Abstract
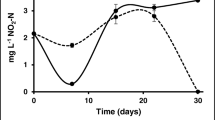
The objective of the present work was to determine the influence of artificial substrates that increase the area for biofilm development on the production performance of Farfantepenaeus paulensis juveniles in pens. Shrimp were stocked at a density of 20/m2 in pen structures (bottom area = 50 m2) that were installed in the Patos Lagoon estuary. Two treatments with three repetitions were analyzed, where artificial substrates (polyethylene nets—1-mm mesh size) were added to increase the area for biofilm development by 100%, and where no substrates were added. During the experimental period, the biomass and the composition of the biofilm were assessed. After 86 days of rearing, no significant differences were found in shrimp performance between the treatments (p > 0.05). However, the examination of the chlorophyll a, dry weight, and composition of the biofilm indicated that the shrimp were actively consuming the biofilm attached to the artificial substrates. Significant decreases in the abundances of nematodes > 500 μm after the 56th day and of tintinnids and rotifers between day 28 and day 42, indicated that the shrimp were selectively predating on these organisms. Moreover, a decrease in the chlorophyll a concentration in the biofilm suggests that the shrimp were consuming the microalgae. Although the increase in the area for biofilm development did not improve shrimp performance, the shrimp presented the highest growth rates when they consumed most of the biofilm microorganisms.






Similar content being viewed by others
References
Abreu PC, Ballester ELC, Odebrecht C, Wasielesky W Jr, Cavalli RO, Granéli W, Anésio AM (2007) Importance of biofilm as food source for shrimp (Farfantepenaeus paulensis) evaluated by stable isotopes (d13C and d15N). J Exp Mar Biol Ecol 347(1-2):88–96. https://doi.org/10.1016/j.jembe.2007.03.012
Akiyama D, Dominy WG, Lawrence AL (1992) Penaeid shrimp nutrition. In: Fast AW, Lester LJ (eds) Marine shrimp culture: principles and practices. Elsevier, Amsterdam, pp 535–568. https://doi.org/10.1016/B978-0-444-88606-4.50031-X
Allan GF, Maguire GB (1992) Effects of stocking density on production of Penaeus monodon Fabricius in model farming ponds. Aquaculture 107(1):49–66. https://doi.org/10.1016/0044-8486(92)90049-Q
Allan GF, Moriarty DJW, Maguire GB (1995) Effects of pond preparation and feeding rate on production of Penaeus monodon Fabricius, water quality, bacteria and benthos in model farming ponds. Aquaculture 130(4):329–349. https://doi.org/10.1016/0044-8486(94)00316-G
Anderson RK, Parker PL, Lawrence AL (1987) A 13C/ 12C tracer study of the utilization of presented feed by commercially important shrimp Penaeus vannamei in a pond growout system. J World Aquacult Soc 18(3):148–155. https://doi.org/10.1111/j.1749-7345.1987.tb00433.x
Ballester ELC, Wasielesky W Jr, Cavalli RO, Abreu PC (2007) Nursery of the pink shrimp Farfantepenaeus paulensis in cages with artificial substrates: biofilm composition and shrimp performance. Aquaculture 265(1-4):355–362. https://doi.org/10.1016/j.aquaculture.2007.04.003
Bratvold D, Browdy CL (2001) Effects of sand sediment and vertical surfaces (AquaMats™) on production, water quality, and microbial ecology in an intensive Litopenaeus vannamei culture system. Aquaculture 195(1-2):81–94. https://doi.org/10.1016/S0044-8486(00)00538-X
Costa RC, Lopes M, Castilho AL, Fransozo A, Simões SM (2008) Abundance and distribution of juvenile pink shrimps Farfantepenaeus spp. in a mangrove estuary and adjacent bay on the northern shore of São Paulo state, southeastern Brazil. Invertebr Reprod Dev 52(1-2):51–58. https://doi.org/10.1080/07924259.2008.9652272
Cuzon G, Lawrence A, Gaxiola G, Guillaume J (2004) Nutrition of Litopenaeus vannamei reared in pond or in tanks. Aquaculture 235(1-4):513–551. https://doi.org/10.1016/j.aquaculture.2003.12.022
D'Incao F (1991) Pesca e biologia de Penaeus paulensis na Lagoa dos Patos, RS. Atlantica 13:159–169 [In Portuguese with English abstract]
D’Incao F, Reis EG (2002) Community-based management and technical advice in Patos Lagoon estuary (Brazil). Ocean Coast Manag 45(8):531–539. https://doi.org/10.1016/S0964-5691(02)00084-4
Dutra FM, Moretto Y, Portz L, Ballester ELC (2016) Pen culture of Macrobrachium amazonicum: use of artificial diet and impact on benthic community. Aquac Res 47(1):266–275. https://doi.org/10.1111/are.12488
EPP MA (2002) Stable isotopes in shrimp aquaculture. World Aquaculture 33:18–19
Fast AW, Lester LJ (1992) Marine shrimp culture: principles and practices. Elsevier, Amsterdam
Jeffrey SW, Humphrey GF (1975) New espectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem Physiol Pflanz 167(2):191–194. https://doi.org/10.1016/S0015-3796(17)30778-3
Jensen L, Domingos JAS, Peixoto S, Santos MHS, Cavalli RO, Poersch LH, Ballester ELC, Wasielesky WJ (2016) Cultivo do camarão rosa em cercados: produção e viabilidade econômica. In: WJr W, Poersch LH (eds) Cultivo de camarões em gaiolas e cercados no estuário da Lagoa dos Patos. Editora da FURG, Rio Grande, pp 150–165
Jensen LV, Wasielesky W Jr, Cavalli RO, Peixoto S, Santos MHS, Ballester ELC (2004) Growth and survival of pink shrimp (Farfantepenaeus paulensis) postlarvae in cages and pen enclosures. Sci Agric 61(3):332–335. https://doi.org/10.1590/S0103-90162004000300016
Jensen LV, Wasielesky W Jr, Ballester ELC, Cavalli RO, Santos MS (2006) Role of the microalgae Thalassiosira fluviatilis in weight gain and survival of the shrimp Farfantepenaeus paulensis reared in indoor nursery tanks. Nauplius 14:37–43
Komárek J, Anagnostidis K (2005) Cyanoprokaryota. In: Büdel B, Gärtner G, Krienitz L, Scagerl M (eds) Süsswasseflora von Mitteleuropa 19/2. Elsevier, Spektrum, Heidelberg, p 759
Krumenauer D, Cavalli RO, Ballester ELC, Wasielesky W Jr (2010) Feasibility of pacific white shrimp Litopenaeus vannamei culture in Southern Brazil: effects of stocking density and single or double crop management strategy in earthen ponds. Aquac Res 41(2):240–248. https://doi.org/10.1111/j.1365-2109.2009.02326.x
Krummenauer D, Zogbi PR, Cavalli RO, Peixoto S, Wasielesky WJ (2016) Análise da viabilidade do cultivo do camarão rosa Farfantepenaeus paulensis em gaiolas em diferentes densidades de estocagem durante o outono no extremo sul do Brasil. In: Wasielesky W Jr, Poersch LH (eds) . Cultivo de camarões em gaiolas e cercados no estuário da Lagoa dos Patos. Editora da FURG, Rio Grande, pp 166–173
Kumlu M, Eroldogan OT, Saglatimur B (2001) The effects of salinity and added substrates on growth and survival of Metapenaeus monoceros (Decapoda: Penaeidae) post-larvae. Aquaculture 196(1-2):177–188. https://doi.org/10.1016/S0044-8486(00)00580-9
Moss KRK, Moss SM (2004) Effects of artificial substrate and stocking density on the nursery production of pacific white shrimp Litopenaeus vannamei. J World Aquacult Soc 35(4):537–542. https://doi.org/10.1111/j.1749-7345.2004.tb00121.x
Nunes AJP, Gesteira TCV, Goddard S (1997) Food ingestion and assimilation by the southern brown shrimp Penaeus subtilis under semi-intensive culture in NE Brazil. Aquaculture 149(1-2):121–136. https://doi.org/10.1016/S0044-8486(96)01433-0
Ostrensky A, Wasielesky W Jr (1995) Acute toxicity of ammonia to various life stages of the São Paulo shrimp, Penaeus paulensis Pérez-Farfante, 1967. Aquaculture 132(3-4):339–347. https://doi.org/10.1016/0044-8486(94)00343-M
Otoshi CA, Montgomery AD, Matsuda EM, Moss SM (2006) Effects of artificial substrate and water source on growth of juvenile pacific white shrimp, Litopenaeus vannamei. J World Aquacult Soc 37(2):210–213. https://doi.org/10.1111/j.1749-7345.2006.00029.x
Peixoto S, Wasielesky W Jr, Louzada L (2003) Comparative analysis of pink shrimp, Farfantepenaeus paulensis, and Pacific White shrimp, Litopenaeus vannamei, culture in extreme southern Brazil. Journal of Applied Aquiculture 14(1-2):47–56. https://doi.org/10.1300/J028v14n01_07
Poersch L, Castello JP, Wasielesky W Jr, Cavalli RO (2007) The challenge of sustainable aquaculture: effects on the environment of the Patos Lagoon estuary. J Coast Res 47:130–135. https://doi.org/10.2112/1551-5036-47.sp1.130
Porter KG, Feig YS (1980) The use of DAPI for identifying and counting aquatic microflora. Limnol Oceanogr 25(5):943–948. https://doi.org/10.4319/lo.1980.25.5.0943
Preto AL, Cavalli RO, Pissetti T, Abreu PC, Wasielesky W Jr (2005) Efeito da densidade de estocagem sobre o biofilme e o desempenho de pós-larvas do camarão-rosa Farfantepenaeus paulensis cultivadas em gaiolas. Ciência Rural 35(6):1417–1423. [In Portuguese with English abstract]. https://doi.org/10.1590/S0103-84782005000600030
Quaresma J, Sugai JK (2005) Circadian profile of feed consumption and amylase and maltase activities in the juvenile shrimp Farfantepenaeus paulensis. J World Aquaculture Soc 36(1):141–147. https://doi.org/10.1111/j.1749-7345.2005.tb00141.x
Round FE, Crawford RM, Mann DV (1990) The diatoms: biology and morphology of the genera. Cambridge University Press, Cambridge
Ruas VM, Dumont LF, D’Incao F (2011) Evaluation of the capture size and abundance of pink shrimp Farfantepenaeus paulensis (Pérez Farfante 1967) around the Marinheiros Island. Patos Lagoon Estuary, RS, Brazil, Atlântica 33:161–172 [In Portuguese with English abstract]
Samocha TM, Lawrence AL, Biedenbach JM (1993) The effect of vertical netting and water circulation pattern on growth and survival of Penaeus vannamei postlarvae in an intensive raceway system. J Appl Aquiculture 2(1):55–64. https://doi.org/10.1300/J028v02n01_04
Schlechtriem C, Ricci M, Focken U, Becker K (2004) Mass produced nematodes Panagrellus redivivus as live food for rearing carp larvae: preliminary results. Aquac Res 35(6):547–551. https://doi.org/10.1111/j.1365-2109.2004.01049.x
Silva CF, Ballester ELC, Monserrat J, Geracitano L, Wasielesky W Jr, Abreu PC (2008) Contribution of microorganisms to the biofilm nutritional quality: protein and lipid contents. Aquac Nutr 14(6):507–514. https://doi.org/10.1111/j.1365-2095.2007.00556.x
Snedecor G, Cochran WG (1982) Statistical Methods, 7th ed. State University Press, Ames - Iowa, USA
Soares R, Peixoto S, Bemvenuti C, Wasielesky W Jr, D’Incao F, Murcia N, Suita S (2004) Composition and abundance of invertebrate benthic fauna in Farfantepenaeus paulensis culture pens (Patos Lagoon estuary, Southern Brazil). Aquaculture 239(1-4):199–215. https://doi.org/10.1016/j.aquaculture.2004.05.041
Soares R, Peixoto S, Wasielesky W Jr, D’Incao F (2005) Feeding rhythms and diet of Farfantepenaeus paulensis under pen culturein Patos Lagoon estuary, Brazil. J Exp Mar Biol Ecol 322(2):167–176. https://doi.org/10.1016/j.jembe.2005.02.019
Sokal RR, Rohlf FJ (1969) Biometry: principle and practices of statistics in biological research. W. H. Freeman and Company, New York, USA
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis. Journal of the Fisheries Research Board of Canada, Ottawa
Thompson FL, Abreu PC, Wasielesky W Jr (2002) Importance of biofilm for water quality and nourishment in intensive shrimp culture. Aquaculture 203(3-4):263–278. https://doi.org/10.1016/S0044-8486(01)00642-1
UNESCO - United Nations Educational, Scientific and Cultural Organization (1983) Manuals and guides 12: chemical methods for use in marine environmental monitoring, UNESCO, Paris, http://www.jodc.go.jp/info/ioc_doc/Manual/055950eo.pdf. Cited 10 nov 2016
Utermöhl H (1958) Zur Vervollkommnung der quantitativen Phytoplankton Methodik. Verhandlungen der Internationalen Vereinigung fur Theoretische und Angewandte Limnologi 9:1–38
Van Wyk P, Scarpa J (1999) Water quality requirements and management. In: Van Wyk P (ed) Farming marine shrimp in recirculating freshwater systems, Florida Department of Agriculture and Consumer Services. Tallahasee - Florida, USA, pp 128–138
Wasielesky W Jr, Abreu PC, Poersh LH, Thompson F, Ballester ELC (2011) Influence of light intensity on biofilm formation and the performance of pink shrimp Farfantepenaeus paulensis juveniles reared in cages. Aquac Res 43(5):706–712. https://doi.org/10.1111/j.1365-2109.2011.02878.x
Wasielesky W Jr, Peixoto S, Jensen L, Poersch L, Bianchini A (2004) Estudo preliminar do cultivo do camarão-rosa Farfantepenaeus paulensis em cercados no estuário da Lagoa dos Patos. Bol Inst Pesca 30:63–70 [In Portuguese with English abstract]
Wasielesky W Jr, Peixoto S, Santos MHS, Cavalli RO (2002) Cultivo do camarão-rosa como alternativa de geração de renda. In: Alderón AI, Sampaio H (eds) Extensão universitária: ação comunitária em Universidades Brasileiras. Olho D'Água, São Paulo, Brasil, pp 17–27
Wasielesky W Jr, Poersh LH, Jensen L, Bianchini A (2001) Effect of stocking density on pen reared pink shrimp Farfantepenaeus paulensis (Pérez-Farfante, 1967) (Decapoda, Penaeidae). Nauplius 9:163–167
Zhukova NV, Kharlamenko VI (1999) Sources of essential fatty acids in the marine microbial loop. Aquat Microb Ecol 17:153–157. https://doi.org/10.3354/ame017153
Acknowledgements
The authors would like to thank the researchers Clarisse Odebrecht, Marli Bergesh, and Taciana Cramer for their assistance in the identification of diatoms, cyanobacteria, and nematodes, respectively. Eduardo Luis Cupertino Ballester, Wilson Wasielesky Jr., Ronaldo O. Cavalli, and Paulo César Abreu are research fellows of CNPq (National Council for the Development of Science and Technology of Brazil), and Tito Luis Pisseti received a CNPq Master’s scholarship during the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ballester, E.L.C., Dutra, F.M., Pisseti, T.L. et al. Influence of biofilm on the production of Farfantepenaeus paulensis in pens in the Patos Lagoon estuary. Aquacult Int 26, 713–726 (2018). https://doi.org/10.1007/s10499-018-0246-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-018-0246-2