Abstract
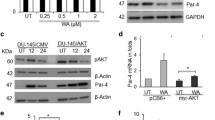
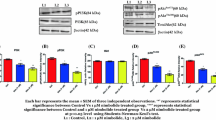
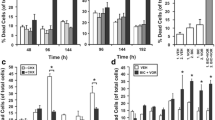
TNF-related apoptosis-inducing ligand (TRAIL) can induce apoptosis in many types of cancer cells. TRAIL is considered a therapeutic target, therefore, it was of interest to examine molecular mechanisms that may modulate sensitivity to TRAIL signaling in prostate cancer cells. LNCaP cells were found to be relatively resistant to TRAIL induced cell death while PC3 cells were sensitive. PI3-kinase (PI3 K) inhibitors were able to render LNCaP cells sensitive to TRAIL but conferred resistance to PC3 cells. PI3 K inhibitors were associated with an increase in p21waf1, cip1 expression in PC3 cells where as p21 decreases in LNCaP cells suggesting that p21 may impart TRAIL resistance. Since androgen receptor (AR) signaling can be modulated by AKT, and p21 is an AR responsive gene, the impact of PI3 K inhibition on TRAIL sensitivity was evaluated in AR transfected PC3 cells (PC3AR). The expression of AR was significantly downregulated by PI3 K inhibition in LNCaP cells, which have an intact AR signaling axis. PC3AR cells expressed higher levels of p21 protein and were relatively resistant to TRAIL compared to control cells. Finally, using adenoviral p21 gene transfer we directly demonstrated that p21 can confer resistance to TRAIL-induced cell death. These results suggest that TRAIL resistance is not regulated simply by a PI3 K/AKT survival pathway associated with inactivating PTEN mutations but may also be modulated by downstream AR responsive targets such as p21. These findings may have significant clinical implications for the utility of TRAIL in the management of prostate cancer.







Similar content being viewed by others
References
Westin P, Bergh A (1998) Apoptosis and other mechanisms in androgen ablation treatment and androgen independent progression of prostate cancer: a review. Cancer Detect Prev 22:476–484
Mitchell RE, Chang SS (2008) Current controversies in the treatment of high-risk prostate cancer. Curr Opin Urol 18:263–268
Syed S, Tolcher A (2003) Innovative therapies for prostate cancer treatment. Rev Urol 5(Suppl 3):S78–S84
Chi KN, Gleave ME (2004) Antisense approaches in prostate cancer. Expert Opin Biol Ther 4:927–936
Walczak H, Miller RE, Ariail K, Gliniak B, Griffith TS, Kubin M et al (1999) Tumoricidal activity of tumor necrosis factor-related apoptosis-inducing ligand in vivo. Nat Med 5:157–163
Sheridan JP, Marsters SA, Pitti RM, Gurney A, Skubatch M, Baldwin D et al (1997) Control of TRAIL-induced apoptosis by a family of signaling and decoy receptors. Science 277:818–821
Griffith TS, Rauch CT, Smolak PJ, Waugh JY, Boiani N, Lynch DH et al (1999) Functional analysis of TRAIL receptors using monoclonal antibodies. J Immunol 162:2597–2605
Guseva NV, Taghiyev AF, Rokhlin OW, Cohen MB (2004) Death receptor-induced cell death in prostate cancer. J Cell Biochem 91:70–99
Kennedy SG, Wagner AJ, Conzen SD, Jordan J, Bellacosa A, Tsichlis PN et al (1997) The PI 3-kinase/Akt signaling pathway delivers an anti-apoptotic signal. Genes Dev 11:701–713
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y et al (1997) Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91:231–241
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E et al (1998) Regulation of cell death protease caspase-9 by phosphorylation. Science 282:1318–1321
Kops GJ, de Ruiter ND, de Vries-Smits AM, Powell DR, Bos JL, Burgering BM (1999) Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature 398:630–634
Rossig L, Jadidi AS, Urbich C, Badorff C, Zeiher AM, Dimmeler S (2001) Akt-dependent phosphorylation of p21(Cip1) regulates PCNA binding and proliferation of endothelial cells. Mol Cell Biol 21:5644–5657
Gorospe M, Cirielli C, Wang X, Seth P, Capogrossi MC, Holbrook NJ (1997) p21(Waf1/Cip1) protects against p53-mediated apoptosis of human melanoma cells. Oncogene 14:929–935
Gartel AL (2009) p21(WAF1/CIP1) and cancer: a shifting paradigm? Biofactors 35:161–164
Marcelli M, Haidacher SJ, Plymate SR, Birnbaum RS (1995) Altered growth and insulin-like growth factor-binding protein-3 production in PC3 prostate carcinoma cells stably transfected with a constitutively active androgen receptor complementary deoxyribonucleic acid. Endocrinology 136:1040–1048
Spurgers KB, Coombes KR, Meyn RE, Gold DL, Logothetis CJ, Johnson TJ et al (2004) A comprehensive assessment of p53-responsive genes following adenoviral-p53 gene transfer in Bcl-2-expressing prostate cancer cells. Oncogene 23:1712–1723
Bigelow RL, Chari NS, Unden AB, Spurgers KB, Lee S, Roop DR et al (2004) Transcriptional regulation of bcl-2 mediated by the sonic hedgehog signaling pathway through gli-1. J Biol Chem 279:1197–1205
Kagawa S, Fujiwara T, Hizuta A, Yasuda T, Zhang WW, Roth JA et al (1997) p53 expression overcomes p21WAF1/CIP1-mediated G1 arrest and induces apoptosis in human cancer cells. Oncogene 15:1903–1909
Leverkus M, Neumann M, Mengling T, Rauch CT, Brocker EB, Krammer PH et al (2000) Regulation of tumor necrosis factor-related apoptosis-inducing ligand sensitivity in primary and transformed human keratinocytes. Cancer Res 60:553–559
Yuan S, Trachtenberg J, Mills GB, Brown TJ, Xu F, Keating A (1993) Androgen-induced inhibition of cell proliferation in an androgen-insensitive prostate cancer cell line (PC-3) transfected with a human androgen receptor complementary DNA. Cancer Res 53:1304–1311
Heisler LE, Evangelou A, Lew AM, Trachtenberg J, Elsholtz HP, Brown TJ (1997) Androgen-dependent cell cycle arrest and apoptotic death in PC-3 prostatic cell cultures expressing a full-length human androgen receptor. Mol Cell Endocrinol 126:59–73
McDonnell TJ, Navone NM, Troncoso P, Pisters LL, Conti C, von Eschenbach AC et al (1997) Expression of bcl-2 oncoprotein and p53 protein accumulation in bone marrow metastases of androgen independent prostate cancer. J Urol 157:569–574
Gleave M, Miyake H, Chi K (2005) Beyond simple castration: targeting the molecular basis of treatment resistance in advanced prostate cancer. Cancer Chemother Pharmacol 56(Suppl 1):47–57
Singh AS, Figg WD (2005) In vivo models of prostate cancer metastasis to bone. J Urol 174:820–826
Munshi A, Pappas G, Honda T, McDonnell TJ, Younes A, Li Y et al (2001) TRAIL (APO-2L) induces apoptosis in human prostate cancer cells that is inhibitable by Bcl-2. Oncogene 20:3757–3765
Parsons R (2004) Human cancer, PTEN and the PI-3 kinase pathway. Semin Cell Dev Biol 15:171–176
Li P, Nicosia SV, Bai W (2001) Antagonism between PTEN/MMAC1/TEP-1 and androgen receptor in growth and apoptosis of prostatic cancer cells. J Biol Chem 276:20444–20450
McDonnell TJ, Chari NS, Cho-Vega JH, Troncoso P, Wang X, Bueso-Ramos CE et al (2008) Biomarker expression patterns that correlate with high grade features in treatment naive, organ-confined prostate cancer. BMC Med Genomics 1:1
Xu SQ, El-Deiry WS (2000) p21(WAF1/CIP1) inhibits initiator caspase cleavage by TRAIL death receptor DR4. Biochem Biophys Res Commun 269:179–190
Acknowledgments
This work was supported by NIH grants P50 CA90270, and PO1 CA78778, and a Grant from the American Cancer Society RSG CDD-101054.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yoshihiko Kadowaki and Nikhil S. Chari contributed equally to this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kadowaki, Y., Chari, N.S., Teo, A.E.K. et al. PI3 Kinase inhibition on TRAIL-induced apoptosis correlates with androgen-sensitivity and p21 expression in prostate cancer cells. Apoptosis 16, 627–635 (2011). https://doi.org/10.1007/s10495-011-0591-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0591-3