Abstract
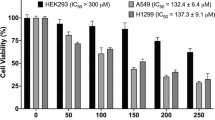
The 2,4-diacteylphloroglucinol (DAPG), a polyketide metabolite extracted from Pseudomonas aeruginosa strain FP10, exhibited selective cytoxicity against lung (A549), breast (MDA MB-231), cervical (HeLa) and colon (HCT-15) cancer cells in differential and dose-dependent manner. The anticancer and antimetastatic activities of DAPG were mediated by the inhibition of ROS, NF-κB, Bcl-2, MMP-2, VEGF and primary inflammatory mediators such as TNF-α, IL-6, IL-1β and NO. The DAPG induced apoptosis in cancer cells by intrinsic and extrinsic pathways via the release of cytochrome-C, upregulation of Bax and the activation of caspases and also, exhibited anti-inflammatory activity by the inhibition of LPS-inflammed cell proliferation of macrophage (Raw 264.7), monocytic cells (THP-1) and peripheral blood mononuclear cells (PBMCs). Results further confirmed that the DAPG inhibited the primary inflammatory mediators in cancer cells and inflammed immune cells through the down regulation of NF-κB. In the present study, for the first time, antiproliferative, proapoptotic, antimetastatic and anti-inflammatory activities of DAPG in various cancer cells and inflammation-induced immune cells have been reported.








Similar content being viewed by others
Abbreviations
- AO:
-
Acridine orange
- Bax:
-
Bcl-2 associated X-protein
- Bcl-2:
-
B-cell lymphoma 2
- CRI:
-
Cancer-related inflammation
- DCF:
-
2,7-Dichlorofluorescein
- DCFDA:
-
2,7-Dichlorofluorescein diacetate
- DMEM:
-
Dulbecco’s modified eagle medium
- RPMI:
-
Rosewell park memorial institute
- EB:
-
Ethidium bromide
- FBS:
-
Fetal bovine serum
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- IL-1:
-
Interleukin-1
- IL-1β:
-
Interleukin-1 beta
- IL-6:
-
Interleukin-6
- JSH:
-
23-4-Methyl-N1-(3-phenyl-propyl)-benzene-1,2-diamine
- LDH:
-
Lactate dehydrogenase
- LPS:
-
Lipopolysaccharride
- MTT:
-
3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide
- NO:
-
Nitric oxide
- PE:
-
Phycoerthyrin
- PI:
-
Propidium iodide
- PPM:
-
Pigment producing media
- RNase:
-
Ribonulease
- ROS:
-
Reactive oxygen species
- VEGF:
-
Vascular endothelial growth factor
- VRSA:
-
Vancomycin resistant Staphylococcus aureus
- VSV:
-
Vascular stomatitis viruses (VSV-1)
References
Floor SL, Dumont JE, Maenhaut C, Raspe E (2012) Hallmarks of cancer: of all cancer cells, all the time? Trends Mol Med 18:509–515
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Meacham CE, Morrison SJ (2013) Tumour heterogeneity and cancer cell plasticity. Nature 501:328–337
Cavallo F, De Giovanni C, Nanni P, Forni G, Lollini PL (2011) 2011: the immune hallmarks of cancer. Cancer Immunol Immunother 60:319–326
Igney FH, Krammer PH (2002) Immune escape of tumors: apoptosis resistance and tumor counterattack. J Leukoc Biol 71:907–920
Bhowmick NA, Neilson EG, Moses HL (2004) Stromal fibroblasts in cancer initiation and progression. Nature 432:332–337
Goel S, Duda DG, Xu L, Munn LL, Boucher Y, Dai Fukumura, Jain RK (2011) Normalization of the vasculature for treatment of cancer and other diseases. Physiol Rev 91:1071–1121
Pietras K, Ostman A (2010) Hallmarks of cancer: interactions with the tumor stroma. Exp Cell Res 316:1324–1331
Nelson D, Ganss R (2006) Tumor growth or regression: powered by inflammation. J Leukoc Biol 80:685–690
Smith GR, Missailidis S (2004) Cancer, inflammation and the AT1 and AT2 receptors. J Inflamm 1:1–12
Lu H, Ouyang W, Huang C (2006) Inflammation, a key event in cancer development. Mol Cancer Res 4:221–233
Shigdar S, Li Y, Bhattacharya S, O’Connor M, Pu C, Lin J, Wang T, Xiang D, Kong L, Wei MQ, Zhu Y, Zhou S, Duan W (2014) Inflammation and cancer stem cells. Cancer Lett 345:271–278
Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA (2007) Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449:557–563
Riss J, Khanna C, Koo S, Chandramouli GV, HuY Yang H H, Kleiner DE, Rosenwald A, Schaefer CF, Ben-Sasson SA, Yang L, Powell J, Kane DW, Star RA, Aprelikova O, Bauer K, Vasselli JR, Maranchie JK, Kohn KW, Buetow KH, Linehan WM, Weinstein JN, Lee MP, Klausner RD, Barrett JC (2006) Cancers as wounds that do not heal: differences and similarities between renal regeneration/repair and renal cell carcinoma. Cancer Res 66:7216–7224
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454:436–444
Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A (2009) Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis 30:1073–1081
Koontongkaew S (2013) The tumor microenvironment contribution to development, growth, invasion and metastasis of head and neck Squamous cell carcinomas. J Cancer 4:66–83
Coussens LM, Werb Z (2001) Inflammatory cells and cancer: think different! J Exp Med 193:F23–F26
Demaria S, Pikarsky E, Karin M, Coussens LM, Chen YC, El-Omar EM, Trinchieri G, Dubinett SM, Mao JT, Szabo E, Krieg A, Weiner GJ, Fox BA, Coukos G, Wang E, Abraham RT, Carbone M, Lotze MT (2010) Cancer and inflammation: promise for biologic therapy. J Immunother 33:335–351
Mbeunkui F, Johann DJ (2009) Cancer and the tumor microenvironment: a review of an essential relationship. Cancer Chemother Pharmacol 63:571–582
Kim Y, Stolarska MA, Othmer HG (2011) The role of the microenvironment in tumor growth and invasion. Prog Biophys Mol Biol 106:353–379
Rivkin TL, Liubomirski Y, Bernstein B, Meshel T, Ben-Baruch A (2013) Inflammatory factors of the tumor microenvironment induce plasticity in non transformed breast epithelial cells: EMT, invasion, and collapse of normally organized breast. Neoplasia 15:1330–1346
Jain RK (2013) Normalizing tumor microenvironment to treat cancer: bench to bedside to biomarkers. J Clin Oncol 31:2205–2218
Gibbs JB (2000) Mechanism-based target identification and drug discovery in cancer research. Science 287:1969–1973
Schenone M, Dancik V, Wagner BK, Clemons PA (2013) Target identification and mechanism of action in chemical biology and drug discovery. Nat Chem Biol 9:232–240
Singh IP, Bharate SP (2006) Phloroglucinol compounds of natural origin. Nat Prod Rep 23:558–591
Bangera MG, Thamashow LS (1999) Identification and characterisation of gene cluster for synthesis of the polyketide antibiotic 2,4-Diacetylphloroglucinol from Pseudomonas Q2-87. J Bacteriol 181:3155–3163
Kidarsa TA, Goebel NC, Zabriskie TM, Loper JE (2011) Phloroglucinol mediates cross-talk between the pyoluteorin and 2,4-diacetylphloroglucinol biosynthetic pathways in Pseudomonas fluorescens Pf-5. Mol Microbiol 81:395–414
Tada M, Takakuwa T, Nagai M, Yoshii T (1990) Antiviral and antimicrobial activity of 2,4-diacetylphloroglucinols, 2-acycyclohexane-1,3-diones and 2-carboxamidocyclo-hexane-1,3-diones. Agric Biol Chem 54:3061–3063
Kamei Y, Isnansetyo A (2003) Lysis of methicillin-resistant Staphylococcus aureus by 2,4-diacetylphloroglucinol produced by Pseudomonas sp. AMSN isolated from a marine alga. Int J Antimicrobial Agents 21:71–74
Isnansetyo A, Horikawa M, Kamei Y (2001) In vitro anti-methicillin-resistant Staphylococcus aureus activity of 2,4-diacetylphloroglucinol produced by Pseudomonas sp. AMSN isolated from a marine alga. J Antimicrobial Chemotherapy 47:719–730
Isnansetyo A (2003) Antibacterial activity of 2,4-diacetylphloroglucinol produced by Pseudomonas sp. AMSN isolated from a marine alga, against vancomycin-resistant Staphylococcus aureus. Int J Anti Agents 22:545–547
Leuner K, Heiser JH, Derksen S, Mladenov MI, Fehske CJ, Schubert R, Gollasch M, Schneider G, Harteneck C, Chatterjee SS, Muller WE (2010) Simple 2,4-diacylphloroglucinols as classic transient receptor potential-6 activators–identification of a novel pharmacophore. Mol Pharmacol 77:368–377
Ayyadurai N, Ravindra Naik P, Sreehari Rao M, Sunish Kumar R, Samrat SK, Manohar M, Sakthivel N (2006) Isolation and characterization of a novel banana rhizosphere bacterium as fungal antagonist and microbial adjuvant in micropropagation of banana. J Appl Microbiol 100:926–937
Zhu Y, Yu G, Zhang Y, Xu Z, Wang Y, Yan G, He Q (2013) A novel andrographolide AL-1 exerts its cytotoxicity on K562 cells through a ROS-dependent mechanism. Proteomics 13:169–178
Nunez R (2001) DNA measurement and cell cycle analysis by flow cytometry. Curr Issues Mol Biol 3:67–70
Kennedy RK, Naik RP, Veena V, Lakshmi BS, Lakshmi P, Krishna R, Sakthivel N (2015) 5-Methyl phenazine-1-carboxylic acid: a novel bioactive metabolite by a rhizosphere soil bacterium that exhibits potent antimicrobial and anticancer activities. Chem-Biol Int 231:71–82
Xu M, Bower KM, Wang S, Frank JA, Chen G, Ding M, Wang S, Shi X, Ke Z, Luo J (2010) Cyanidin-3-glucoside inhibits ethanol-induced invasion of breast cancer cells overexpressing ErbB2. Mol Cancer 9:9–14
Ferlini C, Scambia G (2007) Assay for apoptosis using the mitochondrial probes, rhodamine123 and 10-N-nonyl acridine orange. Nat Protoc 2:3111–3114
Hong S, Kim SH, Rhee MH, Kim AR, Jung JH, Chun T, Yoo ES, Cho JY (2003) In vitro anti-inflammatory and pro-aggregative effects of a lipid compound, petrocortyne A, from marine sponges. Naunyn Schmiedebergs Arch Pharmacol 368:448–456
Kennedy RK, Veena V, Naik PR, Lakshmi P, Krishna R, Sudharani S, Sakthivel N (2015) Phenazine-1-carboxamide (PCN) from Pseudomonas sp. strain PUP6 selectively induced apoptosis in lung (A549) and breast (MDA MB-231) cancer cells by inhibition of antiapoptotic Bcl-2 family proteins. Apoptosis 20:858–868
Krutzik PO, Nolan GP (2003) Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytometry Part A 55A:61–70
Shanmugaraj S, Selvaraj S, Murugan VP, Rajaganapathy BR, Shanmuganathan MV, Devaraj S, Subhadra LB (2010) 3-Hydroxylup-20(29)-ene-27,28-dioic acid dimethyl ester, a novel natural product from Plumbago zeylanica inhibits the proliferation and migration of MDA-MB-231 cells. Chem Biol Interact 188:412–420
Zong H, Wang F, Fan QX, Wang LX (2012) Curcumin inhibits metastatic progression of breast cancer cell through suppression of urokinase-type plasminogen activator by NF-kappa B signaling pathways. Mol Biol Rep 39:4803–4808
Lin C, Hou W, Shen S, Juan S, Ko C, Wang L, Chen Y (2008) Quercetin inhibition of tumor invasion via suppressing PKCδ/ERK/AP-1-dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis 29:1807–1815
Li F, Sethi G (2010) Targeting transcription factor NF-kappaB to overcome chemoresistance and radioresistance in cancer therapy. Biochim Biophys Acta 1805:167–180
Kracht M (2007) Targeting strategies to modulate the NF-κB and JNK signal transduction network. AntiInflammatory Antiallergy Agents Med Chem 6:71–84
Karin M (2006) Nuclear factor-kappaB in cancer development and progression. Nature 441:431–436
Acknowledgments
The joint University Grant Commission-Council for Scientific and Industrial Research (joint UGC-CSIR), Government of India, New Delhi through a research fellowship to Ms. V. Veena and the financial support from the Department of Biotechnology (DBT), New Delhi and University Grants Commission-Special Assistance Programme (UGC-SAP) coordinated by Prof. N. Sakthivel are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Veena, V.K., Popavath, R.N., Kennedy, K. et al. In vitro antiproliferative, pro-apoptotic, antimetastatic and anti-inflammatory potential of 2,4-diacteylphloroglucinol (DAPG) by Pseudomonas aeruginosa strain FP10. Apoptosis 20, 1281–1295 (2015). https://doi.org/10.1007/s10495-015-1162-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1162-9