Abstract
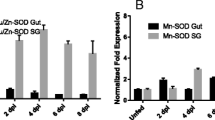
Amblyomma americanum (L.) ticks continue to emerge as disease vectors in many areas of the United States. Tick macrophage migration inhibitory factor (MIF) was first identified in A. americanum females and has been demonstrated to inhibit macrophage movement to the same extent as human MIF. This study was conducted to further characterize and elucidate the physiological role for MIF in tick feeding. A relative quantitative PCR assay was developed to determine the level of MIF gene expression during tick feeding. In addition, RNAi techniques were used to silence MIF prior to blood feeding. Physiological parameters of tick engorgement weight, length of feeding interval, and egg masses were observed to check for phenotypic manifestations of RNA silencing. Specific tick MIF antibody was used to localize MIF protein in frozen tick tissue sections. Tissue specific gene expression indicated that the midgut tissues were the most highly enriched for the MIF. Levels of gene expression did not parallel MIF protein pools seen in tissue sections. Of particular importance was the finding that unfed tick salivary glands appear to contain vesicles that are specific for MIF protein. This is the first demonstration of a pool of MIF that could be secreted during the first hours of tick feeding. While MIF silencing was demonstrated at the molecular level, no physiological phenotype was apparent. The MIF protein pools already available in the tissues may be sufficient to accomplish female tick feeding. Our studies show that the most prominent source of MIF during tick feeding is the midgut tissue. Future studies will address the role of MIF in blood feeding and nutrient digestion in the immature life stages of the tick.




Similar content being viewed by others
References
Abdul AM, Tsuji N, Miyoshi T, Islam MK, Hatti T, Fujisaki K (2009) Legumains from the hard tick Haemaphysalis longicornis play modulatory roles in blood feeding and gut cellular remodeling and impact on embryogenesis. Int J Parasitol 39:97–107
Alberti G, Coons LB (1999) Chapter 5 Acari: Ticks. In: Harrison FW, Foelix RF (eds) Microscopic anatomy of invertebrates, vol 8B. Wiley-Liss, New York, pp 373–403
Aljamali MN, Bior AD, Sauer JR, Essenberg RC (2003) RNA interference in ticks: a study using histamine binding protein dsRNA in the female tick Amblyomma americanum. Insect Mol Biol 12:299–305
Aljamali MN, Hern L, Kupfer D, Downard S, So S, Roe BA, Sauer JR, Essenberg RC (2009) Transcriptome analysis of the salivary glands of the female tick Amblyomma americanum (Acari: Ixodidae). Insect Mol Biol 18:129–154
Anderson JM, Sonenshine DE, Valenzuela JG (2008) Exploring the mialome of ticks: An annotated catalogue of midgut transcripts from the hard tick, Dermacentor variabilis (Acari: Ixodidae). BMC Genomics 9:89
Coons LB, Rosell-Davis R, Tarnowski BI (1986) Bloodmeal digestion in ticks. In: Sauer JR, Hair JA (eds) Morphology, physiology and behavioral biology of ticks. Ellis Horwood Ltd, Chichester, pp 248–279
de la Fuente J, Kocan KM (2006) Strategies for development of vaccines for control of ixodid tick species. Parasite Immunol 28:275–283
de la Fuente JD, Almazan C, Naranjo V, Blouin EF, Kocan KM (2006) Synergistic effect of silencing the expression of tick protective antigens 4D8 and Rs86 in Rhipicephalus sanguineus by RNA interference. Parasitol Res 99:108–113
Dickinson RG, O’Hagen JE, Schotz M, Binnington KC, Hegarty MP (1979) Studies on the significance of smooth muscle contracting substances of the cattle tick, Boophilus microplus. J Aust Entomol Soc 18:199–210
Fernando SC, Buck JS, Ashworth MD, Ross JW, Geisert RD, DeSilva U (2006) Porcine endometrial and conceptus tissue Kalliekrein 1, 4, 11 and 14 gene expression. Reprod Res 132:939–947
Francischetti I, Sa-Nunes A, Mans BJ, Santos IM, Ribeiro JMC (2009) The role of saliva in tick feeding. Front Biosci 14:2051–2088
Jaworski DC, Needham GR (1996) Calreticulin in vector arthropods—the secreted Calreticulin. In: Michalak M (ed) Calreticulin. R.G. Landes, Austin, pp 171–182
Jaworski DC, Jasinskas A, Metz CN, Bucala R, Barbour AG (2001) Identification and characterization of a homologue of the pro-inflammatory cytokine macrophage migration inhibitory factor in the tick, Amblyomma americanum. Insect Mol Biol 10:323–331
Jaworski DC, Bowen CJ, Wasala NB (2009) Amblyomma americanum (L): tick macrophage migration inhibitory factor peptide immunization lengthens lone star tick feeding intervals in vivo. Exp Parasitol 121:384–387
Labuda M, Trimnell AR, Lickova M, Kazimirova M, Davies GM, Lissina O, Hails RS, Nuttall PA (2006) An antivector vaccine protects against a lethal vector-borne pathogen. Plos Pathog 2:251–259
Mulenga A, Khumthong R, Chalaire KC (2009) Ixodes scapularis tick serine proteinase inhibitor (serpin) gene family; annotation and transcriptional analysis. BMC Genomics 10:217
Patrick CD, Hair JA (1975) Laboratory rearing procedures and equipment for multi-host ticks (Acarina: Ixodidae). J Med Entomol 12:389–390
Umemiya R, Hatta T, Liao M, Tanaka M, Zhou JL, Inoue N, Fujisaki K (2007) Haemaphysalis longicornis: molecular characterization of a homologue of the macrophage migration inhibitory factor from the partially fed ticks. Exp Parasitol 115:135–142
Acknowledgments
The authors thank S. C. Fernando and U. DeSilva for assistance in designing primers and developing qPCR assays. In addition, we acknowledge the helpful review of the manuscript by A. Wayandande and U. DeSilva. This article was approved for publication by the Director of Oklahoma Agricultural Experiment Station and supported under project OKL02623.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bowen, C.J., Jaworski, D.C., Wasala, N.B. et al. Macrophage migration inhibitory factor expression and protein localization in Amblyomma americanum (Ixodidae). Exp Appl Acarol 50, 343–352 (2010). https://doi.org/10.1007/s10493-009-9324-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-009-9324-5