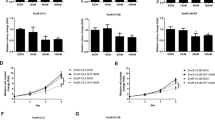
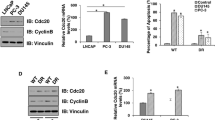
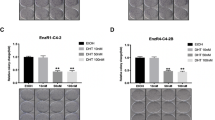
Abstract
Purvalanol and roscovitine are cyclin dependent kinase (CDK) inhibitors that induce cell cycle arrest and apoptosis in various cancer cells. We further hypothesized that co-treatment of CDK inhibitors with rapamycin, an mTOR inhibitor, would be an effective combinatory strategy for the inhibition of prostate cancer regard to androgen receptor (AR) status due to inhibition of proliferative pathway, PI3K/AKT/mTOR, and induction of cell death mechanisms. Androgen responsive (AR+), PTEN−/− LNCaP and androgen independent (AR−), PTEN+/− DU145 prostate cancer cells were exposed to purvalanol (20 µM) and roscovitine (30 µM) with or without rapamycin for 24 h. Cell viability assay, immunoblotting, flow cytometry and fluorescence microscopy was used to define the effect of CDK inhibitors with or without rapamycin on proliferative pathway and cell death mechanisms in LNCaP and DU145 prostate cancer cells. Co-treatment of rapamycin modulated CDK inhibitors-induced cytotoxicity and apoptosis that CDK inhibitors were more potent to induce cell death in AR (+) LNCaP cells than AR (−) DU145 cells. CDK inhibitors in the presence or absence of rapamycin induced cell death via modulating upstream PI3K/AKT/mTOR signaling pathway in LNCaP cells, exclusively only treatment of purvalanol have strong potential to inhibit both upstream and downstream targets of mTOR in LNCaP and DU145 cells. However, co-treatment of rapamycin with CDK inhibitors protects DU145 cells from apoptosis via induction of autophagy mechanism. We confirmed that purvalanol and roscovitine were strong apoptotic and autophagy inducers that based on regulation of PI3K/AKT/mTOR signaling pathway. Co-treatment of rapamycin with purvalanol and roscovitine exerted different effects on cell survival and death mechanisms in LNCaP and DU145 cell due to their AR receptor status. Our studies show that co-treatment of rapamycin with CDK inhibitors inhibit prostate cancer cell viability more effectively than either agent alone, in part, by targeting the mTOR signaling cascade in AR (+) LNCaP cells. In this point, mTOR is a fine-tuning player in purvalanol and roscovitine-induced apoptosis and autophagy via regulation of PI3K/AKT and the downstream targets, which related with cell proliferation.










Similar content being viewed by others
References
Pestell RG, Albanese C, Reutens AT, Segall JE, Lee RJ, Arnold A (1999) The cyclins and cyclin-dependent kinase inhibitors in hormonal regulation of proliferation and differentiation. Endocr Rev 20:501–534
Arisan ED, Obakan P, Coker-Gurkan A, Calcabrini A, Agostinelli E, Unsal NP (2014) CDK inhibitors induce mitochondria-mediated apoptosis through the activation of polyamine catabolic pathway in LNCaP, DU145 and PC3 prostate cancer cells. Curr Pharm Des 20:180–188
Mohapatra S, Chu B, Zhao X, Djeu J, Cheng JQ, Pledger WJ (2009) Apoptosis of metastatic prostate cancer cells by a combination of cyclin-dependent kinase and AKT inhibitors. Int J Biochem Cell Biol 41:595–602
Wesierska-Gadek J, Gritsch D, Zulehner N, Komina O, Maurer M (2011) Roscovitine, a selective CDK inhibitor, reduces the basal and estrogen-induced phosphorylation of ER-alpha in human ER-positive breast cancer cells. J Cell Biochem 112:761–772
Coker-Gurkan A, Arisan ED, Obakan P, Guvenir E, Unsal NP (2014) Inhibition of autophagy by 3-MA potentiates purvalanol-induced apoptosis in Bax deficient HCT 116 colon cancer cells. Exp Cell Res 328:87–98
Gelmann EP (2002) Molecular biology of the androgen receptor. J Clin Oncol 20:3001–3015
Wu Y, Chhipa RR, Cheng J, Zhang H, Mohler JL, Ip C (2010) Androgen receptor-mTOR crosstalk is regulated by testosterone availability: implication for prostate cancer cell survival. Anticancer Res 30:3895–3901
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–293
Noda T, Ohsumi Y (1998) Tor, a phosphatidylinositol kinase homologue, controls autophagy in yeast. J Biol Chem 273:3963–3966
Kulich I, Zarsky V (2014) Autophagy-related direct membrane import from ER/cytoplasm into the vacuole or apoplast: a hidden gateway also for secondary metabolites and phytohormones? Int J Mol Sci 15:7462–7474
Neasta J, Barak S, Hamida SB, Ron D (2014) mTOR complex 1: a key player in neuroadaptations induced by drugs of abuse. J Neurochem 130:172–184
Choo AY, Blenis J (2009) Not all substrates are treated equally: implications for mTOR, rapamycin-resistance and cancer therapy. Cell Cycle 8:567–572
Sarbassov DD, Ali SM, Kim DH et al (2004) Rictor, a novel binding partner of mTOR, defines a rapamycin-insensitive and raptor-independent pathway that regulates the cytoskeleton. Curr Biol 14:1296–1302
Beugnet A, Tee AR, Taylor PM, Proud CG (2003) Regulation of targets of mTOR (mammalian target of rapamycin) signalling by intracellular amino acid availability. Biochem J 372:555–566
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307:1098–1101
Karrasch T, Spaeth T, Allard B, Jobin C (2011) PI3 K-dependent GSK3ss(Ser9)-phosphorylation is implicated in the intestinal epithelial cell wound-healing response. PLoS One 6:e26340
Wang H, Quirion R, Little PJ et al (2015) Forkhead box O transcription factors as possible mediators in the development of major depression. Neuropharmacology 99:527–537
Ouyang DY, Xu LH, He XH et al (2013) Autophagy is differentially induced in prostate cancer LNCaP, DU145 and PC-3 cells via distinct splicing profiles of ATG5. Autophagy 9:20–32
Degtyarev M, De Maziere A, Orr C et al (2008) Akt inhibition promotes autophagy and sensitizes PTEN-null tumors to lysosomotropic agents. J Cell Biol 183:101–116
Nesterov A, Lu X, Johnson M, Miller GJ, Ivashchenko Y, Kraft AS (2001) Elevated AKT activity protects the prostate cancer cell line LNCaP from TRAIL-induced apoptosis. J Biol Chem 276:10767–10774
Xu Y, Chen SY, Ross KN, Balk SP (2006) Androgens induce prostate cancer cell proliferation through mammalian target of rapamycin activation and post-transcriptional increases in cyclin D proteins. Cancer Res 66:7783–7792
Zhang HH, Lipovsky AI, Dibble CC, Sahin M, Manning BD (2006) S6K1 regulates GSK3 under conditions of mTOR-dependent feedback inhibition of Akt. Mol Cell 24:185–197
Suryadinata R, Sadowski M, Steel R, Sarcevic B (2011) Cyclin-dependent kinase-mediated phosphorylation of RBP1 and pRb promotes their dissociation to mediate release of the SAP30.mSin3.HDAC transcriptional repressor complex. J Biol Chem 286:5108–5118
Liu T, Zhu E, Wang L, Okada T, Yamaguchi A, Okada N (2005) Abnormal expression of Rb pathway-related proteins in salivary gland acinic cell carcinoma. Hum Pathol 36:962–970
Seglen PO, Gordon PB (1982) 3-Methyladenine: specific inhibitor of autophagic/lysosomal protein degradation in isolated rat hepatocytes. Proc Natl Acad Sci USA 79:1889–1892
Wang Q, Zheng XL, Yang L et al (2010) Reactive oxygen species-mediated apoptosis contributes to chemosensitization effect of saikosaponins on cisplatin-induced cytotoxicity in cancer cells. J Exp Clin Cancer Res 29:159
Kapuy O, Vinod PK, Banhegyi G (2014) mTOR inhibition increases cell viability via autophagy induction during endoplasmic reticulum stress—an experimental and modeling study. FEBS Open Bio 4:704–713
Facompre ND, Sinha I, El-Bayoumy K, Pinto JT, Sinha R (2012) Remarkable inhibition of mTOR signaling by the combination of rapamycin and 1,4-phenylenebis(methylene)selenocyanate in human prostate cancer cells. Int J Cancer 131:2134–2142
Wu Y, Chhipa RR, Cheng J, Zhang H, Mohler JL, Ip C (2010) Androgen receptor-mTOR crosstalk is regulated by testosterone availability: implication for prostate cancer cell survival. Anticancer Res 30:3895–3901
Chang L, Graham PH, Hao J et al (2014) PI3 K/Akt/mTOR pathway inhibitors enhance radiosensitivity in radioresistant prostate cancer cells through inducing apoptosis, reducing autophagy, suppressing NHEJ and HR repair pathways. Cell Death Dis 5:e1437
Matsuda S, Nakanishi A, Wada Y, Kitagishi Y (2013) Roles of PI3K/AKT/PTEN pathway as a target for pharmaceutical therapy. Open Med Chem J 7:23–29
Ross AH, Gericke A (2009) Phosphorylation keeps PTEN phosphatase closed for business. Proc Natl Acad Sci USA 106:1297–1298
Martin KA, Blenis J (2002) Coordinate regulation of translation by the PI 3-kinase and mTOR pathways. Adv Cancer Res 86:1–39
Wood AR, Esko T, Yang J et al (2014) Defining the role of common variation in the genomic and biological architecture of adult human height. Nat Genet 46:1173–1186
Gwinn DM, Shackelford DB, Egan DF et al (2008) AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 30:214–226
Gwinn DM, Asara JM, Shaw RJ (2010) Raptor is phosphorylated by cdc2 during mitosis. PLoS One 5:e9197
Lee SL, Chou CC, Chuang HC et al (2013) Functional Role of mTORC2 versus Integrin-linked kinase in mediating Ser473-Akt phosphorylation in PTEN-negative prostate and breast cancer cell lines. PLoS One 8:e67149
Dibble CC, Asara JM, Manning BD (2009) Characterization of Rictor phosphorylation sites reveals direct regulation of mTOR complex 2 by S6K1. Mol Cell Biol 29:5657–5670
Vasudevan KM, Barbie DA, Davies MA et al (2009) AKT-independent signaling downstream of oncogenic PIK3CA mutations in human cancer. Cancer Cell 16:21–32
Villerbu N, Gaben AM, Redeuilh G, Mester J (2002) Cellular effects of purvalanol A: a specific inhibitor of cyclin-dependent kinase activities. Int J Cancer 97:761–769
Dong J, Peng J, Zhang H et al (2005) Role of glycogen synthase kinase 3beta in rapamycin-mediated cell cycle regulation and chemosensitivity. Cancer Res 65:1961–1972
Diehl JA, Cheng M, Roussel MF, Sherr CJ (1998) Glycogen synthase kinase-3beta regulates cyclin D1 proteolysis and subcellular localization. Genes Dev 12:3499–3511
Gera JF, Mellinghoff IK, Shi Y et al (2004) AKT activity determines sensitivity to mammalian target of rapamycin (mTOR) inhibitors by regulating cyclin D1 and c-myc expression. J Biol Chem 279:2737–2746
Whittaker SR, Walton MI, Garrett MD, Workman P (2004) The Cyclin-dependent kinase inhibitor CYC202 (R-roscovitine) inhibits retinoblastoma protein phosphorylation, causes loss of Cyclin D1, and activates the mitogen-activated protein kinase pathway. Cancer Res 64:262–272
Xu W, Ngo L, Perez G, Dokmanovic M, Marks PA (2006) Intrinsic apoptotic and thioredoxin pathways in human prostate cancer cell response to histone deacetylase inhibitor. Proc Natl Acad Sci USA 103:15540–15545
Mercalli A, Sordi V, Ponzoni M et al (2006) Rapamycin induces a caspase-independent cell death in human monocytes. Am J Transplant 6:1331–1341
Edelstein CL (2008) Mammalian target of rapamycin and caspase inhibitors in polycystic kidney disease. Clin J Am Soc Nephrol 3:1219–1226
Song X, Dilly AK, Kim SY, Choudry HA, Lee YJ (2014) Rapamycin-enhanced mitomycin C-induced apoptotic death is mediated through the S6K1-Bad-Bak pathway in peritoneal carcinomatosis. Cell Death Dis 5:e1281
Pietrocola F, Izzo V, Niso-Santano M et al (2013) Regulation of autophagy by stress-responsive transcription factors. Semin Cancer Biol 23:310–322
Furuya T, Kim M, Lipinski M et al (2010) Negative regulation of Vps34 by Cdk mediated phosphorylation. Mol Cell 38:500–511
Hu C, Zou MJ, Zhao L et al (2012) E Platinum, a newly synthesized platinum compound, induces autophagy via inhibiting phosphorylation of mTOR in gastric carcinoma BGC-823 cells. Toxicol Lett 210:78–86
Kabeya Y, Mizushima N, Ueno T et al (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Zhang L, Zhao Y, Ding W et al (2015) Autophagy regulates colistin-induced apoptosis in PC-12 cells. Antimicrob Agents Chemother 59:2189–2197
Goodyear S, Sharma MC (2007) Roscovitine regulates invasive breast cancer cell (MDA-MB231) proliferation and survival through cell cycle regulatory protein cdk5. Exp Mol Pathol 82:25–32
Cole AR, Soutar MP, Rembutsu M et al (2008) Relative resistance of Cdk5-phosphorylated CRMP2 to dephosphorylation. J Biol Chem 283:18227–18237
Dulinska-Litewka J, McCubrey JA, Laidler P (2013) Increased Akt signaling resulting from the loss of androgen responsiveness in prostate cancer. Curr Med Chem 20:144–157
Cao C, Subhawong T, Albert JM et al (2006) Inhibition of mammalian target of rapamycin or apoptotic pathway induces autophagy and radiosensitizes PTEN null prostate cancer cells. Cancer Res 66:10040–10047
Boutin B, Tajeddine N, Vandersmissen P et al (2013) Androgen deprivation and androgen receptor competition by bicalutamide induce autophagy of hormone-resistant prostate cancer cells and confer resistance to apoptosis. Prostate 73:1090–1102
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berrak, O., Arisan, E.D., Obakan-Yerlikaya, P. et al. mTOR is a fine tuning molecule in CDK inhibitors-induced distinct cell death mechanisms via PI3K/AKT/mTOR signaling axis in prostate cancer cells. Apoptosis 21, 1158–1178 (2016). https://doi.org/10.1007/s10495-016-1275-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-016-1275-9