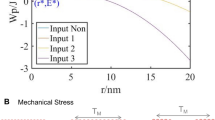
Abstract
With the advantages of biosafety and efficiency, increasing attention has been paid to the devices for gene and macromolecular drug delivery based on mechanoporation. The transient pore formation on the cell membrane allows cargo transportation when the membrane areal strain is beyond the critical pore value and below the lysis tension threshold. Based on this principle, we propose a method to apply the proper fluid stress on cells moving in a microchannel under the action of zero-net-mass-flux (ZNMF) jets. In this study, an immersed finite element method (IFEM) is adopted to simulate the interaction between the cells and the fluid fields so as to investigate the cell movement and deformation in this mechanoporation system. To evaluate the efficiency of the cargo delivery, a pore integral is defined as the mean pore rate when the cell passes through the jet region. By analyzing the effects of the parameters, including the pressure gradient along the microchannel, the jet amplitude, and the jet frequency, on the pore integrals, a group of optimized parameters for cargo delivery efficiency are obtained. Additionally, the stability and safety of this system are analyzed in detail. These results are helpful in designing the mechanoporation devices and improving their efficiency of drug delivery.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
MARTIN, P. S., ROBERT, L., and KLAVS, F. J. Intracellular delivery by membrane disruption: mechanisms, strategies, and concepts. Chemical Reviews, 118(16), 7409–7531 (2018)
STEWART, M. P., SHAREI, A., DING, X., SAHAY, G., LANGER, R., and JENSEN, K. F. In vitro and ex vivo strategies for intracellular delivery. nature, 538(7624), 183–192 (2016)
HUR, J. and CHUNG, A. J. Microfluidic and nanofluidic intracellular delivery. Advanced Science, 8(15), 2004595 (2021)
ZHANG, P., SHAO, N., and QIN, L. Recent advances in microfluidic platforms for programming cell-based living materials. Advanced Materials, 33(46), 2005944 (2021)
SHAREI, A., ZOLDAN, J., ADAMO, A., SIM, W. Y., CHO, N., JACKSON, E., MAO, S., SCHNEIDER, S., HAN, M. J., LYTTON-JEAN, A., BASTO, P. A., JHUNJHUNWALA, S., LEE, J., HELLER, D. A., KANG, J. W., HARTOULAROS, G. C., KIM, K. S., ANDERSON, D. G., LANGER, R., and JENSEN, K. F. A vector-free microfluidic platform for intracellular delivery. Proceedings of the National Academy of Sciences, 110(6), 2082–2087 (2013)
KOLLMANNSPERGER, A., SHAREI, A., RAULF, A., HEILEMANN, M., LANGER, R., JENSEN, K. F., WIENEKE, R., and TAMPE, R. Live-cell protein labelling with nanometre precision by cell squeezing. Nature Communications, 7(1), 1–7 (2016)
DENG, Y., KIZER, M., RADA, M., SAGE, J., WANG, X., CHEON, D. J., and CHUNG, A. J. Intracellular delivery of nanomaterials via an inertial microfluidic cell hydroporator. Nano Letters, 18(4), 2705–2710 (2018)
KIZER, M. E., DENG, Y., KANG, G., MIKAEL, P. E., WANG, X., and CHUNG, A. J. Hydroporator: a hydrodynamic cell membrane perforator for high-throughput vector-free nanomaterial intracellular delivery and DNA origami biostability evaluation. Lab on a Chip, 19(10), 1747–1754 (2019)
KANG, G., CARLSON, D. W., KANG, T. H., LEE, S., HAWARD, S. J., CHOI, I., SHEN, A. Q., and CHUNG, A. J. Intracellular nanomaterial delivery via spiral hydroporation. ACS Nano, 14(3), 3048–3058 (2020)
HUR, J., PARK, I., LIM, K. M., DOH, J., CHO, S. G., and CHUNG, A. J. Microfluidic cell stretching for highly effective gene delivery into hard-to-transfect primary cells. ACS Nano, 14(11), 15094–15106 (2020)
MODARESI, S., PACELLI, S., SUBHAM, S., DATHATHREYA, K., and PAUL, A. Intracellular delivery of exogenous macromolecules into human mesenchymal stem cells by double deformation of the plasma membrane. Advanced Therapeutics, 3(1), 1900130 (2020)
INGRD, U. and LABATE, S. Acoustic circulation effects and the nonlinear impedance of orifices. Journal of the Acoustical Society of America, 22(2), 211–218 (1950)
WILTSE, J. M. and GLEZER, A. Manipulation of free shear flows using piezoelectric actuators. Journal of Fluid Mechanics, 249, 261–285 (1993)
ZHANG, P. F., WANG, J. J., and FENG, L. H. Review of zero-net-mass-flux jet and its application in separation flow control. Science China: Technological Sciences, 51(9), 1315–1344 (2008)
AI, J. F., XIE, J., and HU, G. H. Numerical simulation of red blood cells deformation in microchannel under zero-net-mass-flux jet. Acta Physica Sinica, 69, 234701 (2020)
MALLINSON, S. G., JOHNSON, G., GASTON, M., and HONG, G. Three-Dimensional Numerical Simulations of Synthetic Jet Actuator Flows in a Microchannel, Australia, 341–350 (2004)
PESKIN, C. S. Numerical analysis of blood flow in the heart. Journal of Computational Physics, 25(3), 220–252 (1977)
MITTAL, R. and IACCARINO, G. Immersed boundary methods. Annual Review of Fluid Mechanics, 37, 239–261 (2005)
GOLDSTEIN, D., HANDLER, R., and SIROVICH, L. Modeling a no-slip flow boundary with an external force field. Journal of Computational Physics, 105(2), 354–366 (1993)
PESKIN, C. S. The immersed boundary method. Acta Numerica, 11, 479–517 (2002)
WANG, X. and LIU, W. K. Extended immersed boundary method using FEM and RKPM. Computer Methods in Applied Mechanics and Engineering, 193(12–14), 1305–1321 (2004)
ZHANG, L., GERSTENBERGER, A., WANG, X., and LIU, W. K. Immersed finite element method. Computer Methods in Applied Mechanics and Engineering, 193(21–22), 2051–2067 (2004)
LEE, T. R., CHOI, M., KOPACZ, A. M., YUN, S. H., LIU, W. K., and DECUZZI, P. On the near-wall accumulation of injectable particles in the microcirculation: smaller is not better. Scientific Reports, 3(1), 1–8 (2013)
LIU, W. K., LIU, Y., FARRELL, D., ZHANG, L., WANG, X. S., FUKUI, Y., PATANKAR, N., ZHANG, Y., BAJAJ, C., LEE, J., HONG, J., CHEN, X., and HSU, H. Immersed finite element method and its applications to biological systems. Computer Methods in Applied Mechanics and Engineering, 195(13–16), 1722–1749 (2006)
LIU, Y. and LIU, W. K. Rheology of red blood cell aggregation by computer simulation. Journal of Computational Physics, 220(1), 139–154 (2006)
TAN, J., THOMAS, A., and LIU, Y. Influence of red blood cells on nanoparticle targeted delivery in microcirculation. Soft Matter, 8(6), 1934–1946 (2012)
TEZDUYAR, T. E. Stabilized finite element formulations for incompressible flow computations. Advances in Applied Mechanics, 28, 1–44 (1991)
TEZDUYAR, T. E. Finite element methods for flow problems with moving boundaries and interfaces. Archives of Computational Methods in Engineering, 8(2), 83–130 (2001)
HUGHES, T. J. R., FRANCA, L. P., and BALESTRA, M. A new finite element formulation for computational fluid dynamics: V. circumventing the Babuška-Brezzi condition: a stable Petrov-Galerkin formulation of the Stokes problem accommodating equal-order interpolations. Computer-Methods in Applied Mechanics and Engineering, 59(1), 85–99 (1986)
LIU, W. K., JUN, S., and ZHANG, Y. F. Reproducing kernel particle methods. International Journal for Numerical Methods in Fluids, 20(8–9), 1081–1106 (1995)
MAAS, S. A., ELLIS, B. J., ATESHIAN, G. A., and WEISS, J. A. FEBio: finite elements for biomechanics. Journal of Biomechanical Engineering, 134(1), 011005 (2012)
LIU, F., WU, D., and CHEN, K. Mechanical behavior of cells in microinjection: a minimum potential energy study. Journal of the Mechanical Behavior of Biomedical Materials, 24, 1–8 (2013)
WANG, Q., MANMI, K., and LIU, K. K. Cell mechanics in biomedical cavitation. Interface Focus, 5(5), 20150018 (2015)
SKALAK, R., TOZEREN, A., ZARDA, R. P., and CHIEN, S. Strain energy function of red blood cell membranes. Biophysical Journal, 13(3), 245–264 (1973)
POPEL, A. S. and JOHNSON, P. C. Microcirculation and hemorheology. Annual Review of Fluid Mechanics, 37, 43–69 (2005)
LANOTTE, L., MAUER, J., MENDEZ, S., FEDOSOV, D. A., FROMENTAL, J. M., CLAVERIA, V., NICOUD, F., GOMPPER, G., and ABKARIAN, M. Red cells’ dynamic morphologies govern blood shear thinning under microcirculatory flow conditions. Proceedings of the National Academy of Sciences, 113(47), 13289–13294 (2016)
EVANS, E., HEINRICH, V., LUDWIG, F., and RAWICZ, W. Dynamic tension spectroscopy and strength of biomembranes. Biophysical Journal, 85(4), 2342–2350 (2003)
EVANS, E. A., WAUGH, R., and MELNIK, L. Elastic area compressibility modulus of red cell membrane. Biophysical Journal, 16(6), 585–595 (1976)
SHIGEMATSU, T., KOSHIYAMA, K., and WADA, S. Effects of stretching speed on mechanical rupture of phospholipid/cholesterol bilayers: molecular dynamics simulation. Scientific Reports, 5(1), 1–10 (2015)
MCNEIL, P. L. and STEINHARDT, R. A. Plasma membrane disruption: repair, prevention, adaptation. Annual Review of Cell and Developmental Biology, 19, 697–731 (2003)
OMORI, T., ISHIKAWA, T., BARTHES-BIESEL, D., SALSAC, A. V., IMAI, Y., and YAM-AGUCHI, T. Tension of red blood cell membrane in simple shear flow. Physical Review E, 86(5), 056321 (2012)
TOLPEKINA, T. V., DEN OTTER, W. K., and BRIELS, W. J. Simulations of stable pores in membranes: system size dependence and line tension. Journal of Chemical Physics, 121(16), 8014–8020 (2004)
Author information
Authors and Affiliations
Corresponding author
Additional information
Citation: LIU, X. Y., AI, J. F., XIE, J., and HU, G. H. Numerical study of opposed zero-netmass-flow jet-induced erythrocyte mechanoporation. Applied Mathematics and Mechanics (English Edition), 43(11), 1763–1776 (2022) https://doi.org/10.1007/s10483-022-2931-6
Project supported by the National Natural Science Foundation of China (Nos. 11832017 and 12172209) and the Key Research Project of Zhejiang Laboratory of China (No. 2021PE0AC02)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, X., Ai, J., Xie, J. et al. Numerical study of opposed zero-net-mass-flow jet-induced erythrocyte mechanoporation. Appl. Math. Mech.-Engl. Ed. 43, 1763–1776 (2022). https://doi.org/10.1007/s10483-022-2931-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10483-022-2931-6
Key words
- drug delivery
- membrane mechanoporation
- zero-net-mass-flux (ZNMF) jet
- immersed finite element method (IFEM)