Abstract
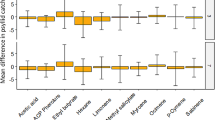
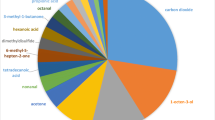
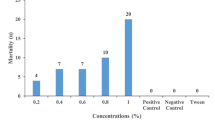
Adults of the Brown Ear Tick (Rhipicephalus appendiculatus) have a predilection for feeding inside and around the ears of cattle and other hosts. A previous study has shown that the tick locates the host ears by ‘push–pull’ deployment of a repellent blend emitted at the anal region and an attractant blend emitted at the ears. Interestingly, the two odours play reverse roles with Rhipicephalus evertsi, which prefer to feed around the anal region. The present study was undertaken to characterize the major constituents of the cattle anal odour and to evaluate their repellence to R. appendiculatus. The anal odour was trapped with reverse-phase C18-bonded silica, Porapak Q and Super Q placed in an oven bag attached at the anal region of the cattle for 6 h. The adsorbents were then removed and extracted with dichloromethane, and the extracted compounds analyzed by linked gas chromatography-mass spectrometry (GC–MS). The major constituents of the odour were o-xylene, 4-hydroxy-4-methyl-2-pentanone, 4-methyl-2-methoxyphenol, ethylbenzene, 2,6,6-trimethyl-[1S(1α,β,5α)]bicycloheptanes, 5-ethoxydihydro-2(3H)-furanone, 3-methylene-2-pentanone, 5-methyl-2-phenyl-1H-indole, and 3-pentanone. The repellency of the available compounds (o-xylene, 4-hydroxy-4-methyl-2-pentanone, 4-methyl-2-methoxyphenol, ethyl benzene, 3-methylene-2-pentanone, and 3-pentanone) and blends was evaluated using a dual choice tick climbing assay at different doses. The anal odour showed repellence with RD75 of 0.39. Of the compounds tested, 4-methyl-2-methoxyphenol was found to be most repellent (RD75 = 0.56) and 3-pentanone least repellent (RD75 = 622.7). The blend of the six constituents showed RD75 of 0.34, comparable to that of the crude anal odour blend. A series of subtractive bioassays with one constituent of the 6-component blend missing was also carried out. Subtraction of 3-methylpentanone gave the most repellent blend (RD75 = 0.097), whereas subtraction of 4-methylguaiacol gave the least repellent blend (RD75 = 160.7) consistent with the high individual activity of this phenol. The study lays down useful groundwork for on-host deployment of controlled-release of a selected repellent or blend to disrupt the tick’s ability to locate its preferred feeding site.

Similar content being viewed by others
References
African Union-Interafrican Bureau for Animal Resources (2011) Pan African animal health yearbook 2011. Pan African animal health yearbook, 2011:xiii + 90 pp. http://www.au-ibar.org/pan-african-animal-health-yearbook. Accessed 2 Dec 2017
Bett MK, Saini RK, Hassanali A (2015) Repellency of tsetse-refractory waterbuck (Kobus defassa) body odour to Glossina pallidipes (Diptera: Glossinidae): assessment of relative contribution of different classes and individual constituents. Acta Trop 146:17–24
Cunningham MP, Brown CG, Burridge MJ, Purnell RE (1973) Cryopreservation of infective particles of Theileria parva. Int J Parasitol 3:583–587
De Deken R, Martin V, Saido A, Madder M, Brandt J, Geysen D (2007) An outbreak of east coast fever on the Comoros: a consequence of the import of immunized cattle from Tanzania? Vet Parasitol 143:245–253
Drummond R (1983) Tick-borne livestock diseases and their vectors. Chemical control of ticks. Wild Anim Revolut (FAO) 36:28–33
Garcia-Garcia JC, Montero C, Redondo M, Vargas M, Canales M, Boue O, Rodriguez M, Joglar M, Machado H, Gonzalez IL (2000) Control of ticks resistant to immunization with Bm86 in cattle vaccinated with recombinant antigen Bm95 isolated from the cattle tick, Boophilus microplus. Vaccine 18:2275–2287
George J (2000) Present and future technologies for tick control. Ann NY Acad Sci 916:583–588
Irvin AD, Brocklesby DW (1970) Rearing and maintaining Rhipicephalus appendiculatus in the laboratory. J Inst Anim Tech 21:106
Kogo K, Michuki G, Okemo P, Skilton R, Kemp S, Muthee M, Hassanal A. The role of microbes in cattle ears in the production of volatile semiochemical blend attractive to the Brown Ear tick. Rhipicephalus appendiculatus (In Preparation)
Laffont C, Alvinerie M, Bousquet-Melou A, Toutain P (2001) Licking behavior and environmental contamination arising from pour-on ivermectin for cattle. Int J Parasitol 31:1687–1692
Mbogo KS, Kariuki PD, McHardy N, Payne R (1995) Training manual for veterinary staff on immunization against East Coast fever using the ECFiM system. Kenya Agricultural Research Institute and Overseas Development Administration of UK
Norval R, Perry D, Young S (1992) The epidemiology of theileriosis in Africa. Academic Press, London, p 481
Saini RK, Hassanali A (2007) A 4-alkyl-substituted analogue of guaiacol shows greater repellency to Savannah Tsetse (Glossina spp.). J Chemo Ecol 33:985–995
Saini RK, Orindi BO, Mbahin N, Andoke JA, Muasa PN, Mbuvi DM, Muya CM, Pickett JA, Borgemeister CW (2017) Protecting cows in small holder farms in East Africa from tsetse flies by mimicking the odor profile of a non-host bovid neglected tropical diseases. PLoS Negl Trop Dis 11:10
Spickett AM (1998) Acaricide and resistance. Vet Ectoparasitol Protozool 1:113
Tholl D, Bolland W, Hansel A, Loreto F, Schnitzer J (2006) Techniques for molecular analysis. Plant J 45:540
Wachira B, Mireji P, Okoth S, Ng’ang’a M, William J, Grace A, Hassanali A (2016) Responses of Glossina pallidipes and Glossina morsitans morsitans tsetse flies to analogues of δ-octalactone and selected blends. Acta Trop 160:53–57
Walker BJ (1974) The ixodid ticks of Kenya: a review of present knowledge of their hosts and distribution. The Eastern Press Limited, London
Wanzala W, Sika N, Gule S, Hassanali A (2004) Attractive and repellent host odours that guide ticks to their respective feeding sites. Chemoecology 14:229–232
Young A, Groocock C, Kariuki D (1988) Integrated control of ticks and tick borne diseases of cattle in Africa. Parasitology 96:403–432
Acknowledgement
This work was supported by funds from National Council of Science Technology (Grant No. NCST/5/003/W44). We also acknowledge Dr Rajindar K. Saini, of ICIPE for facilitating in the purchase of synthetic chemicals, Wanyama Kaye (ICIPE) for assisting in running GC–MS of the cattle anal odours, and E. Maina Kenyatta University for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Human and animal rights statement
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Kariuki, M.W., Hassanali, A. & Ng’ang’a, M.M. Characterisation of cattle anal odour constituents associated with the repellency of Rhipicephalus appendiculatus. Exp Appl Acarol 76, 221–227 (2018). https://doi.org/10.1007/s10493-018-0304-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-018-0304-5