Abstract
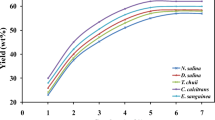
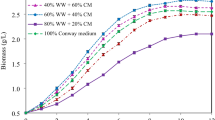
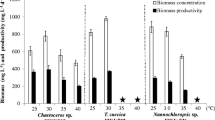
Marine microalgae are used in aquaculture as a suspension to feed young fishes and shrimps. Marine Chlorella sp. is one of the most attractive marine microalgae because in addition to pigments it can accumulate lipids at a high content (>30 %) and release exopolymeric substances (EPSs) into the culture medium. This study aimed to evaluate the optimal cultivation of marine Chlorella sp. as potential sources of lipids, EPSs and pigments. Among the culturing regimes tested, mixotrophic cultivation mode produced the highest yields of biomass, lipids and EPSs. Several factors affecting mixotrophic cultivation were optimized. An increase in light intensity up to 65 μmol m−2 s−1 and carbon dioxide up to 10 % v/v in air enhanced both biomass and product formation. An increase in glucose concentration up to 1 % w/v enhanced biomass and EPS yields but decreased lipid and chlorophyll contents. A semi-continuous cultivation under optimal conditions produced microalgal biomass of 2.76 g L−1 with a high lipid content of 44.9 % and EPS of 1.46 g L−1. This study has also shown that the microalgal lipid and EPS have potential to be used as biodiesel feedstocks and bioflocculants, respectively. The concomitant production of these valuable products together with the microalgal biomass would be a potential way to offset the production cost and contribute greatly to developing industrialized microalgae cultivation.





Similar content being viewed by others
References
Becker EW (1994) Microalgae: biotechnology and microbiology. Cambridge University Press, New York
Bermúdez J, Rosales N, Loreto C, Briceño B, Morales E (2004) Exopolysaccharide, pigment and protein production by the marine microalga Chroomonas sp. in semicontinuous cultures. World J Microbiol Biotechnol 20:179–183
Buckwalter P, Embaye T, Gormly S, Trent JD (2013) Dewatering microalgae by forward osmosis. Desalination 312:19–22
Cheirsilp B, Torpee S (2012) Enhanced growth and lipid production of microalgae under mixotrophic culture condition: effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour Technol 110:510–516
Chen L, Li P, Liu Z, Jiao Q (2009) The released polysaccharide of the cyanobacterium Aphanothece halophytica inhibits flocculation of the microalga with ferric chloride. J Appl Phycol 21:327–331
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306
Chiu SY, Kao CY, Chen CH, Kuan TC, Ong SC, Lin CS (2008) Reduction of CO2 by a high-density culture of Chlorella sp. in a semicontinuous photoreactor. Bioresour Technol 99:3389–3396
Danesi EDG, Rangel-Yagui CO, Sato S, de Carvalho JCM (2011) Growth and content of Spirulina platensis biomass chlorophyll cultivated at different values of light intensity and temperature using different nitrogen sources. Brazil J Microbiol 42(1):362–373
Dayananda C, Sarada R, Rani MU, Shamala TR, Ravishankar GA (2007) Autotrophic cultivation of Botryococcus braunii for the production of hydrocarbons and exopolysaccharides in various media. Biomass Bioenergy 31:87–93
de Oliveira MACL, Monteiro MPC, Robbs PG, Leite SGF (1999) Growth and chemical composition of Spirulina maxima and Spirulina platensis biomass at different temperatures. Aquacult Int 7(4):261–275
Duan J, Gregory J (2003) Coagulation by hydrolysing metal salts. Adv Colloid Interface Sci 100–102:475–502
Fagiri YMA, Salleh A, El-Nagerabi SAF (2013) Influence of chemical and environmental factors on the growth performance of Spirulina platensis strain SZ100. J Algal Biomass Utilization 4(2):7–15
Ferruzzi MG, Blakeslee J (2007) Digestion, absorption, and cancer preventative activity of dietary chlorophyll derivatives. Nutr Res 27(1):1–12
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Frumento D, Casazza AA, Al Arni S, Converti A (2013) Cultivation of Chlorella vulgaris in tubular photobioreactors: a lipid source for biodiesel production. Biochem Eng J 81:120–125
Girard JM, Roy ML, Hafsa MB, Gagnon J, Faucheux N, Heitz M, Tremblay R, Deschênes JS (2014) Mixotrophic cultivation of green microalgae Scenedesmus obliquus on cheese whey permeate for biodiesel production. Algal Res 5:241–248
Goo BG, Baek G, Choi DJ, Park YI, Synytsya A, Bleha R, Seong DH, Lee CG, Park JK (2013) Characterization of a renewable extracellular polysaccharide from defatted microalgae Dunaliella tertiolecta. Bioresour Technol 129:343–350
Gopinath A, Puhan S, Nagarajan G (2009) Theoretical modeling of iodine value and saponification value of biodiesel fuels from their fatty acid composition. Renew Energy 34:1806–1811
Gudin C, Therpenier C (1986) Bioconversion of solar energy into organic chemicals by microalgae. Adv Biotechnol Process 6:73–110
Haass D, Tanner W (1974) Regulation of hexose transport in Chlorella vulgaris. Plant Physiol 53:291–302
Heredia-Arroyo T, Wei W, Hu B (2010) Oil accumulation via heterotrophic/mixotrophic Chlorella protothecoides. Appl Biochem Biotechnol 162:1978–1995
Hossain ABM, Salleh A, Boyce AN, Chowdhurry P, Naqiuddin M (2008) Biodiesel fuel production from algae as renewable energy. Am J Biochem Biotechnol 4:250–254
Jham GN, Teles FFF, Campos LG (1982) Use of aqueous HCl/MeOH as esterification reagent for analysis of fatty acids derived from soybean lipids. J Am Oil Chem Soc 59:132–133
Krisnangkura K (1986) A simple method for estimation of Cetane index of vegetable oil methyl esters. J Am Oil Chem Soc 63:552–553
Lee YK (2004) Algal nutrition. Heterotrophic carbon nutrition. In: Richmond A (ed) Handbook of microalgal culture. Biotechnology and applied phycology. Blackwell Scientific Publications Ltd, Oxford, p 116
Liang YN, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol Lett 31:1043–1049
Liu J, Tao Y, Wu J, Zhu Y, Gao B, Tang Y, Li A, Zhang C, Zhang Y (2014) Effective flocculation of target microalgae with self-flocculating microalgae induced by pH decrease. Bioresour Technol 167:367–375
Lizzul AM, Hellier P, Purton S, Baganz F, Ladommatos N, Campos L (2014) Combined remediation and lipid production using Chlorella sorokiniana grown on wastewater and exhaust gases. Bioresour Technol 151:12–18
Ma L, Dolphin D (1999) The metabolites of dietary chlorophylls. Phytochem 50:195–202
Martínez ME, Camacho F, Jiménez JM, Espínola JB (1997) Influence of light intensity on the kinetic and yield parameters of Chlorella pyrenoidosa mixotrophic growth. Process Biochem 32:93–98
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications, a review. Renew Sust Energy Rev 14:217–232
Metzger P, Largeau C (1999) Chemicals of Botryococcus braunii. In: Cohen Z (ed) Chemicals from microalgae. Taylor and Francis, London, pp 205–260
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–429
Qin J (2005) Bio-hydrocarbons from algae, impacts of temperature, light and salinity on algae growth. A report for the rural industries research and development corporation. Australia
Richmond A (1986) Handbook of microalgal mass culture. CRC Press Inc., Boca Raton
Shelef G, Sukenik A, Green M (1984) Microalgae harvesting and processing: a literature review. Technion Research and Development Foundation Ltd., Haifa
Sterin JR (1973) Handbook of phycological methods, culture methods and growth measurements. Cambridge University Press, Cambridge
Suarez ER, Kralovec JA, Noseda MD, Ewart HS, Barrow CJ, Lumsden MD, Grindley TB (2005) Isolation, characterization and structural determination of a unique type of arabinogalactan from an immunostimulatory extract of Chlorella pyrenoidosa. Carbohydr Res 340:1489–1498
Sudhir P, Murthy SDS (2004) Effect of salt stress on basic processes of phytosynthesis. Photosynthetica 42:481–486
Tansakul P, Savaddiraksa Y, Prasertsan P, Tongurai C (2005) Cultivation of the hydrocarbon-rich alga, Botyococcus braunii in secondary treated effluent from a sea food processing plant. Thai J Agri Sci 38(1–2):71–76
Vandamme D, Foubert I, Meesschaert B, Muylaert K (2010) Flocculation of microalgae using cationic starch. J Appl Phycol 22:525–530
Villay A, Laroche C, Roriz D, Alaoui HE, Delbac F, Michaud P (2013) Optimisation of culture parameters for exopolysaccharides production by the microalga Rhodella violacea. Bioresour Technol 146:732–735
Wang SK, Hu YR, Wang F, Stiles AR, Liu CZ (2014) Scale-up cultivation of Chlorella ellipsoidea from indoor to outdoor in bubble column bioreactors. Bioresour Technol 156:117–122
Wu Z, Zhu Y, Huang W, Zhang C, Li T, Zhang Y, Li A (2012) Evaluation of flocculation induced by pH increase for harvesting microalgae and reuse of flocculated medium. Bioresour Technol 110:496–502
Yeesang C, Chanthachum S, Cheirsilp B (2008) Sago starch as a low-cost carbon source for exopolysaccharide production by Lactobacillus kefiranofaciens. World J Microbiol Biotechnol 24(7):1195–1201
You T, Barnett SM (2004) Effect of light quality on production of extracellular polysaccharides and growth rate of Porphyridium cruentum. Biochem Eng J 19:251–258
Acknowledgments
The authors are grateful to the financial support by Prince of Songkla University in the fiscal year of 2012 under Grant AGR550135S. The first and third authors are also supported by Thailand Research Fund under Grant No. RTA5780002. Thanks are also given to Dr. Brian Hodgson Faculty of Pharmaceutical Science, Prince of Songkla University, for assistance with the English.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheirsilp, B., Mandik, Y.I. & Prasertsan, P. Evaluation of optimal conditions for cultivation of marine Chlorella sp. as potential sources of lipids, exopolymeric substances and pigments. Aquacult Int 24, 313–326 (2016). https://doi.org/10.1007/s10499-015-9927-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-015-9927-2