Abstract
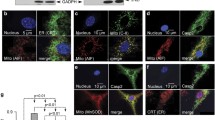
Caspase-3 is one of the main executors of apoptosis. Its zymogen procaspase-3 was localized to cytosol, mitochondria and nuclei. The subcellular location of procaspase-3 in liver was reported by several studies to be either cytosolic or cytosolic and mitochondrial. Our aim was to investigate these separate procaspase-3 pools to differentiate the pathways of their activation. By cell fractionation, immunocytochemistry, and confocal microscopy we report that there is a single procaspase-3 pool located to the cytosol in primary hepatocytes and in fractions of rat liver. In contrast, it depends on the isolation purity whether procaspase-3 is located in mitochondria of non-parenchymal liver cells, or not. All preparations with mitochondrial procaspase-3 fractions contain traces of haemoglobin, indicating the presence of some erythrocytes, which are the source of mitochondrial procaspase-3. Since erythrocytes migrate with mitochondria in subcellular fractionations, it is important to check for haemoglobin, before localizing the protein to mitochondria.




Similar content being viewed by others
References
Chandra D, Tang DG (2003) Mitochondrially localized active caspase-9 and caspase-3 result mostly from translocation from the cytosol and partly from caspase-mediated activation in the organelle. J Biol Chem 278:17408–17420
Samali A, Zhivotovsky B, Jones DP et al (1998) Detection of pro-caspase-3 in cytosol and mitochondria of various tissues. FEBS Lett 431:167–169
Mancini M, Nicholson DW, Roy S et al (1998) The caspase-3 precursor has a cytosolic and mitochondrial distribution: implications for apoptotic signaling. J Cell Biol 140:1485–1495
Ramuz O, Isnardon D, Devilard E et al (2003) Constitutive nuclear localization and initial cytoplasmic apoptotic activation of endogenous caspase-3 evidenced by confocal microscopy. Int J Exp Path 84:75–81
Chandler JM, Cohen GM, MacFarlane M (1998) Different subcellular distribution of caspase-3 and caspase-7 following Fas-induced apoptosis in mouse liver. J Biol Chem 273:10815–10818
van Loo G, Saelens X, Matthijssens F et al (2002) Caspases are not localized in mitochondria during life or death. Cell Death Diff 9:1207–1211
van Berkel TJC, Kruijt JK (1977) Different types of mitochondria in parenchymal and non-parenchymal rat-liver cells. Eur J Biochem 73:223–229
Krajewska M, Wang H-G, Krajewski S et al (1997) Immunohistochemical analysis of in vivo patterns of expression of CPP32 (caspase-3), a cell death protease. Cancer Res 57:1605–1613
Berry MN, Friend DS (1969) High yield preparation of isolated rat liver parenchymal cells. J Cell Biol 48:506–520
Werner S, Neupert W (1972) Functional and biogenetical heterogeneity of the inner membrane of rat-liver mitochondria. Eur J Biochem 25:379–396
Milisav I, Šuput D (2007) Procaspase-9 is attached to the mitochondrial outer membrane in the early stages of apoptosis. Cell Mol Biol Lett 12:509–522
Duan H, Orth K, Chinnaiyan AM et al (1996) ICE-LAP-6, a novel member of the ICE/Ced-3 gene family, is activated by the cytotoxic T cell protease granzyme B. J Biol Chem 271:16720–16724
Matocha MF, Waterman MR (1984) Discriminatory processing of the precursor forms of cytochrome P-450SCC and adrenodoxin by adrenocortical and heart mitochondria. J Biol Chem 259:8672–8678
Pelham HR, Jackson RJ (1976) An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem 67:247–256
Colin M, Moriz S, Schneider H et al (2000) Haemoglobin interferes with the ex vivo luciferase luminescence assay: consequence for detection of luciferase reporter gene expression in vivo. Gene Therapy 7:1333–1336
Castell JV, Jover R, Martinez-Jimenez CP et al (2006) Hepatocyte cell lines: their use, scope and limitations in drug metabolism studies. Expert Opin Drug Metab Toxicol 2:183–212
Okado-Matsumoto A, Fridovich I (2001) Subcellular distribution of superoxide dismutases (SOD) in rat liver. J Biol Chem 276:38388–38393
Geller BL, Winge DR (1982) Rat liver Cu, Zn-superoxide dismutase. J Biol Chem 257:8945–8952
Berg CP, Engels IH, Rothbart A et al (2001) Human mature red blood cells express caspase-3 and caspase-8, but are devoid of mitochondrial regulators of apoptosis. Cell Death Diff 8:1197–1206
Bratosin D, Estaquier J, Petit F et al (2001) Programmed cell death in mature erythrocytes: a model for investigating death effector pathways operating in the absence of mitochondria. Cell Death Diff 8:1143–1156
Acknowledgments
We thank Dr. Rok Romih for assistance with immunoelectron microscopy, Dr. Helena Podgornik for advice on erythrocyte detection, Dr. Marko Živin and Andrej Vovk for advices on band densitometry and statistics, respectively. Alenka Frangež Štrukelj and Dejan Bijelič helped us with preparation of this manuscript. This work was supported by grant P3-0019 from Ministry of Higher Education, Science and Technology of Republic of Slovenia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Milisav, I., Nipič, D. & Šuput, D. The riddle of mitochondrial caspase-3 from liver. Apoptosis 14, 1070–1075 (2009). https://doi.org/10.1007/s10495-009-0381-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-009-0381-3