Abstract
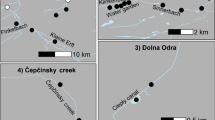
Intracellular bacteria of the genus Wolbachia (alpha Proteobacteria) induce cytoplasmic incompatibility (CI) in many arthropod species, including spider mites, but not all Wolbachia cause CI. In spider mites CI becomes apparent by a reduced egg hatchability and a lower daughter:son ratio: CI in haplodiploid organisms in general was expected to produce all-male offspring or a male-biased sex ratio without any death of eggs. In a previous study of Japanese populations of Tetranychus urticae, two out of three green-form populations tested were infected with non-CI Wolbachia strains, whereas none of six red-form populations harbored Wolbachia. As the survey of Wolbachia infection in T. urticae is still fragmentary in Japan, we checked Wolbachia infection in thirty green-form populations and 29 red-form populations collected from a wide range of Japanese islands. For Wolbachia-infected populations, we tested the effects of Wolbachia on the reproductive traits and determined the phylogenetic relationships of the different strains of Wolbachia. All but one green-form populations were infected with Wolbachia and all strains belonged to the subgroup Ori when the wsp gene was used to determine the phylogenetic relationships of different strains of Wolbachia. Six out of 29 red-form populations harbored Wolbachia and the infected strains belonged to the subgroups Ori and Bugs. Twenty-four of 29 infected green-form populations and five of six infected red-form populations induced CI among the hosts. Thus, CI-Wolbachia strains are widespread in Japan, and no geographical trend was observed in the CI-Wolbachia. Although three red-form populations harbored other intracellular bacteria Cardinium, they did not affect host reproduction.


Similar content being viewed by others
References
Baldo L, Lo N, Werren JH (2005) Mosaic nature of the Wolbachia surface protein. J Bacteriol 187:5406–5418
Baldo L, Bordenstei S, Wernegreen JJ, Werren JH (2006) Widespread recombination throughout Wolbachia genomes. Mol Biol Evol 23: 437–449
Breeuwer JAJ (1997) Wolbachia and cytoplasmic incompatibility in the spider mites Tetranychus urticae and T. turkestani. Heredity 79:41–47
Breeuwer JAJ, Werren JH (1990) Microorganisms associated with chromosome destruction and reproductive isolation between two insect species. Nature 346:558–560
Dupont LM (1979) On gene flow between Tetranychus urticae Koch, 1836 and Tetranychus cinnabarinus(Boisduval) Boudreaux, 1956 (Acari: Tetranychidae): synonymy between the two species. Entomol Exp Appl 25:297–303
Giordano R, O’Neill SL, Robertson HM (1995) Wolbachia infections and the expression of cytoplasmic incompatibility in Drosophila sechellia and D. mauritiana. Genetics 140: 1307–1317
Goka K, Takafuji A, Toda S, Hamamura T, Osakabe Mh, Komazaki S (1996) Genetic distinctness between two forms of Tetranychus urticae Koch (Acari: Tetranychidae) detected by electrophoresis. Exp Appl Acarol 20:683–693
Gomi K, Gotoh T, Noda H (1997) Wolbachiahaving no effect on reproductive incompatibility in Tetranychus kanzawai Kishida (Acari: Tetranychidae). Appl Entomol Zool 32:485–490
Gotoh T, Noda H, Fujita T, Iwadate K, Higo Y, Saito S, Ohtsuka S (2005) Wolbachia and nuclear-nuclear interactions contribute to reproductive incompatibility in the spider mite Panonychus mori (Acari: Tetranychidae). Heredity 94:237–246
Gotoh T, Noda H, Hong X-Y (2003) Wolbachia distribution and cytoplasmic incompatibility based on a survey of 42 spider mite species (Acari:Tetranychidae) in Japan. Heredity 91:208–216
Gotoh T, Noda H, Ito S (2007) Cardinium symbionts cause cytoplasmic incompatibility in spider mites. Heredity 98:13–20
Gotoh T, Oku H, Moriya K, Odawara M (1995) Nucleus-cytoplasm interactions causing reproductive incompatibility between two populations of Tetranychus quercivorus Ehara et Gotoh (Acari: Tetranychidae). Heredity 74:405–414
Gotoh T, Sugasawa J, Nagata T (1999) Reproductive compatibility of the two-spotted spider mite (Tetranychus urticae) infected with Wolbachia. Entomol Sci 2:289–295
Groot TVM, Breeuwer JAJ (2006) Cardinium symbionts induce haploid thelytoky in most clones of three closely related Brevipalpus species. Exp Appl Acarol 39:257–271
Helle W, van de Bund CF (1962) Crossbreeding experiments with some species of the Tetranychus urticae group. Entomol Exp Appl 5: 159–165
Hoffmann AA, Clancy DJ, Duncan J (1996) Naturally-occurring Wolbachia infection in Drosophila simulans that does not cause cytoplasmic incompatibility. Heredity 76:1–8
Hoffmann AA, Clancy DJ, Merton E (1994) Cytoplasmic incompatibility in Australian populations of Drosophila melanogaster. Genetics 136:993–999
Holden PR, Brookfield JFY, Jones P (1993) Cloning and characterization of an ftsZ homologue from a bacterial symbiont of Drosophila melanogaster. Mol Gen Genet 240: 213–220
Hunter MS, Perlman SJ, Kelly SE (2003) A bacterial symbiont in the Bacteroides induces cytoplasmic incompatibility in the parasitoid wasp Encarsia pergandiella. Proc R Soc London B270: 2185–2190
Ikeda T, Ishikawa H, Sasaki T (2003) Infection density of Wolbachia and level of cytoplasmic incompatibility in the Mediterranean flour moth, Ephestia kuehniella. J Invert Pathol 84:1–5
Jiggins FG, Bentley JK, Majerus MEN, Hurst GDD (2002) Recent changes in phenotype and patterns of host specialization in Wolbachia bacteria. Mol Ecol 11:1275–1283
Kittayapong P, Mongkalangoon P, Baimai V, O’Neill SL (2002) Host age effect and expression of cytoplasmic incompatibility in field populations of Wolbachia-superinfected Aedes albopictus. Heredity 88:270–274
Muraji M, Kawasaki K, Shimizu T (2000) Phylogenetic utility of nucleotide sequences of mitochondrial 16S ribosomal RNA and cytochrome b genes in anthocorid bugs (Heteroptera:Anthocoridae). Appl Entomol Zool 35:293–300
Noda H, Koizumi Y, Zhang Q, Deng K (2001) Infection density of Wolbachia and incompatibility level in two planthopper species, Laodelphax striatellus and Sogatella furcifera. Insect Biochem Mol Biol 31:727–737
O’Neill SL, Giordano R, Colbert AME, Karr TL, Robertson HM (1992) 16S rRNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc Natl Acad Sci USA 89:2699–2702
O’Neill SL, Hoffmann AA, Werren JH (eds) (1997) Influential passengers: Inherited microorganisms and arthropod reproduction. Oxford Univ Press, New York, p 214
Perrot-Minnot MJ, Cheval B, Migeon A, Navajas M (2002) Contrasting effects of Wolbachia on cytoplasmic incompatibility and fecundity in the haplodiploid mite Tetranychus urticae. J Evol Biol 15:808–817
Sokal RR, Rohlf FJ (1995) Biometry 3rd edn. WH Freeman & Company, New York, p 887
SPSS. (2002) SPSS 11.5J Brief guide. SPSS Inc, Tokyo, p 160 (in Japanese)
Stouthamer R, Luck RF, Hamilton WD (1990) Antibiotics cause parthenogenetic Trichogramma (Hymenoptera/ Trichogrammatidae) to revert to sex. Proc Natl Acad Sci 87:2424–2427
Stouthamer R, Breeuwer JAJ, Hurst HGD (1999) Wolbachia pipientis: microbial manipulator of arthropod reproduction. Annu Rev Microbiol 53:71–102
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL X windows interface:flexible strategies for multiple sequence alignment aided by quality analysis tool. Nucl Acids Res 25:4876–4882
Vala F, Breeuwer JAJ, Sabelis MW (2000) Wolbachia-induced ‘hybrid breakdown’ in the two-spotted spider mite Tetranychus urticae Koch. Proc R Soc Lond Ser B 267:1931–1937
Vala F, Breeuwer JAJ, Sabelis MW (2003) No variation for Wolbachia-induced hybrid breakdown in two populations of a spider mite. Exp Appl Acarol 29:1–12
van Opijnen T, Breeuwer JAJ (1999) High temperatures eliminate Wolbachia, a cytoplasmic incompatibility inducing endosymbiont, from the two-spotted spider mite. Exp Appl Acarol 23:871–881
Vavre F, Fleury F, Varaldi J, Fouillet P, Bouletreau M (2000) Evidence for female mortality in Wolbachia-mediated cytoplasmic incompatibility in haplodiploid insects:epistemiologic and evolutionary consequences. Evolution 54:191–200
Weeks AR, Marec B, Breeuwer JAJ (2001) A mite species that consists entirely of haploid females. Science 292:2479–2482
Werren JH, Windsor DM, Guo LR (1995a) Distribution of Wolbachia among neotropical arthropods. Proc R Soc Lond Ser B 262: 197–204
Werren JH, Zhang W, Guo LR (1995b) Evolution and phylogeny of Wolbachia: reproductive parasites of arthropods. Proc R Soc Lond Ser B 261:55–63
Xie R-R, Liu Y, Hong X-Y, Gotoh T (2006) Effect of infection rate of Wolbachia on the reproduction in Tetranychus kanzawai Kishida (Acari:Tetranychidae) in China and Taiwan. Internat J Acarol 32: 407–415
Yamamura K (2002) Transformation formulae for performing correct ANOVA. Plant Protect 56:436–441 (in Japanese)
Zchori-Fein E, Gottlieb Y, Kelly SE, Brown JK, Wilson JM, Karr TL, Hunter MS (2001) A newly discovered bacterium associated with parthenogenesis and a change in host selection behavior in parasitoid wasps. Proc Natl Acad Sci USA 98:12555–12560
Zhou WG, Rousset F, O’Neill SL (1998) Phylogeny and PCR-based classification of Wolbachia strains using wsp gene sequences. Proc R Soc Lond Ser B 265:509–515
Acknowledgements
We thank Sawako Kawai, Saiko Ito and Yuki Nakamura for their kind assistance. We are also grateful to two anonymous reviewers for their critical comments on an earlier draft. This work was supported in part by Grants-in-Aid for Scientific Research (Nos. 15380040 and 16580041) from the Japan Society of Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Gotoh, T., Sugasawa, J., Noda, H. et al. Wolbachia-induced cytoplasmic incompatibility in Japanese populations of Tetranychus urticae (Acari: Tetranychidae). Exp Appl Acarol 42, 1–16 (2007). https://doi.org/10.1007/s10493-007-9072-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-007-9072-3