Abstract
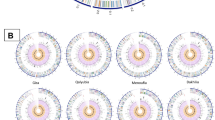
Trueperella pyogenes, an opportunistic pathogen causes various ailments in different animals. Different strains from different animals have distinct characters phenotypically and genotypically. Hence understanding the strains in a particular geographical location helps in framing the preventive measures. Comparative genomics of all the available T. pyogenes genome in the NCBI was conducted to understand the relatedness among strains. Whole genome phylogeny showed host associated clustering of strains recovered from swine lungs. Core genome phylogeny also showed host associated clustering mimicking whole genome phylogeny results. MLST analysis showed that there was higher diversity among cattle strains. Multidimensional scaling revealed five swine clusters, two cattle and buffalo clusters. Pangenome analysis also showed that T. pyogenes had an open genome with 57.09% accessory genome. Host specific genes were identified by pangenome analysis, and (R)-citramalate synthase was specific for swine strains of Asian origin. Host specifc genes identified by pangenome analysis can be exploited for developing a molecular assay to specifically identify the strains. The study shows that MLST having higher discriminatory power can be used as an epidemiological tool for strain discrimination of T. pyogenes.





Similar content being viewed by others
Data availability
The data used to support the findings of this study is included in the article. Supporting data are included as supportive tables.
References
Ashrafi Tamai I, Mohammadzadeh A, Zahraei Salehi T, Mahmoodi P (2018) Genomic characterisation, detection of genes encoding virulence factors and evaluation of antibiotic resistance of Trueperella pyogenes isolated from cattle with clinical metritis. Antonie Van Leeuwenhoek 111:2441–2453
Croucher NJ, Page AJ, Connor TR, Delaney AJ, Keane JA, Bentley SD, Parkhill J, Harris SR (2015) Rapid phylogenetic analysis of large samples of recombinant bacterial whole genome sequences using Gubbins. Nucleic Acids Res 43:e15
Diene SM, Merhej V, Henry M, El Filali A, Roux V, Robert C, Azza S, Gavory F, Barbe V, La Scola B, Raoult D, Rolain JM (2013) The rhizome of the multidrug-resistant Enterobacter aerogenes genome reveals how new “Killer Bugs” are created because of a sympatric lifestyle. Mol Biol Evol 30:369–383
Fujimoto H, Shimoji N, Sunagawa T, Kubozono K, Nakajima T, Chuma C (2020) Differences in phenotypic and genetic characteristics of Trueperella pyogenes detected in slaughtered cattle and pigs with septicemia. J Vet Med Sci 82:626–631. https://doi.org/10.1292/jvms.19-0370
Grim CJ, Kotewicz ML, Power KA, Gopinath G, Franco AA, Jarvis KG, Yan QQ, Jackson SA, Sathyamoorthy V, Hu L, Pagotto F, Iversen C, Lehner A, Stephan R, Fanning S, Tall BD (2013) Pan-genome analysis of the emerging foodborne pathogen Cronobacter spp. suggests a species-level bidirectional divergence driven by niche adaptation. BMC Genomics 14:366
Juhas M, Van der Meer JR, Gaillard M, Harding RM, Hood D, Crook DW (2009) Genomic islands: tools of bacterial horizontal gene transfer and evolution. FEMS Microbiol Rev 33:376–393. https://doi.org/10.1111/j.1574-6976.2008.00136.x
Karthik K, Anbazhagan S, Thomas P, Ananda Chitra M, Senthilkumar TMA, Sridhar R, Dhinakar Raj G (2021) Genome sequencing and comparative Genomics of Indian isolates of Brucella melitensis. Front Microbiol 12:698069. https://doi.org/10.3389/fmicb.2021.698069
Karthik K, Anbazhagan S, Chitra MA, Bharathi R, Venkataramanan R, Manobhavan M, Sundaram SM, Sridhar R (2022) Comparative genomics of Trueperella pyogenes available in the genome database reveals multidrug resistance genomic islands. J Global Antimicrob Resist S2213-7165(22)00225-9. https://doi.org/10.1016/j.jgar.2022.09.011
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547
Letunic I, Bork P (2021) Interactive tree of life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res 49:W293–W296
Marschall T, Marz M, Abeel T, Dijkstra L, Dutilh BE, Ghaffaari A, Kersey P, Kloosterman WP, Mäkinen V, Novak AM (2018) The computational Pan-Genomics Consortium. Computational pan-genomics: Status, promises and challenges. Brief Bioinform 19:118–135
Medini D, Donati C, Tettelin H, Masignani V, Rappuoli R (2005) The microbial pan-genome. Curr Opin Genet Dev 5:589–594
Nagib S, Glaeser SP, Eisenberg T, Sammra O, Lämmler C, Kämpfer P, Schauerte N, Geiger C, Kaim U, Prenger-Berninghoff E, Becker A, Abdulmawjood A (2017) Fatal infection in three Grey Slender Lorises (Loris lydekkerianus nordicus) caused by clonally related Trueperella pyogenes. BMC Vet Res 13:273. https://doi.org/10.1186/s12917-017-1171-8
Nourdin-Galindo G, Sánchez P, Molina CF, Espinoza-Rojas DA, Oliver C, Ruiz P, Vargas-Chacoff L, Cárcamo JG, Figueroa JE, Mancilla M, Maracaja-Coutinho V, Yañez AJ (2017) Comparative pan-genome analysis of Piscirickettsia salmonis reveals genomic divergences within genogroups. Front Cell Infect Microbiol 7:459. https://doi.org/10.3389/fcimb.2017.00459
Rasko DA, Rosovitz MJ, Myers GS, Mongodin EF, Fricke WF, Gajer P, Crabtree J, Sebaihia M, Thomson NR, Chaudhuri R, Henderson IR (2008) The pangenome structure of Escherichia coli: comparative genomic analysis of E. coli commensal and pathogenic isolates. J Bacteriol 190:6881–6893
Rogovskyy AS, Lawhon S, Kuczmanski K, Gillis DC, Wu J, Hurley H, Rogovska YV, Konganti K, Yang CY, Duncan K (2018) Phenotypic and genotypic characteristics of Trueperella pyogenes isolated from ruminants. J Vet Diagn Invest 30:348–353. https://doi.org/10.1177/1040638718762479
Rzewuska M, Kwiecień E, Chrobak-Chmiel D, Kizerwetter-Świda M, Stefańska I, Gieryńska M (2019) Pathogenicity and virulence of Trueperella pyogenes: a review. Int J Mol Sci 20:2737
Seemann T (2019) snp-dists. GitHub repository. GitHub. Retrieved from https://github.com/tseemann/snp-dists
Silva E, Gaivão M, Leitão S, Jost BH, Carneiro C, Vilela CL, Lopes da Costa L, Mateus L (2008) Genomic characterization of Arcanobacterium pyogenes isolates recovered from the uterus of dairy cows with normal puerperium or clinical metritis. Vet Microbiol 132:111–118
Tettelin H, Masignani V, Cieslewicz MJ, Donati C, Medini D, Ward NL, Angiuoli SV, Crabtree J, Jones AL, Durkin AS, Deboy RT, Davidsen TM, Mora M, Scarselli M, Margarit /Dodson RJ, Rosovitz MJ, Sullivan SA, Daugherty SC, Haft DH, Selengut J, Gwinn ML, Zhou L, Zafar N, Khouri H, Radune D, Dimitrov G, Watkins K, O’Connor KJ, Smith S, Utterback TR, White O, Rubens CE, Grandi G, Madoff LC, Kasper DL, Telford JL, Wessels MR, Rappuoli R, Fraser CM (2005) Genome analysis of multiple pathogenic isolates of Streptococcus agalactiae: implications for the microbial “pan-genome.” Proc Natl Acad Sci 102:13950–13955. https://doi.org/10.1073/pnas.0506758102
Tomasini N, Lauthier JJ, Llewellyn MS, Diosque P (2013) MLSTest: novel software for multi-locus sequence data analysis in eukaryotic organisms. Infect Genet Evol 20:188–196
Tonkin-Hill G, MacAlasdair N, Ruis C, Weimann A, Horesh G, Lees JA, Gladstone RA, Lo S, Beaudoin C, Floto RA, Frost SD (2020) Producing polished prokaryotic pangenomes with the Panaroo pipeline. Genome Biol 21:1–21
Trifinopoulos J, Nguyen LT, von Haeseler A, Minh BQ (2016) IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235
Zhao Y, Jia X, Yang J, Ling Y, Zhang Z, Yu J, Wu J, Xiao J (2014) PanGP: a tool for quickly analyzing bacterial pan-genome profile. Bioinformatics 30:1297–1299
Acknowledgements
The authors are thankful to Tamil Nadu Veterinary and Animal Sciences University for the facilities provided for carrying out the work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Kumaragurubaran Karthik: Conceptualization, isolation of T. pyogenes from the samples, formal analysis, writing original draft; Subbaiyan Anbazhagan: data curation, software analysis, updating original manuscript; Murugesan Ananda Chitra: Data curation, reviewing and editing of the draft; Ramaswamy Sridhar: Fund acquisition and administration.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karthik, K., Anbazhagan, S., Chitra, M.A. et al. Comparative phylogenomics of Trueperella pyogenes reveals host-based distinction of strains. Antonie van Leeuwenhoek 116, 343–351 (2023). https://doi.org/10.1007/s10482-022-01806-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-022-01806-9