Abstract
Understanding the identity and stability of the hydrolysis products of metals is required in order to predict their behavior in natural aquatic systems. Despite this need, the hydrolysis constants of many metals are only known over a limited range of temperature and ionic strengths. In this paper, we show that the hydrolysis constants of 31 metals [i.e. Mn(II), Cr(III), U(IV), Pu(IV)] are nearly linearly related to the values for Al(III) over a wide range of temperatures and ionic strengths. These linear correlations allow one to make reasonable estimates for the hydrolysis constants of +2, +3, and +4 metals from 0 to 300°C in dilute solutions and 0 to 100°C to 5 m in NaCl solutions. These correlations in pure water are related to the differences between the free energies of the free ion and complexes being almost equal
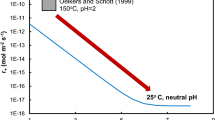
The correlation at higher temperatures is a result of a similar relationship between the enthalpies of the free ions and complexes
The correlations at higher ionic strengths are the result of the ratio of the activity coefficients for Al(III) being almost equal to that of the metal.
The results of this study should be useful in examining the speciation of metals as a function of pH in natural waters (e.g. hydrothermal fresh waters and NaCl brines).




Similar content being viewed by others
References
Baes CF Jr, Mesmer RE (1976) The hydrolysis of cations. Wiley, New York
Benézéth P, Palmer DA, Wesolowski DJ (2001) Aqueous high-temperature solubility studies. II. The solubility of Boehmite at 0.03 m ionic strength as a function of temperature and pH as determined by in situ measurements. Geochim Cosmochim Acta 65:2097–2111
Bourcier WL, Knauss KG, Jackson KJ (1993) Aluminum hydrolysis constants to 250°C from boehmite solubility measurements. Geochim Cosmochim Acta 57:747–762
Castet S, Dandurand JL, Schott J, Gout R (1993) Boehmite solubility and aqueous aluminum speciation in hydrothermal solutions (90–350°). Experimental study and modelling. Geochim Cosmochim Acta 57:4869–4884
Choppin GR, Bond AH, Hromadka PM (1997) Redox speciation of plutonium. J Radioanal Nuclear Chem 219:203–210
Couturier Y, Michard G, Sarazin G (1984) Constantes de formation des complexes hydroxydés de l’aluminum en solution aqueuse de 20 et 70°C. Geochim Cosmochim Acta 48:649–659
Ekberg C, Albinsson Y, Comarmound MJ, Brown PL (2000) Studies on the complexation behavior of Thorium(IV). 1. Hydrolysis equilibria. J Soln Chem 29:63–86
Ekberg C, Källvenius G, Albinsson Y, Brown PL (2004) Studies on the hydrolytic behavior of zirconium(IV). J Soln Chem 33:47–79
Frink CR, Peech M (1963) Hydrolysis of the aluminum ion in dilute solutions. Inorg Chem 2:473–478
Klungness GD, Byrne RH (2000) Comparative hydrolysis behavior of the rare earths and yttrium: the influence of temperature and ionic strength. Polyhedron 19:99–107
Manfredi C, Caruso V, Vasca E, Vero S, Ventimiglia E, Pallidino G, Ferri D (2006) On the hydrolysis of tetravalent uranium ion U4+. J Soln Chem 35:927–937
Millero FJ, Pierrot D (2007) The activity coefficients of Fe(III) complexes with hydroxide in NaCl and NaClO4 solutions. Geochim Cosmochim Acta 71:4825–4833
Millero FJ, Woosley RJ (2009) The hydrolysis of Al(III) in NaCl solutions—a model for Fe(III). Environ Sci Technol 43(6):1818–1823
Neck V, Kim JI (2001) Solubility and hydrolysis of tetravalent actinides. Radiochem Acta 89:1–16
Palmer DA, Wesolowski DJ (1992) Aluminum speciation and equilibria in aqueous solution: II. The solubility of gibbsite in acidic sodium chloride solutions from 30 to 70°C. Geochim Cosmochim Acta 56:1092–1111
Palmer DA, Wesolowski DJ (1993) Aluminum speciation and equilibria in aqueous solution: III. Potentiometric determination of the first hydrolysis constant of aluminum (III) in sodium chloride solutions to 125°C. Geochim Cosmochim Acta 57:2929–2938
Paulson AJ, Kester DR (1980) Copper(II) ion hydrolysis in aqueous solution. J Solution Chem 9:269–277
Pivovarov S (2005) Modeling of ionic equilibria of trace metals (Cu2+, Zn2+, Cd2+) in concentrated aqueous electrolyte solutions at 25°C. J Colloid Interface Sci 291:421–432
Rai D, Xia Y, Hess NJ, Strachan DM, McGrail BP (2001) Hydroxo and chloro complexes/Ion interactions of Hf4+ and the solubility product of HfO2(am). J Solution Chem 30:949–967
Schofield RK, Taylor AW (1954) The hydrolysis of aluminum salt solutions. J Chem Soc 4445–4448
Tarapcik P, Fourest B, Giffaut E (2005) Comparative approach of the solubility of protactinium oxy/hydroxides. Radiochem Acta 93:27–33
Verdes G, Gout R, Castet S (1992) Thermodynamic properties of aluminate ion and of bayerite, boehmite, diaspore and gibbsite. Eur J Mineral 4:767–792
Wesolowski DJ (1992) Aluminum speciation and equilibria in aqueous solution: I. The solubility of gibbsite in the system Na-K-Cl-OH-Al(OH)4 from 0 to 100°C. Geochim Cosmochim Acta 56:1065–1091
Wesolowski DJ, Palmer DA (1994) Aluminum speciation and equilibria in aqueous solutions: V. Gibbsite solubility at 50°C and pH 3–9 in 1 molal NaCl solutions (a general model for aluminum speciation; analytical methods). Geochim Cosmochim Acta 58:2947–2969
Zakanova-Herzog VP, Seward TM, Sulemenov OM (2006) Arsenous acid ionization in aqueous solutions from 25 to 300°C. Geochim Cosmochim Acta 70:1928–1938
Acknowledgments
The authors acknowledge the support of the Oceanographic Section of the National Science Foundation and the National Oceanic and Atmospheric Administration for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woosley, R.J., Millero, F.J. The Hydrolysis of Al(III) in NaCl solutions: A Model for M(II), M(III), and M(IV) Ions. Aquat Geochem 16, 317–324 (2010). https://doi.org/10.1007/s10498-009-9075-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10498-009-9075-2