Abstract
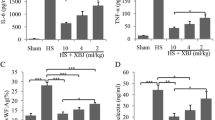
Intolerance to heat exposure is believed to be associated with hypothalamo-pituitary-adrenocortical (HPA) axis impairment [reflected by decreases in blood concentrations of both adrenocorticotrophic-hormone (ACTH) and corticosterone]. The purpose of this study was to determine the effect of human recombinant factor VIIa (rfVIIa) on heat intolerance, HPA axis impairment, and hypothalamic inflammation, ischemic and oxidative damage, and apoptosis in mice under heat stress. Immediately after heat stress (41.2 °C for 1 h), mice were treated with vehicle (1 mL/kg of body weight) or rfVIIa (65–270 µg/kg of body weight) and then returned to room temperature (26 °C). Mice still alive on day 4 of heat exposure were considered survivors. Cellular ischemia markers (e.g., glutamate, lactate-to-pyruvate ratio), oxidative damage markers (e.g., nitric oxide metabolite, hydroxyl radials), and pro-inflammatory cytokines (e.g., interleukin-6, interleukin-1β, tumor necrosis factor-α) in hypothalamus were determined. In addition, blood concentrations of both ACTH and corticosterone were measured. Hypothalamic cell damage was assessed by determing the neuronal damage scores, whereas the hypothalamic cell apoptosis was determined by assessing the numbers of cells stained with terminal deoxynucleotidyl transferase-mediated αUTP nick-end labeling, caspase-3-positive cells, and platelet endothelial cell adhesion molecula-1-positive cells in hypothalamus. Compared with vehicle-treated heated mice, rfVIIa-treated heated mice had significantly higher fractional survival (8/10 vs 1/10), lesser thermoregulatory deficit (34.1 vs 24.8 °C), lesser extents of ischemic, oxidative, and inflammatory markers in hypothalamus, lesser neuronal damage scores and apoptosis in hypothalamus, and lesser HPA axis impairment. Human recombinant factor VIIa appears to exert a protective effect against heatstroke by attenuating hypothalamic cell apoptosis (due to ischemic, inflammatory, and oxidative damage) in mice.








Similar content being viewed by others
References
Chen SH, Lin MT, Chang CP (2013) Ischemic and oxidative damage to the hypothalamus may be responsible for heat stroke. Curr Neuropharmacol 11:129–140
Chang CK, Chang CP, Chiu WT, Lin MT (2006) Prevention and repair of circulatory shock and cerebral ischemia/injury by various agents in experimental heatstroke. Curr Med Chem 13:3145–3154
Michel V, Peinnequin A, Alonso A, Buguet A, Cespuglio R, Canini F (2007) Decreased heat tolerance is associated with hypothalamo–pituitary– adrenocortical axis impairment. Neuroscience 147:522–531
Malamud N, Haymaker W, Custer RP (1946) Heat stroke; a clinico-pathologic study of 125 fatal cases. Mil Surg 99:397–449
Cure M (1989) Plasma corticosterone response in continuous versus discontinuous chronic heat exposure in rat. Physiol Behav 45:1117–1122
Djordjevic J, Cvijic G, Davidovic V (2003) Different activation of ACTH and corticosterone release in response to various stressors in rats. Physiol Res 52:67–72
Shapiro AD (2000) Recombinant factor VIIa in the treatment of bleeding in hemophilic children with inhibitors. Semin Thromb Hemost 26:413–419
Ingerslev J (2000) Efficacy and safety of recombinant factor VIIa in the prophylaxis of bleeding in various surgical procedures in hemophilic patients with factor VIII and factor 1× inhibitiors. Semin Thromb Hemost 26:425–432
Negrier C, Hay CRM (2000) The treatment of bleeding in hemophilic patients with inhibitors with recombinant factor VIIa. Semin Thromb Hemost 26:407–412
Hedner U (1998) Recombinant activated factor VII as a universal haemostatic agent. Blood Coagul Fibrin 9:S147–S152
Chao TC, Sinniah S, Pakiam JE (1981) Acute heat stroke deaths. Pathology 13:145–156
Sohal RS, Sun SC, Colcolough HL, Burch GE (1968) Heat stroke. An electron microscopic study of endothelia cell damage and disseminated intravascular coagulation. Arch Intern Med 122:43–47
Chatterjee S, Premachandran S, Bagewadikar RS, Bhattacharya S, Chattopadhyay S, Poduval TB (2006) Arginine metabolic pathways determine its therapeutic benefit in experimental heatstroke: role of Th1/Th2 cytokine balance. Nitric Oxide 15:408–416
Chatterjee S, Premachandran S, Sharma D, Bagewadikar RS, Poduval TB (2005) Therapeutic treatment with l-arginine rescues mice from heat shock-induced death: physiological and molecular mechanisms. Shock 24:341–347
Pulsinelli WA, Brierley JB, Plum F (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11:491–498
Newman PJ, Newman DK (2003) Signal transduction pathways mediated by PECAM-1: new roles for an old molecule in platelet and vascular cell biology. Arterioscler Thromb Vasc Biol 23:953–964
Gao C, Sun W, Christofidou-Solomidou M, Sawada M, Newman DK, Bergom C, Albelda SM, Matsuyama S, Newman PJ (2003) PECAM-1 functions as a specific and potent inhibitor of mitochondrial-dependent apoptosis. Blood 102:169–179
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407:770–776
Mullane KM, Kraemer R, Smith B (1985) Myeloperoxidase activity as a quantitative assessment of neutrophil infiltration into ischemic myocardium. J Pharmacol Methods 14:157–167
Chen SH, Chang FM, Tsai YC, Huang KF, Lin MT (2005) Resuscitation from experimental heatstroke by transplantation of human umbilical cord blood cells. Crit Care Med 33:1377–1383
Togashi H, Mori K, Ueno K, Matsumoto M, Suda N, Saito H et al (1998) Consecutive evaluation of nitric oxide production after transient cerebral ischemia in the rat hippocampus using in vivo brain microdialysis. Neurosci Lett 240:53–57
Poduval TB, Chatterjee S, Sainis KB (2003) Effect of nitric oxide on mortality of mice after whole body hyperthermia. Int J Hyperther 19:35–44
Lin CY, Hsu CC, Lin MT, Chen SH (2012) Flutamide, an androgen receptor antagonist, improves heatstroke outcomes in mice. Eur J Pharmacol 688:62–67
Lin CY, Lin MT, Cheng RT, Chen SH (2010) Testosterone depletion by castration may protect mice from heat-induced multiple organ damage and lethality. J Biomed Biotechnol 2010:485306
Berg DJ, Kühn R, Rajewsky K, Müller W, Menon S, Davidson N et al (1995) Interleukin-10 is a central regulator of the response to LPS in murine models of endotoxic shock and the Shwartzman reaction but not endotoxin tolerance. J Clin Invest 96:2339–2347
Rennick DM, Fort MM, Davidson NJ (1997) Studies with IL-10-/- mice: an overview. J Leukoc Biol 61:389–396
Endo S, Inada K, Kasai T, Takakuwa T, Yamada Y, Koike S et al (1995) Levels of soluble adhesion molecules and cytokines in patients with septic multiple organ failure. J Inflamm 46:212–219
Gérard C, Bruyns C, Marchant A, Abramowicz D, Vandenabeele P, Delvaux A et al (1993) Interleukin 10 reduces the release of tumor necrosis factor and prevents lethality in experimental endotoxemia. J Exp Med 177:547–550
Standiford TJ, Strieter RM, Lukacs NW, Kunkel SL (1995) Neutralization of IL-10 increases lethality in endotoxemia. Cooperative effects of macrophage inflammatory protein-2 and tumor necrosis factor. J Immunol 155:2222–2229
Leon LR, Blaha MD, Dubose DA (2006) Time course of cytokine, corticosterone, and tissue injury response in mice during heat strain recovery. J Appl Physiol 100:1400–1409
Kadhim HJ, Duchateau J, Sébire G (2008) Cytokines and brain injury: invited review. J Intensive Care Med 23:236–249
Bowen KK, Dempsey RJ, Vemuganti R (2011) Adult interleukin-6 knockout mice show compromised neurogenesis. NeuroReport 22:126–130
Monje ML, Toda H, Palmer TD (2003) Inflammatory blockade restores adult hippocampal neurogenesis. Science 302(5651):1760
Vallieres L, Campbell IL, Gage FH, Sawchenko PE (2002) Reduced hippocampal neurogenesis in adult transgenic mice with chronic astrocytic production of interluekin-6. J Neurosci 22:486–492
Ji R, Tian S, Lu HJ, Lu Q, Zheng Y, Wang X et al (2013) TAM receptors affect adult brain neurogenesis by negative regulation of microglial cell activation. J Immunol 191:6165–6177
Jongeneel VC (1994) Regulation of the TNF-α gene. Prog Clin Biol Res 388:367–381
Acknowledgments
This work was supported in part by Grants NSC99-2314-B-384-006-MY2, NSC99-2314-B-384-004-MY3, and NSC98-2314-B-218-MY2 from the Taiwan National Science Council; Grant DOH99-TD-B-111-003 from the Taiwan Department of Health; funds from the Taiwan Center of Excellence for Clinical Trials and Research in Neuroscience. We would like to thank four anonymous reviewers and the editor for their comments.
Conflict of interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Chuan-Chih Hsu and Sheng-Hsien Chen have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Hsu, CC., Chen, SH., Lin, CH. et al. Human recombinant factor VIIa may improve heat intolerance in mice by attenuating hypothalamic neuronal apoptosis and damage. Apoptosis 19, 1484–1496 (2014). https://doi.org/10.1007/s10495-014-1020-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-1020-1