown on different carbon sources. Fish Shellfish Immunol 41(2):332–339. https://doi.org/10.1016/j.fsi.2014.09.004
Emerenciano M, Gaxiola G, Cuzon G (2013) Biofloc Technology (BFT): a review for aquaculture application and Animal Food Industry. Matovic. M.D. Biomass now – cultivation and utilization. Rijeka. InTech, Croatia, pp 301–328. https://doi.org/10.5772/53902
Engle CR, McNevin A, Racine P, Boyd CE, Paungkaew D, Viriyatum R, Tinh HQ, Minh HN (2017) Economics of sustainable intensification of aquaculture: evidence from shrimp farms in Vietnam and Thailand. J World Aquacult Soc 48:227–239. https://doi.org/10.1111/jwas.12423
Esteves FA (2011) Fundamentos de limnologia. 3rd ed. Interciência. 826p
Ferreira LMMHM (2008) Formação de flocos microbianos em cultivo do camarão rosa Farfantepenaeus paulensis e do Camarão-branco Litopenaeus vannamei. Master Thesis, Federal University of Rio Grande, Rio Grande, Brazil, 96 pp
Ferreira GS, Bolívar NC, Pereira SA, Guertler C, Vieira FN, Mouriño JLP, Seiffert WQ (2015) Microbial biofloc as source of probiotic bacteria for the culture of Litopenaeus vannamei Aquaculture 448:273–279. https://doi.org/10.1016/j.aquaculture.2015.06.006
Fox J, Weisberg S (2019) An {R} Companion to applied regression. 3rd edn. Sage, Thousand Oaks. https://socialsciences.mcmaster.ca/jfox/Books/Companion/
Furtado PS, Poersch LH, Wasielesky W (2011) Effect of calcium hydroxide. Carbonate and sodium bicarbonate on water quality and zootechnical performance of shrimp Litopenaeus vannamei reared in bio-flocs technology (BFT) systems. Aquaculture 321:130–135. https://doi.org/10.1016/j.aquaculture.2011.08.034
Gaona CAP, Poersch LH, Krummenauer D, Foes GK, Wasielesky WJ (2011) The effect of solids removal on water quality. Growth and survival of Litopenaeus vannamei in a biofloc technology culture system. Int J Recirc Aquac 12:54–73. https://doi.org/10.21061/ijra.v12i1.1354
García-Robledo E, Corzo A, Papaspyrou S (2014) A fast and direct spectrophotometric method for the sequential determination of nitrate and nitrite at low concentrations in small volumes. Mar Chem 162:30–36. https://doi.org/10.1016/j.marchem.2014.03.002
Gebrezgi D (2019) Proximate composition of complementary food prepared from maize (Zea mays). Soybean (Glycine max) and Moringa leaves in Tigray. Ethiopia Cogent Food Agric 5:1. https://doi.org/10.1080/23311932.2019.1627779
Godoy LC, Odebrecht C, Ballester E, Martins TG, Wasielesky W (2012) Effect of diatom supplementation during the nursery rearing of Litopenaeus vannamei (Boone, 1931) in a heterotrophic culture system. Aquacult Int 20:559–569. https://doi.org/10.1007/s10499-011-9485-1
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analysis. Paleontol Eletron 4(1):9
Hargreaves JA (1998) Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 166(3–4):181–212. https://doi.org/10.1016/S0044-8486(98)00298-1
Hargreaves JA (2006) Photosynthetic suspended-growth systems in aquaculture. Aquacult Eng 34(3):344–363. https://doi.org/10.1016/j.aquaeng.2005.08.009
Hargreaves JA (2013) Biofloc production systems for aquaculture. Stoneville. Southern Regional Aquaculture Center, MS, p 4503
Hobbie JE, Daley RJ, Jasper S (1977) Use of nuclepore filters for counting bacteria by fluorescence microscopy. Appl Environ Microbiol Microbiol 33:1225–1228. https://doi.org/10.1128/aem.33.5.1225-1228.1977
Hope RM (2022) Rmisc: Ryan Miscellaneous. R package version 1.5.1. https://CRAN.R-project.org/package=Rmisc
Hostins B, Lara G, Decamp O, Cesar DE, Wasielesky W (2017) Efficacy and variations in bacterial density in the gut of Litopenaeus vannamei reared in a BFT system and in clear water supplemented with a commercial probiotic mixture. Aquaculture 480:58–64. https://doi.org/10.1016/j.aquaculture.2017.07.036
Hussain AS, Mohammad DA, Sallam WS, Shoukry NM, Davis DA (2021) Effects of culturing the Pacific white shrimp Penaeus vannamei in biofloc vs synbiotic systems on the growth and immune system. Aquaculture 542:736905. https://doi.org/10.1016/j.aquaculture.2021.736905
Jory DE, Cabrera TR, Dugger DM, Fegan D, Lee PG, Lawrence AL, Jackson CJ, Mcintosh RP, Castañeda J (2001) A global review of shrimp feed management: status and perspectives. In: Browdy CL, Jory DE (Eds.), The New Wave: Proceedings of the Special Session on Sustainable Shrimp Culture. They World Aquaculture Society, Baton Rouge, LA, 104–152
Katalani K, Wasielesky W, Zuñiga R, De Souza MS, Foes G (2023) Comparison between biofloc technology system and aquamimicry in the cultivation of Litopenaeus vannamei in lined ponds in Southern Brazil. Bol do Instituto De Pesca 49:e787. https://doi.org/10.20950/1678-2305/bip.2023.49.e787
Kawahigashi D (2018) Synbiotics as a management tool for improving nursery efficiency. Hatcheryfeed 6:36–39
Khanjani MH, Sharifinia M (2020) Biofloc technology as a promising tool to improve aquaculture production. Rev Aquac 12:1836–1850. https://doi.org/10.1111/raq.12412
Khanjani MH, Mohammadi A, Emerenciano MGC (2022) Microorganisms in biofloc aquaculture system. Aquac Rep 26:101300. https://doi.org/10.1016/j.aqrep.2022.101300
Khanjani MH, Brito LO, Fóes GK, Vieira FN, Poli MA, Santos M, Emerenciano MGC (2023) Synbiotics and aquamimicry as alternative microbial-based approaches in intensive shrimp farming and biofloc: novel disruptive techniques or complementary management tools? A scientific-based overview. Aquaculture 567:739273. https://doi.org/10.1016/j.aquaculture.2023.739273
Krummenauer D, Peixoto S, Cavalli RO, Poersch LH, Wasielesky W (2011) Superintensive culture of white shrimp. Litopenaeus vannamei. In a biofloc technology system in southern Brazil at different stocking densities. J World Aquac Soc 42:726–733. https://doi.org/10.1111/j.1749-7345.2011.00507.x
Krummenauer D, Samocha T, Poersch L, Lara G, Wasielesky W (2014a) The reuse of water on the culture of Pacific white shrimp. Litopenaeus vannamei. In BFT system. J World Aquac Soc 45:3–14. https://doi.org/10.1111/jwas.12093
Krummenauer D, Poersch L, Romano LA, Lara GR, Encarnação P, Wasielesky W Jr (2014b) The Effect of Probiotics in a Litopenaeus vannamei Biofloc Culture System infected with Vibrio parahaemolyticus J Appl Aquac 26(4):370–379. https://doi.org/10.1080/10454438.2014.965575
Lima PCM, da Silva AEM, da Silva DA, da Silva SMBC, Brito LO, Gálvez AO (2021) Effect of stocking density of Crassostrea sp. In a multitrophic biofloc system with Litopenaeus vannamei in nursery. Aquaculture 530:735913. https://doi.org/10.1016/j.aquaculture.2020.735913
Lin Y-C, Chen J-C (2003) Acute toxicity of nitrite on Litopenaeus vannamei (Boone) juveniles at different salinity levels. Aquaculture 224(1–4):193–201. https://doi.org/10.1016/S0044-8486(03)00220-5
Liñan-Vidriales MA, Peña-Rodríguez A, Tovar-Ramírez D, Elizondo-González R, Barajas-Sandoval DR, Ponce-Gracía EI, Rodríguez-Jaramillo C, Balcázar JL, Quiroz-Guzmán E (2020) Effect of rice bran fermented with Bacillus and Lysinibacillus species on dynamic microbial activity of Pacific white shrimp (Penaeus vannamei). Aquaculture 531:735958. https://doi.org/10.1016/j.aquaculture.2020.735958
Liu H, Li H, Wei H, Zhu X, Han D, Jin J, Yang Y, Xie S (2019) Biofloc formation improves water quality and fish yield in a freshwater pond aquaculture system. Aquaculture 506:256–269. https://doi.org/10.1016/j.aquaculture.2019.03.031
Marinho YF, Brito LO, Silva Campos CVF, Severi W, Andrade HA, Galvez AO (2017) Effect of the addition of Chaetoceros calcitrans, Navicula sp. and Phaeodactylum tricornutum (diatoms) on phytoplankton composition and growth of Litopenaeus vannamei (Boone) postlarvae reared in a biofloc system. Aquac Res 48:4155–4164. https://doi.org/10.1111/are.13235
Martins TG, Odebrecht C, Jensen LV, D’Oca MG, Wasielesky W (2016) The contribution of diatoms to bioflocs lipid content and the performance of juvenile Litopenaeus vannamei (Boone, 1931) in a BFT culture system. Aquac Res 47:1315–1326. https://doi.org/10.1111/are.12592
Moro JD, Rosa CS, Hoelzel SCS (2004) Composição centesimal e ação antioxidante do farelo de arroz e seus benefícios à saúde. Discip Sci 4(1):33–44. https://doi.org/10.37777/866
Onipe OO, Jideani AIO, Beswa D (2015) Composition and functionality of wheat bran and its application in some cereal food products. Int J Food Sci Technol 50(12):2509–2518. https://doi.org/10.1111/ijfs.12935
Páez-Osuna F (2001) The environmental impact of shrimp aquaculture: a global perspective. Environ Pollut 112(2):229–231. https://doi.org/10.1016/S0269-7491(00)00111-1
Panigrahi A, Sundaram M, Saranya C, Swain S, Dash RR, Dayal JS (2019) Carbohydrate sources deferentially influence growth performances. Microbial dynamics and immunomodulation in Pacific white shrimp (Litopenaeus vannamei) under biofloc system. Fish Shellfish Immunol 86:1207–1216. https://doi.org/10.1016/j.fsi.2018.12.040
Pimentel OALF, de Oliveira VQ, Oliveira CRR, Severi W, Gálvez AO, Amado AM, Brito LO (2022) Assessment of different ionic adjustment strategies in low-salinity water on the growth of Litopenaeus vannamei and microbial community stoichiometry in a synbiotic nursery system. Aquac Res 53:50–62. https://doi.org/10.1111/are.15552
Pohlert T (2022) PMCMRplus: Calculate pairwise multiple comparisons of mean rank sums extended. R package version 1.9.6. https://CRAN.R-project.org/package=PMCMRplus
R Core Team (2023) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Rajkumar M, Pandey PK, Aravind R, Vennila A, Bharti V, Purushothaman CS (2016) Effect of different biofloc system on water quality. Biofloc composition and growth performance in Litopenaeus vannamei (Boone. 1931). Aquac Res 47(11):3432–3444. https://doi.org/10.1111/are.12792
Rashid NYA, Razak DLA, Jamaluddin A, Sharifuddin SA, Long K (2015) Bioactive compounds and antioxidant activity of rice bran fermented with lactic acid bacteria. Malaysian J Microbiol 11:156–162. https://doi.org/10.21161/mjm.12714
Reis WG, Wasielesky W, Abreu PC, Brandão H, Krummenauer D (2019) Rearing of the Pacific white shrimp Litopenaeus vannamei (Boone. 1931) in BFT system with different photoperiods: effects on the microbial community. Water quality and zootechnical performance. Aquaculture 508:19–29. https://doi.org/10.1016/j.aquaculture.2019.04.067
Robles-Porchas GR, Gollas‐Galván T, Martínez‐Porchas M, Martínez‐Cordova LR, Miranda‐Baeza A, Vargas‐Albores F (2020) The nitrification process for nitrogen removal in biofloc system aquaculture. Rev Aquac 12:2228–2249. https://doi.org/10.1111/raq.12431
Romano N (2017) Aquamimicry: A revolutionary concept for shrimp farming. Global aquaculture advocate. https://www.globalseafood.org/advocate/aquamimicry-a-revolutionary-concept-for-shrimp-farming/. Accessed Mar 2023
Romano N, Kumar V (2017) Vegetarian shrimp: pellet-free shrimp farming. World Aquaculture Magazine. December
Romano N, Dauda AB, Ikhsan N, Karim M, Kamarudin MS (2018) Fermenting rice bran as a carbon source for biofloc technology improved the water quality. Growth. Feeding efficiencies. And biochemical composition of African catfish Clarias gariepinus juveniles. Aquac Res 49:3691–3701. https://doi.org/10.1111/are.13837
Sadh PK, Suresh K, Prince C, Joginder SD (2018) Fermentation: a Boon for production of Bioactive compounds by Processing of Food Industries Wastes (By-Products). Molecules 23:2560. https://doi.org/10.3390/molecules23102560
Samocha TM (2019) Sustainable Biofloc systems for Marine shrimp. Academic, San Diego
Santos SFM (2020) Sistema aquamimicry: cultivo de Litopenaeus vannamei em diferentes densidades de estocagem. Master Thesis, Federal University of Rio Grande, Rio Grande, Brazil, 58pp
Santos RB, Coelho Filho PA, Gonçalves AP, Santos RA, Lins Rodrigues M, Correia ES, Oliveira VQ, Brito LO (2022) Effects of organic carbon sources on water quality, microbial flocs protein and performance of Macrobrachium rosenbergii post-larvae reared in biofloc and synbiotic systems. Aquac Res 53:388–397. https://doi.org/10.1111/are.15580
Scheffer M (2004) Ecology of shallow lakes. Kluwer Academic
Serra FP, Gaona CAP, Furtado PS, Poersch LH, Wasielesky W (2015) Use of different carbon sources for the biofloc system adopted during the nursery and grow-out culture of Litopenaeus vannamei Aquacult Int 23:1325–1339. https://doi.org/10.1007/s10499-015-9887-6
Silva DA, de Lima PC, da Silva AE, de Oliveira Filho PRC, da Silva SMBC, Gálvez AO, Brito LO (2021) Effects of adding rotifers on the water quality. Plankton composition and growth of Pacific white shrimp. Litopenaeus vannamei juvenile. When cultured with biofloc technology. Aquac Res 00:1–14. https://doi.org/10.1111/are.15276
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis, 2nd edn. Fisheries research board of Canada, Ottawwa
Suita SM, Ballester ELC, Abreu PC, Wasielesky W (2015) Dextrose as carbon source in the culture of Litopenaeus vannamei (Boone. 1931) in a zero exchange system lat. Am J Aquat Res 43(3):526–533. https://doi.org/10.3856/vol43-issue3-fulltext-13
Timmons MB, Ebeling JM (2013) Recirculating Aquaculture 3rd edition. Ithaca Publishing Company LLC, Ithaca
Tinh TH, Koppenol T, Hai TN, Verreth JAJ, Verdegem MCJ (2021) Effects of carbohydrate sources on a biofloc nursery system for whiteleg shrimp (Litopenaeus vannamei). Aquaculture 735795. https://doi.org/10.1016/j.aquaculture.2020.735795
UNESCO (1983) Chemical methods for use in marine environmental monitoring, Intergovernamental Oceanographic commission – Manuals and guides 12
Utermöhl H (1958) Zur Vervollkommnung Der Quantitativen Phytoplankton-Methodik. Mitt int Ver Theor Angew Limnol 9:38
Van Wyk P, Davis-Hodgkins M, Laramore R, Main KL, Mounta J, Scarpa J (1999) Farming marine shrimp in recirculating freshwater systems. Florida Department of Agriculture and Consumer Services – Harbor Branch Oceanographic Institution, Florida
Vidal JMA, Pessôa MNC, Santos FL, Mendes PP, Mendes ES (2018) Probiotic potential of Bacillus cereus against Vibrio spp. in post-larvae shrimps. Rev Caatinga 31(2):495–503. https://doi.org/10.1590/1983-21252018v31n226rc
Wasielesky W, Bezerra A, Poersch L, Hoffling FB, Krummenauer D (2020) Effect of feeding frequency on the white shrimp Litopenaeus vannamei during the pilot-scale nursery phase of a superintensive culture in a biofloc system. J World Aquac Soc 51:1175–1191. https://doi.org/10.1111/jwas.12694
Wei Y, Liao S-A, Wang A (2016) The effect of different carbon sources on the nutritional composition. Microbial community and structure of bioflocs. Aquaculture 465:88–93. https://doi.org/10.1016/j.aquaculture.2016.08.040
Wetzel RG (2001) Limnology: lake and river ecosystems, 3rd edn. Academic, San Diego
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-, New York
Xavier M, Wasielesky W, Hostins B, Bequé E, Krummenauer D (2022) The use of a flocculant additive and its effect on biofloc formation. Nitrification. And zootechnical performance during the culture of Pacific white shrimp Penaeus vannamei (Boone. 1931) in a BFT system. Lat Am J Aquat Res 50(2):181–196. https://doi.org/10.3856/vol50-issue2-fulltext-2777
Zar JH (2010) Biostatistical Analysis, 5th edn. Pearson Prentice Hall, Hoboken
Zhu Y, Wang S, Huang L, Yan M, Dong P, Chen C, Guo H, Zhang D (2021) Effects of sucrose addition on water quality and bacterioplankton community in the Pacific White Shrimp (Litopenaeus vannamei) culture system. Aquac Res. https://doi.org/10.1111/are.15257
Funding
This work was supported by the Foundation for Research Support of the State of Rio Grande do Sul – FAPERGS (process number: 21/2551-0002225-6); Coordination for the Improvement of Higher-Level Personnel (CAPES; process number: 88887.612993/2021-00 and 88887.601721/2021-00); and National Council for Scientific and Technological Development (CNPq; process number: 307741/2022-2). GUABI Animal Health and Nutrition, AQUATEC, TREVISAN, and Al Aqua donated the experimental diets, post-larvae and Aeration system, respectively.
Author information
Authors and Affiliations
Contributions
Otávio Augusto L. F. Pimentel: conceptualization, investigation, methodology, data curation, visualization, formal analysis, and writing—original draft. Wilson Wasielesky Jr: resources, supervision, visualization, funding acquisition, project administration, and writing—review and editing. Natália P. da Silva: investigation, data curation, and writing—review and editing. Lucélia do Valle Borges: investigation, data curation, and writing—review and editing. Dariano Krummenauer: conceptualization, methodology, resources, supervision, funding acquisition, project administration, visualization, and writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The research undertaken complies with the current animal welfare laws in Brazil. All the authors agree to participate in this experiment.
Consent for publication
All the authors of this article agree to the publication.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pimentel, O.A.L.F., Wasielesky, W., da Silva, N.P. et al. Fertilizing synbiotic system with different vegetable brans: effects on nitrification, plankton composition, and growth of Penaeus vannamei in the nursery phase. Aquacult Int (2024). https://doi.org/10.1007/s10499-024-01471-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-024-01471-x