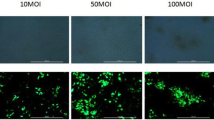
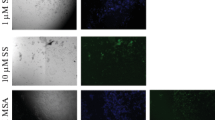
Abstract
Using siRNA as a tool, the channelization of pathway in H2O2 induced apoptosis of primary Leydig cells was investigated in vitro. Exposure (4 h) to H2O2 (250 μM) induced maximum apoptosis but affected Leydig cell viability significantly. Therefore, expression of apoptotic marker genes, caspase-8, -9, -3 and polyadenosine ribose polymerase was subsequently investigated using the same concentration post 1 h exposure. Incubation with siRNA (20 nM) either for caspase-8 or -9, inhibited their individual expressions by 55–60 % and activity, 50–55 %. The inhibition efficiency using siRNA was comparable with post- or pre-H2O2 treatment of cells. Like siRNA, Eugenia jambolana (100 μg/ml) plant extract too, effectively countered over-expression of all apoptotic marker proteins. Silencing expressions of caspase 8 but not 9 through siRNA leads to a profound inhibition of caspase 3 implying that H2O2 induced Leydig cell apoptosis is preferably channeled through extrinsic and later extending to other pathways.








Similar content being viewed by others
References
Svchnikov K, Izzo G, Landreh L, Weisser J, Soder O (2010) Endocrine disruptors and Leydig cell function. J Biomed Biotech. doi:10.1155/2010/684504
Skakkebaek NE, Rajpert-De Meyts E, Main KM (2001) Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum Reprod 16:972–978
Virtanena HE, Rajpert-De Meyts E, Main KM, Skakkebaek NE, Topparia J (2005) Testicular dysgenesis syndrome and the development and occurrence of male reproductive disorders. Toxicol Appl Pharmacol 207:S501–S505
Hedger MP, de Kretser DM (2000) Leydig cell function and regulation. Results Probl Cell Differ 28:69–110
Habert R, Lejeune H, Saez JM (2001) Origin, differentiation and regulation of fetal and adult Leydig cells. Mol Cell Endocrinol 179:47–74
Qin J, Tsai MJ, Tsai SY (2008) Essential roles of COUP-TFII in Leydig cell differentiation and male fertility. PLoS One 3:e3285. doi:10.1371/journal.pone.0003285
Barbouti A, Doulias PT, Nousis L, Tenopoulou M, Galaris D (2002) DNA damage and apoptosis in hydrogen peroxide-exposed jurkat cells: bolus versus continuous generation of H2O2. Free Radic Biol Med 33:691–702
Zhai L, Zhang P, Sun RY, Liu XY, Liu WG, Guo XL (2011) Cytoprotective effects of CSTMP, a novel stilbene derivative, against H2O2-induced oxidative stress in human endothelial cells. Pharmacol Rep 63:1469–1480
Liu RH, Yang MH, Xiang H, Bao LM, Yang HA, Yue LW, Jiang X, Ang N, Wu LY, Huang Y (2012) Depletion of OLFM4 gene inhibits cell growth and increases sensitization to hydrogen peroxide and tumor necrosis factor-alpha induced-apoptosis in gastric cancer cells. J Biomed Sci 19:38
Moon DO, Kim BY, Jang JH, Kim MO, Jayasooriya RG, Kang CH, Choi YH, Moon SK, Kim WJ, Ahn JS, Kim GY (2012) K-RAS transformation in prostate epithelial cell overcomes -induced apoptosis via upregulation of gamma-glutamyltransferase-2. Toxicol In Vitro 26:429–434
Gautam DK, Misro MM, Chaki SP, Sehgal N (2006) H2O2 at physiological concentrations modulates Leydig cell function inducing oxidative stress and apoptosis. Apoptosis 11:39–46
Aggarwal A, Misro MM, Maheshwari A, Sehgal N, Nandan D (2009) Adverse effects associated with persistent stimulation of Leydig cells with hCG in vitro. Mol Reprod Dev 76:1076–1083
Aggarwal A, Misro MM, Maheshwari A, Sehgal N, Nandan D (2010) N-Acetylcysteine counteracts oxidative stress and prevents hCG induced apoptosis in rat Leydig cells through caspase-8 and JNK down regulation. Mol Reprod Dev 77:900–909
Dean NM (2001) Functional genomics and target validation approaches using antisense oligonucleotide technology. Curr Opin Biotechnol 12:622–625
Bennett CF, Chiang M-Y, Chan H, Shoemaker JEE, Mirabelli CK (1992) Cationic lipids enhance cellular uptake and activity of phosphorothioate antisense oligonucleotides. Mol Pharmacol 41:1023–1033
Anand H, Misro MM, Sharma SB, Prakash S (2012) Cytoprotective effects of fruit pulp of Eugenia jambolana on H2O2 induced oxidative stress and apoptosis in rat Leydig cells in vitro. Andrologia. doi:10.1111/j.1439-0272.2012.01323.x. Epub ahead of print
Khan S, Teerds K, Dorrington J (1992) Growth factor requirements for DNA synthesis by Leydig cells from the immature rat. Biol Reprod 46:335–341
Maheshwari A, Misro MM, Aggarwal A, Sharma RK, Nandan D (2011) N-Acetyl-l-Cysteine counteracts oxidative stress and prevents H2O2 induced germ cell apoptosis through down-regulation of caspase-9 and JNK/c-Jun. Mol Reprod Dev 78:69–79
Benton L, Shan LX, Hardy MP (1995) Differentiation of adult Leydig cells. J Steroid Biochem Mol Biol 53:61–68
Kumar TR (2004) Divide and differentiate: ghrelin instructs the Leydig cells. Endocrinol 145:4822–4844
Hutson JC (2006) Physiologic interactions between macrophages and Leydig cells. Exp Biol Med (Maywood) 231:1–7
Hutson JC (1992) Development of cytoplasmic digitations between Leydig cells and testicular macrophages of the rat. Cell Tissue Res 267:385–389
Maheshwari A, Misro MM, Aggarwal A, Sharma RK (2012) N-Acetylcysteine modulates multiple signaling pathways to rescue male germ cells from apoptosis induced by chronic hCG administration to rats. Apoptosis 17:551–565
Tamura H, Ohtsuru A, Kamohara Y, Fujioka H, Yanaga K, Kanematsu T, Yamashita S (2003) Bax cleavage implicates caspase-dependent H2O2-induced apoptosis of hepatocytes. Int J Mol Med 11:369–374
Wu Y, Wang D, Wang X, Wang Y, Ren F, Chang D, Chang Z, Jia B (2011) Caspase 3 is activated through caspase 8 instead of caspase 9 during H2O2-induced apoptosis in HeLa cells. Cell Physiol Biochem 27:539–546
Maheshwari A, Misro MM, Aggarwal A, Sharma RK, Nandan D (2009) Pathways involved in testicular germ cell apoptosis induced by H2O2 in vitro. FEBS J 276:870–881
Kole R, Sazani P (2001) Antisense effects in the cell nucleus: modification of splicing. Curr Opin Mol Ther 3:229–234
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T (2001) Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411:494–498
Veldhoen S, Laufer SD, Restle T (2008) Recent developments in peptide-based nucleic acid delivery. Int J Mol Sci 9:1276–1320
Zetsepin TS, Turner JJ, Oretskaya TS, Gait MJ (2005) Conjugates of oligonucleotides and analogues with cell penetrating peptides as gene silencing agents. Curr Pharm Des 11:3639–3654
Ding SW (2005) RNAi: mechanisms, biology and applications. FEBS Lett 579:5821–6007
Rana TM (2007) Illuminating the silence: understanding the structure and function of small RNAs. Nat Rev Mol Cell Biol 8:23–36
Simeoni F, Morris MC, Heitz F, Divita G (2003) Insight into the mechanism of the peptide-based gene delivery system MPG: implications for delivery of siRNA into mammalian cells. Nucleic Acids Res 31:2717–2724
Chiu YL, Ali A, Chu CY, Cao H, Rana TM (2004) Visualizing a correlation between siRNA localization, cellular uptake and RNAi in living cells. Chem Biol 11:1165–1175
Kaufmann SH, Hengartner MO (2001) Programmed cell death: alive and well in the new millennium. Trends Cell Biol 11:526–534
Ghobrial IM, Witzig TE, Adjei AA (2005) Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin 55:178–194
Zapata JM, Pawlowski K, Haas E et al (2001) A diverse family of proteins containing tumor necrosis factor receptor-associated factor domains. J Biol Chem 276:24242–24252
Hockenbery D, Nunez G, Milliman C et al (1990) Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 348:334–336
Taylor MF, de Boer-Brouwer M, Woolveridge I, Teerds KJ, Morris ID (1999) Leydig cell apoptosis after the administration of ethane dimethane sulfonate to the adult male rat is Fas-mediated process. Endocrinology 140:3797–3804
Fruehauf JP Jr, Meyskens FL (2007) Reactive oxygen species: a breath of life or death. Clin Cancer Res 13:789–794
Sen CK (2000) Cellular thiols and redox-regulated signal transduction. Curr Top Cell Regul 36:1–30
Biswas S, Chida AS, Rahman I (2006) Redox modification of protein-thiols: emerging roles in cell signaling. Biochem Pharmacol 71:551–564
Acknowledgments
The generous gift of EJE extract from Dr. Suman Bala Sharma, Professor, University College of Medical Sciences, Delhi, is greatly acknowledged. The study was funded by NIHFW.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anand, H., Misro, M.M., Sharma, S.B. et al. siRNA as a tool to delineate pathway channelization in H2O2 induced apoptosis of primary Leydig cells in vitro. Apoptosis 17, 1131–1143 (2012). https://doi.org/10.1007/s10495-012-0749-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-012-0749-7