Abstract
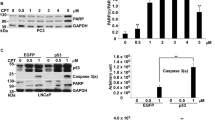
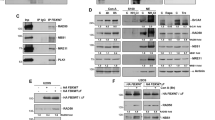
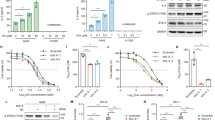
Various DNA-targeting agents may initiate p53-dependent as well as p53-independent response and subsequent apoptosis via alternative cellular systems which include for instance p73, caspase-2 or Bcl-2 family proteins. The scope of involvement of individual molecules in this process and the mechanisms governing their potential interplay are still not entirely understood, in particular in highly aggressive cancers such as in malignant melanoma. In this work we investigated the role and involvement of both p53-dependent and -independent mechanisms in selected melanoma cell lines with differing status of p53 using a model DNA topoisomerase I inhibitor camptothecin (CPT). Here we report that CPT induced in Bowes melanoma cells apoptosis which is essentially p53 and mitochondria-dependent but with some involvement of caspase-2 and p73. Conversely, in mutant p53 melanoma cells overall levels of CPT-induced apoptosis are significantly lower, with p73 and caspase-2 signaling playing important roles. In addition, in these cells the expression of micro RNAs family 34 (miR-34) were low compared to wild-type p53 cells. The ectopic expression of wild type p53 than restored apoptotic response of cells to CPT despite the fact that the expression of miR-34 and miR-155 were not influenced. These results suggest that CPT induces multivariate cellular stress responses including activation of DNA-damage response-p53 pathway as well as p53-independent signaling and their mutual crosstalk play the decisive role in the efficient triggering of apoptosis in melanoma cells.








Similar content being viewed by others
References
Gray-Schopfer V, Wellbrock C, Marais R (2007) Melanoma biology and new targeted therapy. Nature 445:851–857
Ivanov VN, Bhoumik A, Ronai Z (2003) Death receptors and melanoma resistance to apoptosis. Oncogene 22:3152–3161
Soengas MS, Lowe SW (2003) Apoptosis and melanoma chemoresistance. Oncogene 22:3138–3151
Sigalotti L, Covre A, Fratta E, Parisi G, Colizzi F, Rizzo A, Danielli R, Nicolay HJ, Coral S, Maio M (2010) Epigenetics of human cutaneous melanoma: setting the stage for new therapeutic strategies. J Transl Med 8:56
Li G, Bush JA, Ho VC (2000) p53-dependent apoptosis in melanoma cells after treatment with camptothecin. J Invest Dermatol 114:514–519
Bae I, Smith ML, Sheikh MS, Zhan Q, Scudiero DA, Friend SH, O’Connor PM, Fornace AJ Jr (1996) An abnormality in the p53 pathway following gamma-irradiation in many wild-type p53 human melanoma lines. Cancer Res 56:840–847
Satyamoorthy K, Chehab NH, Waterman MJ, Lien MC, El-Deiry WS, Herlyn M, Halazonetis TD (2000) Aberrant regulation and function of wild-type p53 in radioresistant melanoma cells. Cell Growth Differ 11:467–474
Qin JZ, Stennett L, Bacon P, Bodner B, Hendrix MJ, Seftor RE, Seftor EA, Margaryan NV, Pollock PM, Curtis A, Trent JM, Bennett F, Miele L, Nickoloff BJ (2004) p53-independent NOXA induction overcomes apoptotic resistance of malignant melanomas. Mol Cancer Ther 3:895–902
Chen X, Zheng Y, Zhu J, Jiang J, Wang J (2001) p73 is transcriptionally regulated by DNA damage, p53, and p73. Oncogene 20:769–774
Robertson JD, Enoksson M, Suomela M, Zhivotovsky B, Orrenius S (2002) Caspase-2 acts upstream of mitochondria to promote cytochrome c release during etoposide-induced apoptosis. J Biol Chem 277:29803–29809
Bauer MK, Vogt M, Los M, Siegel J, Wesselborg S, Schulze-Osthoff K (1998) Role of reactive oxygen intermediates in activation-induced CD95 (APO-1/Fas) ligand expression. J Biol Chem 273:8048–8055
Breitschopf K, Haendeler J, Malchow P, Zeiher AM, Dimmeler S (2000) Posttranslational modification of Bcl-2 facilitates its proteasome-dependent degradation: molecular characterization of the involved signaling pathway. Mol Cell Biol 20:1886–1896
Klumpp S, Krieglstein J (2002) Serine/threonine protein phosphatases in apoptosis. Curr Opin Pharmacol 2:458–462
Cichorek M, Kozlowska K, Bryl E (2007) Mitochondrial transmembrane potential in spontaneous and camptothecin-induced apoptosis of melanotic and amelanotic melanoma cells. Neoplasma 54:29–36
Costanzo A, Merlo P, Pediconi N, Fulco M, Sartorelli V, Cole PA, Fontemaggi G, Fanciulli M, Schiltz L, Blandino G, Balsano C, Levrero M (2002) DNA damage-dependent acetylation of p73 dictates the selective activation of apoptotic target genes. Mol Cell 9:175–186
Urist M, Tanaka T, Poyurovsky MV, Prives C (2004) p73 induction after DNA damage is regulated by checkpoint kinases Chk1 and Chk2. Genes Dev 18:3041–3054
Zhivotovsky B, Orrenius S (2005) Caspase-2 function in response to DNA damage. Biochem Biophys Res Commun 331:859–867
Jimenez GS, Khan SH, Stommel JM, Wahl GM (1999) p53 regulation by post-translational modification and nuclear retention in response to diverse stresses. Oncogene 18:7656–7665
Wang Y, Prives C (1995) Increased and altered DNA binding of human p53 by S and G2/M but not G1 cyclin-dependent kinases. Nature 376:88–91
Lambert PF, Kashanchi F, Radonovich MF, Shiekhattar R, Brady JN (1998) Phosphorylation of p53 serine 15 increases interaction with CBP. J Biol Chem 273:33048–33053
D’Orazi G, Cecchinelli B, Bruno T, Manni I, Higashimoto Y, Saito S, Gostissa M, Coen S, Marchetti A, Del Sal G, Piaggio G, Fanciulli M, Appella E, Soddu S (2002) Homeodomain-interacting protein kinase-2 phosphorylates p53 at Ser 46 and mediates apoptosis. Nat Cell Biol 4:11–19
He L, He X, Lim LP, de Stanchina E, Xuan Z, Liang Y, Xue W, Zender L, Magnus J, Ridzon D, Jackson AL, Linsley PS, Chen C, Lowe SW, Cleary MA, Hannon GJ (2007) A microRNA component of the p53 tumour suppressor network. Nature 447:1130–1134
Chang TC, Wentzel EA, Kent OA, Ramachandran K, Mullendore M, Lee KH, Feldmann G, Yamakuchi M, Ferlito M, Lowenstein CJ, Arking DE, Beer MA, Maitra A, Mendell JT (2007) Transactivation of miR-34a by p53 broadly influences gene expression and promotes apoptosis. Mol Cell 26:745–752
Lodygin D, Tarasov V, Epanchintsev A, Berking C, Knyazeva T, Korner H, Knyazev P, Diebold J, Hermeking H (2008) Inactivation of miR-34a by aberrant CpG methylation in multiple types of cancer. Cell Cycle 7:2591–2600
Levati L, Alvino E, Pagani E, Arcelli D, Caporaso P, Bondanza S, Di Leva G, Ferracin M, Volinia S, Bonmassar E, Croce CM, D’Atri S (2009) Altered expression of selected microRNAs in melanoma: antiproliferative and proapoptotic activity of miRNA-155. Int J Oncol 35:393–400
He L, He X, Lowe SW, Hannon GJ (2007) microRNAs join the p53 network–another piece in the tumour-suppression puzzle. Nat Rev Cancer 7:819–822
Yamashita T, Tokino T, Tonoki H, Moriuchi T, Jin HY, Omori F, Jimbow K (2001) Induction of apoptosis in melanoma cell lines by p53 and its related proteins. J Invest Dermatol 117:914–919
Chakraborty J, Banerjee S, Ray P, Hossain DM, Bhattacharyya S, Adhikary A, Chattopadhyay S, Das T, Sa G (2010) Gain of cellular adaptation due to prolonged p53 impairment leads to functional switchover from p53 to p73 during DNA damage in acute myeloid leukemia cells. J Biol Chem 285:33104–33112
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12:440–450
Mhaidat NM, Wang Y, Kiejda KA, Zhang XD, Hersey P (2007) Docetaxel-induced apoptosis in melanoma cells is dependent on activation of caspase-2. Mol Cancer Ther 6:752–761
Melino G, Bernassola F, Ranalli M, Yee K, Zong WX, Corazzari M, Knight RA, Green DR, Thompson C, Vousden KH (2004) p73 Induces apoptosis via PUMA transactivation and Bax mitochondrial translocation. J Biol Chem 279:8076–8083
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB, Korsmeyer SJ (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292:727–730
Acknowledgment
This work was supported by Ministry of Education of Czech Republic Research Project MSM 0021620820.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rudolf, E., Rudolf, K. & Cervinka, M. Camptothecin induces p53-dependent and -independent apoptogenic signaling in melanoma cells. Apoptosis 16, 1165–1176 (2011). https://doi.org/10.1007/s10495-011-0635-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0635-8