Abstract
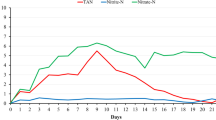
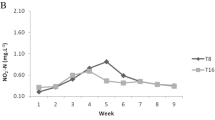
This study aimed to evaluate the effect of different management strategies on the toxic nitrogen compounds and alkalinity patterns in nurseries and pre-grow-out of Penaeus vannamei culture with low salinity water (2.5 g L−1) and synbiotic system. A study case is presented using three individual experiments (E): two nurseries (EI and EII: 2000 shrimp m−3 or 600 shrimp m−2) and one pre-grow-out (EIII: 300 shrimp m−3 or 90 shrimp m−2). Key water quality management strategies included the adoption of artificial substrate and water reuse with a microbial-based synbiotic system. In EI experiment, mean TAN was less than 0.50 mg L−1 throughout the culture, NO2−-N reached a maximum concentration of 0.80 mg L−1, and NO3−-N showed a decrease over the time. In EII, mean TAN was 0.64 mg L−1 with a peak on day 10 of the experimental course, with the same pattern occurring for NO2−-N. On the other hand, NO3−-N constantly increased from 0.10 mg L−1 at the beginning to 2.32 mg L−1 at the end of the trial. In EIII, mean TAN was less than 0.50 mg L−1 reaching a maximum concentration of 0.93 mg L−1; NO2−-N reached a maximum mean concentration of 0.70 mg L−1 at the end of the experiment. NO3−-N had a mean of 1.50 mg L−1 at the beginning of the experiment and a reduction to 0.53 mg L−1 at the end of the trial. Alkalinity showed mean concentrations of 114.90 mg L−1, 88.04 mg L−1, and 88.27 mg L−1 for EI, EII, and EIII, respectively. The results demonstrated that the proposed management protocols adopted in EI, EII, and EIII were efficient for toxic nitrogen control in culture conditions using low salinity water and synbiotic system.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Abakari G, Luo G, Kombat EO (2021) Dynamics of nitrogenous compounds and their control in biofloc technology (BFT) systems: a review. Aquac Fish 6(5):441–447. https://doi.org/10.1016/j.aaf.2020.05.005
AOAC (2016) Official methods of analysis, 20th edn. Association of Official Analytical Chemistry, Washington, DC, p 3172
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association/American Water Works Association/Water Environment Federation, Washington DC, USA, p 2005
APHA (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association, Washington, DC, USA
Avnimelech Y (2012) Biofloc technology – a practical guide book, 2nd edn. The World Aquaculture Society, Baton Rouge, Louisiana, United States
Boyd CE, Thunjai T (2003) Concentrations of major ions in waters of inland shrimp farms in China, Ecuador, Thailand, and the United States. J World Aquac Soc 34:524–532. https://doi.org/10.1111/j.1749-7345.2003.tb00092.x
Boyd CE, Tucker CS (1998) Pond aquaculture water quality management. Kluwer Academic Publishers, Massachusetts, EUA
Brito LO, Junior LC, Abreu JL, Severi W, Moraes LBS, Galvez AO (2018) Effects of two commercial feeds with high and low crude protein content on the performance of white shrimp Litopenaeus vannamei raised in an integrated biofloc system with the seaweed Gracilaria birdiae. Span J Agric Res 16(1):e0603. https://doi.org/10.5424/sjar/2018161-11451
Chen JC, Chen SF (1992) Effects of nitrite on growth and molting of Penaeus monodon juveniles. Comp Biochem Physiol Part C: Comp Pharmacol 101(3):453–458. https://doi.org/10.1016/0742-8413(92)90069-J
Cui Y, Ren X, Li J, Zhai Q, Feng Y, Xu Y, Ma L (2017) Effects of ammonia-N stress on metabolic and immune function via the neuroendocrine system in Litopenaeus vannamei. Fish Shellfish Immunol 64:270–275. https://doi.org/10.1016/j.fsi.2017.03.028
Da Silva DA, de Lima PC, da Silva AE, de Oliveira Filho PRC, da Silva SMBC, Gálvez AO, Brito LO (2021) Effects of adding rotifers on the water quality, plankton composition and growth of Pacific white shrimp, Litopenaeus vannamei juvenile, when cultured with biofloc technology. Aquac Res 00:1–14. https://doi.org/10.1111/are.15276
De Lima PCM, da Silva AEM, da Silva DA, da Silva SMBC, Brito LO, Gálvez AO (2021) Effect of stocking density of Crassostrea sp. in a multitrophic biofloc system with Litopenaeus vannamei in nursery. Aquaculture 530:735913. https://doi.org/10.1016/j.aquaculture.2020.735913
Eaton DE, Clesceri LS, Greenberg AE (1995) Standard methods for the examination of water and wastewater. 19th ed. Publication Office, American Public Health Association, Washington D.C
Ebeling JM, Timmons MB, Bisogni JJ (2006) Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia–nitrogen in aquaculture systems. Aquaculture 257:346–358. https://doi.org/10.1016/j.aquaculture.2006.03.019
El-Sayed A-FM (2021) Use of biofloc technology in shrimp aquaculture: a comprehensive review, with emphasis on the last decade. Rev Aquacult 13:676–705. https://doi.org/10.1111/raq.12494
Emerenciano MGC, Rombenso AN, Vieira FdN, Martins MA, Coman GJ, Truong HH, Noble TH, Simon CJ (2022) Intensification of penaeid shrimp culture: an applied review of advances in production systems, nutrition and breeding. Animals 12:236. https://doi.org/10.3390/ani12030236
Esparza-Leal HM, Xavier JAA, Wasielesky W (2016) Performance of Litopenaeus vannamei postlarvae reared in indoor nursery tanks under biofloc conditions at different salinities and zero-water exchange. Aquacult Int 24(5):1435–1447. https://doi.org/10.1007/s10499-016-0001-5
FAO (2020) The State of World Fisheries and Aquaculture 2020. Sustainability in action, Rome
FAO (2022) The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation. FAO, Rome. https://doi.org/10.4060/cc0461en
Ferreira LMH, Lara G, Wasielesky W, Abreu PC (2016) Biofilm versus biofloc: are artificial substrates for biofilm production necessary in the BFT system? Aquacult Int 24(4):921–930. https://doi.org/10.1007/s10499-015-9961-0
Fries J (1971) Análisis de Trazas. Métodos Fotometricos Comprobados, Merck, Darmstadt
Furtado PS, Poersch LH, Wasielesky W (2011) Effect of calcium hydroxide, carbonate and sodium bicarbonate on water quality and zootechnical performance of shrimp Litopenaeus vannamei reared in bio-flocs technology (BFT) systems. Aquaculture 321:130–135. https://doi.org/10.1016/j.aquaculture.2011.08.034
Furtado PS, Poersch H, Wasielesky W (2015) The effect of different alkalinity levels on Litopenaeus vannamei reared with biofloc technology (BFT). Aquacult Int 23(1):345–358. https://doi.org/10.1007/s10499-014-9819-x
Galkanda-Arachchige HSC, Roy LA, Davis DA (2020) Evaluation of an alternative salt mixture to culture Pacific white shrimp (Litopenaeus vannamei) in inland aquaculture. Aquac Res 51:3540–3550. https://doi.org/10.1111/are.14691
Garza de Yta A, Rouse DB, Davis DA (2004) Influence of nursery period on the growth and survival of Litopenaeus vannamei under pond production conditions. J World Aquacult Soc 35:357–365. https://doi.org/10.1111/j.1749-7345.2004.tb00099.x
Gosling E (2003) Marine bivalve Molluscs: biology, ecology and culture (456 p). Wiley-Blackwell, UK
Gross A, Abutbul S, Zilberg D (2004) Acute and chronic effects of nitrite on white shrimp, Litopenaeus vannamei, cultured in low-salinity brackish water. J World Aquac Soc 35(3):315–321. https://doi.org/10.1111/j.1749-7345.2004.tb00095.x
Hargreaves JA (1998) Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 166(3–4):181–212. https://doi.org/10.1016/S0044-8486(98)00298-1
Hart JP, Lovis WA, Schulenberg JK, Urquhart GR (2007) Paleodietary implications from stable carbon isotope analysis of experimental cooking residues. J Archaeol Sci 34:804–813. https://doi.org/10.1016/j.jas.2006.08.006
Hope RM (2022) Rmisc: Ryan Miscellaneous. R package version 1.5.1. https://CRAN.R-project.org/package=Rmisc
Jensen FB (2003) Nitrite disrupts multiple physiological functions in aquatic animals. Comp Biochem Physiol Part A: Mol Integr Physiol 135(1):9–24. https://doi.org/10.1016/S1095-6433(02)00323-9
Jiann-Chu C, Chi-Yuan L (1991) Lethal effects of ammonia and nitrite on Penaeus penicillatus juveniles at two salinity levels. Comp Biochem Physiol Part C: Comp Pharmacol 100(3):477–482. https://doi.org/10.1016/0742-8413(91)90026-P
Kawahigashi D (2018) Synbiotics as a management tool for improving nursery efficiency. Hatcheryfeed 6(3):36–39
Khanjani MH, Brito LO, Fóes GK, Vieira FN, Poli MA, Santos M, Emerenciano MGC (2023) Synbiotics and aquamimicry as alternative microbial-based approaches in intensive shrimp farming and biofloc: novel disruptive techniques or complementary management tools? A scientific-based overview. Aquaculture 567:739273. https://doi.org/10.1016/j.aquaculture.2023.739273
Krummenauer D, Samocha T, Poersch L, Lara G, Wasielesky W (2014) The reuse of water on the culture of pacific white shrimp, Litopenaeus vannamei, in BFT system. J World Aquac Soc 45(1):3–14. https://doi.org/10.1111/jwas.12093
Lara GS, Furtado P, Hostins B, Poersch L, Wasielesky W (2017) Addition of sodium nitrite and biofilm in a Litopenaeus vannamei biofloc culture system. Lat Am J Aquat Res 44(4):760–768. https://doi.org/10.3856/vol44-issue4-fulltext-11
Lara GR, Poersch LH, Wasielesky W (2021) The quantity of artificial substrates influences the nitrogen cycle in the biofloc culture system of Litopenaeus vannamei. Aquacult Eng 94:102171. https://doi.org/10.1016/j.aquaeng.2021.102171
Lin Y-C, Chen J-C (2001) Acute toxicity of ammonia on Litopenaeus vannamei Boone juveniles at different salinity levels. J Exp Mar Biol Ecol 259(1):109–119. https://doi.org/10.1016/S0022-0981(01)00227-1
Maicá PM, Borba MR, Martins TG, Wasielesky W (2014) Effect of salinity on performance and body composition of Pacific white shrimp juveniles reared in a super-intensive system. Rev Bras Zootec 43:343–350. https://doi.org/10.1590/S1516-35982014000700001
Nunes AJP, López CV (2001) Low-salinity, inland shrimp culture in Brazil and Ecuador. Responsible Seafood Advocate. https://www.globalseafood.org/advocate/low-salinity-inland-shrimp-culture-in-brazil-and-ecuador/. Accessed 14 july 2023
R Core Team (2023) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ray AJ, Dillon KS, Lotz JM (2011) Water quality dynamics and shrimp (Litopenaeus vannamei) production in intensive, mesohaline culture systems with two levels of biofloc management. Aquacult Eng 45(3):127–136. https://doi.org/10.1016/j.aquaeng.2011.09.001
Robles-Porchas GR, Gollas-Galván T, Martínez-Porchas M, Martínez-Cordova LR, Miranda-Baeza A, Vargas-Albores F (2020) The nitrification process for nitrogen removal in biofloc system aquaculture. Rev Aquacult 12:2228–2249. https://doi.org/10.1111/raq.12431
Romano N, Zeng C (2013) Toxic effects of ammonia, nitrite, and nitrate to decapod crustaceans: a review on factors influencing their toxicity, physiological consequences, and coping mechanisms. Rev Fish Sci 21(1):1–21. https://doi.org/10.1080/10641262.2012.753404
Romano N, Dauda AB, Ikhsan N, Karim M, Kamarudin MS (2018) Fermenting rice bran as a carbon source for biofloc technology improved the water quality, growth, feeding efficiencies, and biochemical composition of African catfish Clarias gariepinus juveniles. Aquac Res 49:3691–3701. https://doi.org/10.1111/are.13837
Roy LA, Davis DA, Saoud IP, Boyd CA, Pine HJ, Boyd CE (2010) Shrimp culture in inland low salinity waters. Rev Aquac 2(4):191–208. https://doi.org/10.1111/j.1753-5131.2010.01036.x
Samocha TM (2019) Sustainable biofloc systems for marine shrimp – The Texas A&M AgriLife Research Experience. The World Aquaculture Society, Baton Rouge, p 463p
Santos NBV, Furtado PS, César DE, Wasielesky W (2019) Assessment of the nitrification process in a culture of pacific white shrimp, using artificial substrate and bacterial inoculum in a biofloc technology system (BFT). Ciênc Rural 49:06. https://doi.org/10.1590/0103-8478cr20180306
Schveitzer R, Arantes R, Francisco M, Fóes P, Costódio S (2013a) Use of artificial substrates in the culture of Litopenaeus vannamei (Biofloc System) at different stocking densities: effects on microbial activity, water quality and production rates. Aquacult Eng 54:93–103. https://doi.org/10.1016/j.aquaeng.2012.12.003
Schveitzer R, Arantes R, Costódio PFS, do Espírito Santo CM, Arana LV, Seiffert WQ, Andreatta ER (2013) Effect of different biofloc levels on microbial activity, water quality and performance of Litopenaeus vannamei in a tank system operated with no water exchange. Aquacult Eng 56:59–70. https://doi.org/10.1016/j.aquaeng.2013.04.006
Sowers A, Young SP, Isely JJ, Browdy CL, Tomasso JR (2004) Nitrite toxicity to Litopenaeus vannamei in water containing low concentrations of sea salt or mixed salts. J World Aquacult Soc 35(4):445–451. https://doi.org/10.1111/j.1749-7345.2004.tb00109.x
Tomasso JR (2012) Environmental nitrite and aquaculture: a perspective. Aquacult Int 20:1107–1116. https://doi.org/10.1007/s10499-012-9532-6
Valencia-Castañeda G, Frías-Espericueta MG, Vanegas-Pérez RC, Pérez-Ramírez JA, Chávez-Sánchez MC, Páez-Osuna F (2018) Acute toxicity of ammonia, nitrite and nitrate to shrimp Litopenaeus vannamei postlarvae in low-salinity water. Bull Environ Contam Toxicol 101(2):229–234. https://doi.org/10.1007/s00128-018-2355-z
Valencia-Castañeda G, Frías-Espericueta MG, Vanegas-Pérez RC, Chávez-Sánchez MC, Páez-Osuna F (2019) Toxicity of ammonia, nitrite and nitrate to Litopenaeus vannamei juveniles in low-salinity water in single and ternary exposure experiments and their environmental implications. Environ Toxicol Pharmacol 70:103–193. https://doi.org/10.1016/j.etap.2019.05.002
Van Wyk P, Davis-Hodgkins M, Laramore R, Main KL, Mounta J, Scarpa J (1999) Farming marine shrimp in recirculating freshwater systems. Florida Department of Agriculture and Consumer Services – Harbor Branch Oceanographic Institution, Florida
Venero JA, McAbee B, Lawson A, Lewis BL, Stokes AD, Leffer JW, Browdy CL (2009) Greenhouse-enclosed superintensive shrimp production: alternative to traditional ponds in U.S. Responsible Seafood Advocate. https://www.globalseafood.org/advocate/greenhouse-enclosed-superintensive-shrimp-production/. Accessed 14 july 2023
Wickham H (2016) Ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag, New York
Xu W-J, Morris TC, Samocha TM (2016) Effects of C/N ratio on biofloc development, water quality, and performance of Litopenaeus vannamei juveniles in a biofloc-based, high-density, zero-exchange, outdoor tank system. Aquaculture 453:169–175. https://doi.org/10.1016/j.aquaculture.2015.11.021
Xu S, Chen M, Feng T, Zhan L, Zhou L, Yu G (2021) Use ggbreak to effectively utilize plotting space to deal with large datasets and outliers. Front Genet 12:774846. https://doi.org/10.3389/fgene.2021.774846
Zacarias S, Schveitzer R, Arantes R, Galasso H, Pinheiro I, Espirito Santo C, Vinatea L (2019) Effect of different concentrations of potassium and magnesium on performance of Litopenaeus vannamei postlarvae reared in low-salinity water and a biofloc system. J Appl Aquac 31(1):85–96. https://doi.org/10.1080/10454438.2018.1536009
Funding
The Federal Rural University of Pernambuco (Pró-Reitoria de Pesquisa e Pós-Graduação—PRPPG 015/2018); the Brazilian National Council for Scientific and Technological Development (CNPq) (PQ 308063/2019–8; PQ309669/2021–9); the Coordination for the Improvement of Higher Education Personnel (CAPES) (88882.436207/2019–01; 88882.436206/2019–01; 88882.436231/2019–01); and the Brazilian Agricultural Research Corporation (EMBRAPA) (21188.005941/2018–04).
Author information
Authors and Affiliations
Contributions
Otávio Augusto L. F. Pimentel: conceptualization, data curation, investigation, methodology, formal analysis, visualization, and writing—original draft. Valdemir Queiroz de Oliveira and Caio Rubens do Rêgo Oliveira: conceptualization, data curation, investigation, methodology, visualization, and writing—review and editing. Elizabeth Pereira dos Santos: data curation, visualization, and writing—review and editing. William Severi: resources and writing—review and editing. Jesus Malpartida Pasco: writing—review and editing. Mauricio G. C. Emerenciano: visualization, and writing—review and editing. Alfredo Olivera Gálvez: resources and writing—review and editing. Luis Otavio Brito: conceptualization, investigation, methodology, funding acquisition, project administration, resources, supervision, visualization, and writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The research undertaken complies with the current animal welfare laws in Brazil. Penaeus vannamei used in this experimental work does not need approval from the Ethics Committee for Animal Use in Brazil. All the authors agree to participate in this experiment.
Human and animal ethics
The authors followed international and institutional animal management guidelines for the experiments.
Consent for publication
All the authors of this article agree to the publication.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pimentel, O.A.L.F., de Oliveira, V.Q., do Rêgo Oliveira, C.R. et al. Nitrogenous compounds and alkalinity patterns in Penaeus vannamei nurseries and pre-grow-out with low salinity water and synbiotic system: a case study. Aquacult Int 32, 1703–1718 (2024). https://doi.org/10.1007/s10499-023-01237-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01237-x