Abstract
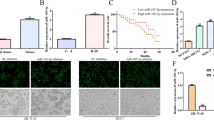
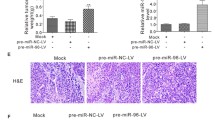
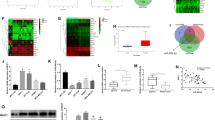
Breast cancer is a common malignancy in women with poor prognosis. This study aimed to investigate the molecular mechanism of microRNA-944 (miR-944) mediated secreted phosphoprotein-1 (SPP1) in breast cancer progression and its regulatory effect on the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway. Differential gene analysis was performed to identify key genes associated with breast cancer development by screening breast cancer-related microarray data. The expression of miR-944 and SPP1 and their relationship were determined in clinical samples and cells. sh-SPP1, oe-SPP1, LY294002 or miR-944 mimic were transfected into MCF-7 cells to investigate the role of miR-944 mediated SPP1 in breast cancer development and its regulatory effect on the PI3K/Akt pathway. Finally, the tumorigenicity of breast cancer cells was observed in nude mice. Through bioinformatics analysis, we identified SPP1 as a key gene in breast cancer, and miR-944 as an upstream miRNA of SPP1. In breast cancer tissues and cells, the expression of miR-944 was decreased while that of SPP1 was increased. miR-944 negatively regulated the expression of SPP1. In breast cancer cells, SPP1 activated the PI3K/Akt pathway to promote cell proliferation and inhibit apoptosis. In vitro cell experiments showed that the downregulation of miR-944 promoted the high expression of SPP1, which then activated the PI3K/Akt signaling pathway, promoting breast cancer cell proliferation. In vivo experiments further confirmed the anti-cancer role of miR-944 mediated SPP1 in breast cancer. Our study highlights the role of miR-944 mediated SPP1 in inhibiting breast cancer progression by blocking the PI3K/Akt pathway.









Similar content being viewed by others
Data Availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Abbreviations
- SPP1:
-
Secreted phosphoprotein-1
- PI3K:
-
Phosphoinositide 3-kinase
- OS:
-
Overall survival, BRCA, breast cancer, DEGs, differentially expressed genes
- PPI:
-
Protein-protein interaction
- GSEA:
-
Gene set enrichment analysis
References
Liang Y, Zhang H, Song X, Yang Q (2020) Metastatic heterogeneity of breast cancer: molecular mechanism and potential therapeutic targets. Semin Cancer Biol 60:14–27. https://doi.org/10.1016/j.semcancer.2019.08.012
Medeiros B, Allan AL (2019) Molecular mechanisms of breast Cancer metastasis to the lung: clinical and experimental perspectives. Int J Mol Sci 20:2272. https://doi.org/10.3390/ijms20092272
Yang F, Xu J, Tang L, Guan X (2017) Breast cancer stem cell: the roles and therapeutic implications. Cell Mol Life Sci 74:951–966. https://doi.org/10.1007/s00018-016-2334-7
Hasanpourghadi M, Pandurangan AK, Mustafa MR (2018) Modulation of oncogenic transcription factors by bioactive natural products in breast cancer. Pharmacol Res 128:376–388. https://doi.org/10.1016/j.phrs.2017.09.009
Yim A, Smith C, Brown AM (2022) Osteopontin/secreted phosphoprotein-1 harnesses glial-, immune-, and neuronal cell ligand-receptor interactions to sense and regulate acute and chronic neuroinflammation. Immunol Rev 311:224–233. https://doi.org/10.1111/imr.13081
Liang L, Lu G, Pan G, Deng Y, Liang J, Liang L, Liu J, Tang Y, Wei G (2019) A case-control study of the Association between the SPP1 gene SNPs and the susceptibility to breast Cancer in Guangxi, China. Front Oncol 9:1415. https://doi.org/10.3389/fonc.2019.01415
Saleh S, Thompson DE, McConkey J, Murray P, Moorehead RA (2016) Osteopontin regulates proliferation, apoptosis, and migration of murine claudin-low mammary tumor cells. BMC Cancer 16:359. https://doi.org/10.1186/s12885-016-2396-9
Christodoulatos GS, Spyrou N, Kadillari J, Psallida S, Dalamaga M (2019) The role of adipokines in breast Cancer: current evidence and perspectives. Curr Obes Rep 8:413–433. https://doi.org/10.1007/s13679-019-00364-y
Dongre A, Rashidian M, Eaton EN, Reinhardt F, Thiru P, Zagorulya M, Nepal S, Banaz T, Martner A, Spranger S, Weinberg RA (2021) Direct and indirect regulators of epithelial-mesenchymal transition-mediated immunosuppression in breast carcinomas. Cancer Discov 11:1286–1305. https://doi.org/10.1158/2159-8290.CD-20-0603
Pompura SL, Dominguez-Villar M (2018) The PI3K/AKT signaling pathway in regulatory T-cell development, stability, and function. J Leukoc Biol. https://doi.org/10.1002/JLB.2MIR0817-349R
Yuen HF, Chan KK, Grills C, Murray JT, Platt-Higgins A, Eldin OS, O’Byrne K, Janne P, Fennell DA, Johnston PG, Rudland PS, El-Tanani M (2012) Ran is a potential therapeutic target for cancer cells with molecular changes associated with activation of the PI3K/Akt/mTORC1 and Ras/MEK/ERK pathways. Clin Cancer Res 18:380–391. https://doi.org/10.1158/1078-0432.CCR-11-2035
Zhang H, Guo M, Chen JH, Wang Z, Du XF, Liu PX, Li WH (2014) Osteopontin knockdown inhibits alphav,beta3 integrin-induced cell migration and invasion and promotes apoptosis of breast cancer cells by inducing autophagy and inactivating the PI3K/Akt/mTOR pathway. Cell Physiol Biochem 33:991–1002. https://doi.org/10.1159/000358670
Haines CN, Klingensmith HD, Komara M, Burd CJ (2020) GREB1 regulates PI3K/Akt signaling to control hormone-sensitive breast cancer proliferation. Carcinogenesis 41:1660–1670. https://doi.org/10.1093/carcin/bgaa096
Shen J, Wang Q, Liang C, Su X, Ke Y, Mao Y, Fang J, Duan S (2022) Novel insights into miR-944 in Cancer. Cancers (Basel) 14:4232. https://doi.org/10.3390/cancers14174232
Fang T, Yan YX, Yang Y, Lv YX, Chang QQ, Zhang DD (2020) Ethyl acetate fraction from Hedyotis diffusa plus Scutellaria barbata suppresses Migration of bone-metastatic breast Cancer cells via OPN-FAK/ERK/NF-kappaB Axis. Evid Based Complement Alternat Med 2020:3573240. https://doi.org/10.1155/2020/3573240
Butti R, Nimma R, Kundu G, Bulbule A, Kumar TVS, Gunasekaran VP, Tomar D, Kumar D, Mane A, Gill SS, Patil T, Weber GF, Kundu GC (2021) Tumor-derived osteopontin drives the resident fibroblast to myofibroblast differentiation through Twist1 to promote breast cancer progression. Oncogene 40:2002–2017. https://doi.org/10.1038/s41388-021-01663-2
Ghanbarnasab Behbahani R, Danyaei A, Teimoori A, Tahmasbi MJ, Neisi N (2022) CRISPR/Cas9 mediated knocking out of OPN gene enhances radiosensitivity in MDA-MB-231 breast cancer cell line. J Cancer Res Clin Oncol. https://doi.org/10.1007/s00432-022-04304-7
Lv W, Tan Y, Xiong M, Zhao C, Wang Y, Wu M, Wu Y, Zhang Q (2021) Analysis and validation of m6A regulatory network: a novel circBACH2/has-miR-944/HNRNPC axis in breast cancer progression. J Transl Med 19:527. https://doi.org/10.1186/s12967-021-03196-4
Hill M, Tran N (2021) miRNA interplay: mechanisms and consequences in cancer. Dis Model Mech 14:dmm047662. https://doi.org/10.1242/dmm.047662
Yu G, Wang LG, Han Y, He QY (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287. https://doi.org/10.1089/omi.2011.0118
Fang T, Lv H, Lv G, Li T, Wang C, Han Q, Yu L, Su B, Guo L, Huang S, Cao D, Tang L, Tang S, Wu M, Yang W, Wang H (2018) Tumor-derived exosomal mir-1247-3p induces cancer-associated fibroblast activation to foster lung metastasis of liver cancer. Nat Commun 9:191. https://doi.org/10.1038/s41467-017-02583-0
Huan J, Hornick NI, Shurtleff MJ, Skinner AM, Goloviznina NA, Roberts CT Jr, Kurre P (2013) RNA trafficking by acute myelogenous leukemia exosomes. Cancer Res 73:918–929. https://doi.org/10.1158/0008-5472.CAN-12-2184
Lin Z, Tian XY, Huang XX, He LL, Xu F (2019) microRNA-186 inhibition of PI3K-AKT pathway via SPP1 inhibits chondrocyte apoptosis in mice with osteoarthritis. J Cell Physiol 234:6042–6053. https://doi.org/10.1002/jcp.27225
Li J, Xu H, Wang Q, Wang S, Xiong N (2019) 14-3-3zeta promotes gliomas cells invasion by regulating snail through the PI3K/AKT signaling. Cancer Med 8:783–794. https://doi.org/10.1002/cam4.1950
Li Z, Yu D, Li H, Lv Y, Li S (2019) Long noncoding RNA UCA1 confers tamoxifen resistance in breast cancer endocrinotherapy through regulation of the EZH2/p21 axis and the PI3K/AKT signaling pathway. Int J Oncol 54:1033–1042. https://doi.org/10.3892/ijo.2019.4679
Januskeviciene I, Petrikaite V (2019) Heterogeneity of breast cancer: the importance of interaction between different tumor cell populations. Life Sci 239:117009. https://doi.org/10.1016/j.lfs.2019.117009
Lindahl G, Rzepecka A, Dabrosin C (2019) Increased extracellular osteopontin levels in normal human breast tissue at high risk of developing Cancer and its Association with inflammatory biomarkers in situ. Front Oncol 9:746. https://doi.org/10.3389/fonc.2019.00746
Sharon Y, Raz Y, Cohen N, Ben-Shmuel A, Schwartz H, Geiger T, Erez N (2015) Tumor-derived osteopontin reprograms normal mammary fibroblasts to promote inflammation and tumor growth in breast cancer. Cancer Res 75(6):963–973. https://doi.org/10.1158/0008-5472.CAN-14-1990
Gothlin Eremo A, Lagergren K, Othman L, Montgomery S, Andersson G, Tina E (2020) Evaluation of SPP1/osteopontin expression as predictor of recurrence in tamoxifen treated breast cancer. Sci Rep 10:1451. https://doi.org/10.1038/s41598-020-58323-w
Song Y, Lu M, Feng L, Chen Q, Huang H, Lin Q (2022) Identification of potential immunotherapy biomarkers for breast cancer by bioinformatics analysis. Biosci Rep 42:BSR20212035. https://doi.org/10.1042/BSR20212035
Krstic M, Hassan HM, Kolendowski B, Hague MN, Anborgh PH, Postenka CO, Torchia J, Chambers AF, Tuck AB (2020) Isoform-specific promotion of breast cancer tumorigenicity by TBX3 involves induction of angiogenesis. Lab Invest 100:400–413. https://doi.org/10.1038/s41374-019-0326-6
Insua-Rodriguez J, Pein M, Hongu T, Meier J, Descot A, Lowy CM, De Braekeleer E, Sinn HP, Spaich S, Sutterlin M, Schneeweiss A, Oskarsson T (2018) Stress signaling in breast cancer cells induces matrix components that promote chemoresistant metastasis. EMBO Mol Med 10:e9003. https://doi.org/10.15252/emmm.201809003
Kothari C, Clemenceau A, Ouellette G, Ennour-Idrissi K, Michaud A, R CG, Diorio C, Durocher F (2021) TBC1D9: an important modulator of tumorigenesis in breast Cancer. Cancers (Basel) 13:3557. https://doi.org/10.3390/cancers13143557
Spangle JM, Roberts TM, Zhao JJ (2017) The emerging role of PI3K/AKT-mediated epigenetic regulation in cancer. Biochim Biophys Acta Rev Cancer 1868(1):123–131. https://doi.org/10.1016/j.bbcan.2017.03.002
Xue C, Li G, Lu J, Li L (2021) Crosstalk between circRNAs and the PI3K/AKT signaling pathway in cancer progression. Signal Transduct Target Ther 6(1):400. https://doi.org/10.1038/s41392-021-00788-w
He Y, Sun MM, Zhang GG, Yang J, Chen KS, Xu WW, Li B (2021) Targeting PI3K/Akt signal transduction for cancer therapy. Signal Transduct Target Ther 6(1):425. https://doi.org/10.1038/s41392-021-00828-5
Nunnery SE, Mayer IA (2020) Targeting the PI3K/AKT/mTOR pathway in hormone-positive breast Cancer. Drugs 80(16):1685–1697. https://doi.org/10.1007/s40265-020-01394-w
Mi Z, Guo H, Russell MB, Liu Y, Sullenger BA, Kuo PC (2009) RNA aptamer blockade of osteopontin inhibits growth and metastasis of MDA-MB231 breast cancer cells. Mol Ther 17:153–161. https://doi.org/10.1038/mt.2008.235
Zhu K, Wu Y, He P, Fan Y, Zhong X, Zheng H, Luo T (2022) PI3K/AKT/mTOR-Targeted therapy for breast Cancer. Cells 11:2508. https://doi.org/10.3390/cells11162508
Di Leo A, Johnston S, Lee KS, Ciruelos E, Lonning PE, Janni W, O’Regan R, Mouret-Reynier MA, Kalev D, Egle D, Csoszi T, Bordonaro R, Decker T, Tjan-Heijnen VCG, Blau S, Schirone A, Weber D, El-Hashimy M, Dharan B, Sellami D, Bachelot T (2018) Buparlisib plus fulvestrant in postmenopausal women with hormone-receptor-positive, HER2-negative, advanced breast cancer progressing on or after mTOR inhibition (BELLE-3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 19:87–100. https://doi.org/10.1016/S1470-2045(17)30688-5
Ooms LM, Binge LC, Davies EM, Rahman P, Conway JR, Gurung R, Ferguson DT, Papa A, Fedele CG, Vieusseux JL, Chai RC, Koentgen F, Price JT, Tiganis T, Timpson P, McLean CA, Mitchell CA (2015) The Inositol Polyphosphate 5-Phosphatase PIPP regulates AKT1-Dependent breast Cancer Growth and Metastasis. Cancer Cell 28:155–169. https://doi.org/10.1016/j.ccell.2015.07.003
Gao X, Qin T, Mao J, Zhang J, Fan S, Lu Y, Sun Z, Zhang Q, Song B, Li L (2019) PTENP1/miR-20a/PTEN axis contributes to breast cancer progression by regulating PTEN via PI3K/AKT pathway. J Exp Clin Cancer Res 38:256. https://doi.org/10.1186/s13046-019-1260-6
Mao M, Chen Y, Jia Y, Yang J, Wei Q, Li Z, Chen L, Chen C, Wang L (2019) PLCA8 suppresses breast cancer apoptosis by activating the PI3k/AKT/NF-kappaB pathway. J Cell Mol Med 23:6930–6941. https://doi.org/10.1111/jcmm.14578
Abolghasemi M, Tehrani SS, Yousefi T, Karimian A, Mahmoodpoor A, Ghamari A, Jadidi-Niaragh F, Yousefi M, Kafil HS, Bastami M, Edalati M, Eyvazi S, Naghizadeh M, Targhazeh N, Yousefi B, Safa A, Majidinia M, Rameshknia V (2020) MicroRNAs in breast cancer: roles, functions, and mechanism of actions. J Cell Physiol 235(6):5008–5029. https://doi.org/10.1002/jcp.29396
Flores-Perez A, Marchat LA, Rodriguez-Cuevas S, Bautista VP, Fuentes-Mera L, Romero-Zamora D, Maciel-Dominguez A, de la Cruz OH, Fonseca-Sanchez M, Ruiz-Garcia E, la Vega HA, Lopez-Camarillo C (2016) Suppression of cell migration is promoted by miR-944 through targeting of SIAH1 and PTP4A1 in breast cancer cells. BMC Cancer 16:379. https://doi.org/10.1186/s12885-016-2470-3
Ali Syeda Z, Langden SSS, Munkhzul C, Lee M, Song SJ (2020) Regulatory mechanism of MicroRNA expression in Cancer. Int J Mol Sci 21:1723. https://doi.org/10.3390/ijms21051723
He H, Tian W, Chen H, Jiang K (2016) MiR-944 functions as a novel oncogene and regulates the chemoresistance in breast cancer. Tumour Biol 37:1599–1607. https://doi.org/10.1007/s13277-015-3844-x
Pei Q, Liu GS, Li HP, Zhang Y, Xu XC, Gao H, Zhang W, Li T (2019) Long noncoding RNA SNHG14 accelerates cell proliferation, migration, invasion and suppresses apoptosis in colorectal cancer cells by targeting miR-944/KRAS axis through PI3K/AKT pathway. Eur Rev Med Pharmacol Sci 23:9871–9881. https://doi.org/10.26355/eurrev_201911_19551
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Conceptualization, Ying Zhang, Shan Li, Xiangguo Cui, Yiliang Wang; Formal analysis, Ying Zhang, Shan Li; Investigation, Ying Zhang, Shan Li; Methodology, Ying Zhang, Shan Li; Project administration, Ying Zhang, Shan Li, Xiangguo Cui, Yiliang Wang; Resources, Xiangguo Cui, Yiliang Wang; Supervision, Xiangguo Cui, Yiliang Wang; Writing – original draft, Ying Zhang, Shan Li; Writing – review & editing, Xiangguo Cui, Yiliang Wang. All authors have read and approved the final submitted manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interest.
Ethical approval
This study was approved by the clinical ethics committee of the First Hospital of China Medical University, in strict accordance with the Declaration of Helsinki. All patients signed the informed consent form. During animal experiments, the principles of completing experiments with the least number of animals and minimizing the pain of experimental animals were strictly obeyed.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Li, S., Cui, X. et al. microRNA-944 inhibits breast cancer cell proliferation and promotes cell apoptosis by reducing SPP1 through inactivating the PI3K/Akt pathway. Apoptosis 28, 1546–1563 (2023). https://doi.org/10.1007/s10495-023-01870-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01870-0