Abstract
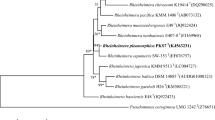
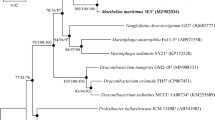
A Gram-stain-positive, motile, and rod-shaped bacterium, designated as strain MB25T, was isolated from the gut of Cyprinus carpio from the highly polluted river Yamuna, India. Phylogenetic analysis based on 16S rRNA gene sequence revealed that strain MB25T belonged to the genus Sporosarcina, sharing the highest sequence similarity with S. luteola Y1T (98.98%) and S. koreensis S-K12T (98.91%). Digital DNA-DNA hybridization and average nucleotide identity values of strain MB25T with strain Y1T and S-K12T were 18.9, 77.69, and 18.2, 76.80 respectively. Genome analysis of strain MB25T revealed its biotechnological properties such as tolerance to potent heavy metals, genes for the production of carbohydrate-active enzymes, antimicrobial compounds, and also degradation of aromatic compounds. The G + C content of strain MB25T genome was 45%. Growth observed at 10–40 °C (optimum, 28–30 °C), pH 6.0–8.5 (optimum pH 7.5–8.0); NaCl concentrations up to 6.0% (w/v). The dominant respiratory quinone was MK-7, cell wall peptidoglycan is of the A-4 type containing amino acids Lys-Glu and the major fatty acids are anteiso-C11:0 and iso-C15: 0. The major polar lipids of strain MB25T are diphosphatidylglycerol, phosphatidylglycerol, and phosphatidylethanolamine. On the basis of phenotypic, chemotaxonomic, phylogenetic, and phylogenomic data, strain MB25T represents a novel species of the genus Sporosarcina, for which the name Sporosarcina cyprini sp. nov. is proposed. The type strain is MB25T (= MCC 4366 T = JCM 34521 T = CCM 9113 T).


Similar content being viewed by others
Abbreviations
- DNA:
-
Deoxyribonucleic acid
- RNA:
-
Ribonucleic acid
- MUSCLE:
-
Multiple Sequence Comparison by Log-Expectation
- MEGA:
-
Molecular Evolutionary Genetics Analysis
- MCC:
-
Microbial Culture Collection
- JCM:
-
Japan Collection of Microorganisms
- CCM:
-
Czech Collection of Microorganisms
- DSMZ:
-
Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH
References
An SY, Haga T, Kasai H, Goto K, Yokota A (2007) Sporosarcina saromensis sp. nov., an aerobic endospore-forming bacterium. Int J Syst Evol Microbiol 57:1868–1871. https://doi.org/10.1099/ijs.0.649620
Asim M, Nageswara Rao K (2021) Assessment of heavy metal pollution in Yamuna River, Delhi-NCR, using heavy metal pollution index and GIS. Environ Monit Assess 193:1–16. https://doi.org/10.1007/s10661-021-08886-6
Aziz RK, Bartels D, Best AA et al (2008) The RAST server: rapid annotations using subsystems technology. BMC Genom 9:75. https://doi.org/10.1186/1471-2164-9-75
Bauer AW (1966) Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol 45:149–158
Belfort M, Wulff DL (1973) Genetic and biochemical investigation of the escherichia coli mutant hfl-1 which is lysogenized at high frequency by bacteriophage lambda. J Bacteriol 115:299–306. https://doi.org/10.1128/jb.115.1.299-306.1973
Bharti M, Nagar S, Khurana H, Negi RK (2022) Metagenomic insights to understand the role of polluted river Yamuna in shaping the gut microbial communities of two invasive fish species. Arch Microbiol 204:1–12. https://doi.org/10.1007/s00203-022-03127-x
Bitterlich G (1985) Digestive enzyme pattern of two stomachless filter feeders, silver carp, Hypophthalmichthys molitrix Val., and bighead carp Aristichthys Nobilis Rich. J Fish Biol 27:103–112. https://doi.org/10.1111/j.1095-8649.1985.tb04013.x
Blin K, Shaw S, Steinke K, Villebro R, Ziemert N, Lee SY, Medema MH, Weber T (2019) AntiSMASH 5.0: updates to the secondary metabolite genome mining pipeline. Nucl Acid Res 47:W81–W87. https://doi.org/10.1093/nar/gkz310
Brezna B, Kweon O, Stingley RL, Freeman JP, Khan AA, Polek B, Jones RC, Cerniglia CE (2006) Molecular characterization of cytochrome P450 genes in the polycyclic aromatic hydrocarbon degrading Mycobacterium vanbaalenii PYR-1. Appl Microbiol Biotechnol 71:522–532. https://doi.org/10.1007/s00253-005-0190-8
Claus D, Fahmy F, Rolf HJ, Tosunoglu N (1983) Sporosarcina halophila sp. nov., an obligate, slightly halophilic bacterium from salt marsh soils. Syst Appl Microbiol 4:496–506. https://doi.org/10.1016/S0723-2020(83)80007-1
Colpaert JV, Van Assche JA (1992) Zinc toxicity in ectomycorrhizal Pinus sylvestris. Plant Soil 143:201–211. https://doi.org/10.1007/BF00007874
Cowan ST, Steel KJ (1965) Manual for the identification of medical bacteria. Cambridge University Press, London
Dong X, Cai M (2001) Manual of systematic and determinative bacteriology beijing. Academic Press, China
Dubey AK, Baker CS, Suzuki K, Jones AD, Pandit P, Romeo T, Babitzke P (2003) CsrA regulates translation of the Escherichia coli carbon starvation gene, cstA, by blocking ribosome access to the cstA transcript. J Bacteriol 185:4450–4460. https://doi.org/10.1128/JB.185.15.4450-4460.2003
Edwards U, Rogall T, Blöcker H, Emde M, Böttger EC (1989) Isolation and direct complete nucleotide determination of entire genes. Characterisation of a gene coding for 16S ribosomal RNA. Nucleic Acid Res 17:7843–7853. https://doi.org/10.1093/nar/17.19.7843
Elsemore DA, Ornston LN (1995) Unusual ancestry of dehydratases associated with quinate catabolism in Acinetobacter calcoaceticus. J Bacteriol 177:5971–5978. https://doi.org/10.1128/jb.177.20.5971-5978.1995
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
FishBase (2021) Retrieved April 12, 2021. https://www.fishbasese/search.php?c_code=356#country
Frey AD, Kallio PT (2003) Bacterial hemoglobins and flavohemoglobins: versatile proteins and their impact on microbiology and biotechnology. FEMS Microbiol Rev 27:525–545. https://doi.org/10.1016/S0168-6445(03)00056-1
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, Tiedje JM (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Gupta RS, Patel S (2020) Robust demarcation of the family Caryophanaceae (Planococcaceae) and its different genera including three novel genera based on phylogenomics and highly specific molecular signatures. Front Microb 10:2821. https://doi.org/10.3389/fmicb.2019.02821
Huang X, Zhang R, Cui M, Lai H (2022) Experimental investigation on bioremediation of heavy metal contaminated solution by Sporosarcina pasteurii under some complex conditions. Water 14:595. https://doi.org/10.3390/w14040595
Huerta-Cepas J, Szklarczyk D, Forslund K, Cook H, Heller D et al (2016) eggNOG 4.5: a hierarchical orthology framework with improved functional annotations for eukaryotic, prokaryotic and viral sequences. Nucleic Acid Res 44:D286–D293. https://doi.org/10.1093/nar/gkv1248
Huyben D, Chiasson M, Lumsden JS, Pham PH, Chowdhury MAK (2021) Dietary microencapsulated blend of organic acids and plant essential oils affects intestinal morphology and microbiome of rainbow trout (Oncorhynchus mykiss). Microorganisms 9:2063. https://doi.org/10.3390/microorganisms9102063
Ianeva OD (2009) Mechanisms of bacteria resistance to heavy metals. Mikrobiol Z 71:54–65
Jackman SD, Vandervalk BP, Mohamadi H, Chu J, Yeo S, Hammond SA, Jahesh G, Khan H, Coombe L, Warren RL, Birol I (2017) ABySS 2.0: resource-efficient assembly of large genomes using a bloom filter. Genome Res 27(5):768–777. https://doi.org/10.1101/gr.214346.116
Joffe M (2003) Infertility and environmental pollutants. Br Med Bull 68:47–70. https://doi.org/10.1093/bmb/ldg025
Jones MP, McCarthy AJ, Cross T (1979) Taxonomic and serologic studies on Micropolyspora faeni and Micropolyspora strains from soil bearing the specific epithet rectivirgula. J Gen Microbiol 115:343–354. https://doi.org/10.1099/00221287-115-2-343
Jukes TH, Cantor CR (1969) Evolution of protein molecules. Mamm Protein Metab 3:21–132
Kämpfer P, Falsen E, Lodders N, Schumann P (2010) Sporosarcina contaminans sp. nov. and Sporosarcina thermotolerans sp. nov., two endospore-forming species. Int J Syst Evol Microbiol 60:1353–1357. https://doi.org/10.1099/ijs.0.014423-0
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Khurana H, Sharma M, Verma H, Lopes BS, Lal R, Negi RK (2020) Genomic insights into the phylogeny of Bacillus strains and elucidation of their secondary metabolic potential. Genomics 112:3191–3200. https://doi.org/10.1016/j.ygeno.2020.06.005
Khurana H, Sharma M, Bharti M, Singh DN, Negi RK (2021) Gut milieu shapes the bacterial communities of invasive silver carp. Genomics 113:815–826. https://doi.org/10.1016/j.ygeno.2021.01.013
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721. https://doi.org/10.1099/ijs.0.038075-0
Kluyver AJ, van Neil CB (1936) Prospects for a natural classification of bacteria. Zentbl Bakteriol Parasitenkd Infektkrankh Hyg Abt II 94:369–403
Komagata K, Suzuki K (1987) Lipids and cell-wall analysis in bacterial systematics. Method Microbiol 19:161–207. https://doi.org/10.1016/S0580-9517(08)70410-0
Krossøy C, Waagbø R, Ørnsrud R (2011) Vitamin K in fish nutrition. Aquac Nutr 17:585–594. https://doi.org/10.1111/j.1365-2095.2011.00904.x
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Kwon SW, Kim BY, Song J, Weon HY, Schumann P, Tindall BJ, Stackebrandt E, Fritze D (2007) Sporosarcina koreensis sp. nov. and Sporosarcina soli sp. nov, isolated from soil in Korea. Int J Syst Evol Microbiol 57:1694–1698. https://doi.org/10.1099/ijs.0.64352-0
Mahizan NA, Yang SK, Moo CL, Song AA, Chong CM, Chong CW, Abushelaibi A, Lim SH, Lai KS (2019) Terpene derivatives as a potential agent against antimicrobial resistance (AMR) pathogens. Molecules 24:2631. https://doi.org/10.3390/molecules24142631
Mao B, Yin R, Li X, Cui S, Zhang H, Zhao J, Chen W (2021) Comparative genomic analysis of Lactiplantibacillus plantarum isolated from different niches. Genes 12:241. https://doi.org/10.3390/genes12020241
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:60. https://doi.org/10.1186/1471-2105-14-60
Minnikin DE, O’Donnell AG, Goodfellow M, Alderson G, Athalye M, Schaal A, Parlett JH (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Method 2:233–241. https://doi.org/10.1016/0167-7012(84)90018-6
Montaner B, Pérez-Tomás R (2001) Prodigiosin-induced apoptosis in human colon cancer cells. Life Sci 68:2025–2036. https://doi.org/10.1016/S0024-3205(01)01002-5
Nakamura LK (1984) Bacillus psychrophilus sp. nov., nom. rev. Int J Syst Bacteriol 34:121–123. https://doi.org/10.1099/00207713-34-2-121
Navarrete P, Fuentes P, De la Fuente L, Barros L, Magne F, Opazo R, Ibacache C, Romero J (2013) Short-term effects of dietary soybean meal and lactic acid bacteria on the intestinal morphology and microbiota of Atlantic salmon (Salmo salar). Aquac Nutr 19:827–836. https://doi.org/10.1111/anu.12047
Nelson JS, Grande TC, Wilson MV (2016) Fishes of the world. Wiley, New York
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Nies DH, Nies A, Chu L, Silver S (1989) Expression and nucleotide sequence of a plasmid-determined divalent cation efflux system from Alcaligenes eutrophus. Proc Natl Acad Sci 86:7351–7355. https://doi.org/10.1073/pnas.86.19.7351
Oktari A, Supriatin Y, Kamal M, Syafrullah H (2017) The bacterial endospore stain on schaeffer fulton using variation of methylene blue solution. J Phys Conf Ser 812:012066. https://doi.org/10.1088/1742-6596/812/1/012066
Page AJ, Cummins CA, Hunt M, Wong VK, Reuter S et al (2015) Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics 31:3691–3693. https://doi.org/10.1093/bioinformatics/btv421
Pal A, Dutta S, Mukherjee P, Paul A (2005) Occurrence of heavy metal-resistance in microflora from serpentine soil of andaman. J Basic Microbiol 45:207–218. https://doi.org/10.1002/jobm.200410499
Pantos O, Cooney RP, Le Tissier MD, Barer MR, O’Donnell AG, Bythell JC (2003) The bacterial ecology of a plague-like disease affecting the caribbean coral Montastrea annularis. Environ Microbiol 5:370–382. https://doi.org/10.1046/j.1462-2920.2003.00427
Plessl C, Otachi EO, Körner W, Avenant-Oldewage A, Jirsa F (2017) Fish as bioindicators for trace element pollution from two contrasting lakes in the eastern rift Valley, Kenya: spatial and temporal aspects. Environ Sci Pollut Res 24:19767–19776. https://doi.org/10.1007/s11356-017-9518-z
Price-Whelan A, Dietrich LE, Newman DK (2006) Rethinking’secondary’metabolism: physiological roles for phenazine antibiotics. Nat Chem Biol 2:71–78. https://doi.org/10.1038/nchembio764
Priyodip P, Balaji S (2019) A preliminary study on probiotic characteristics of Sporosarcina spp. for poultry applications. Curr Microbiol 76:448–461. https://doi.org/10.1007/s00284-019-01647-2
Rabus R (2005) Functional genomics of an anaerobic aromatic-degrading denitrifying bacterium, strain EbN1. Appl Microbiol Biotechnol 68:580–587. https://doi.org/10.1007/s00253-005-0030-x
Ran ZH, Bi WA, Cai QT, Li XX, Min LI, Dong HU, Guo DB, Juan WA, Chun FA (2016) Bioremediation of hexavalent chromium pollution by Sporosarcina saromensis M52 isolated from offshore sediments in Xiamen, China. Biomed Environ Sci 29:127–136. https://doi.org/10.3967/bes2016.014
Reddy GS, Matsumoto N, Shivaji S (2003) Sporosarcina macmurdoensis sp. nov., from a cyanobacterial mat sample from a pond in the McMurdo dry valleys. Antarct Int J Syst Evol Microbiol 53:1363–1367. https://doi.org/10.1099/ijs.0.02628-0
Rix L, Ribes M, Coma R, Jahn MT, de Goeij JM, van Oevelen D, Eschrig S, Meibom A, Hentschel U (2020) Heterotrophy in the earliest gut: a single-cell view of heterotrophic carbon and nitrogen assimilation in sponge-microbe symbioses. ISME J 14:2554–2567. https://doi.org/10.1038/s41396-020-0706-3
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sambrook J, Russell DW (2000) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Serra CR, Almeida EM, Guerreiro I, Santos R, Merrifield DL, Tavares F, Oliva-teles EP (2019) Selection of carbohydrate-active probiotics from the gut of carnivorous fish fed plant-based diets. Sci Rep 9:1–15. https://doi.org/10.1038/s41598-019-42716-7
Silver S, Phung LT (2005) Genes and enzymes involved in bacterial oxidation and reduction of inorganic arsenic. Appl Environ Microbiol 71:599–608. https://doi.org/10.1128/AEM.71.2.599-608.2005
Singh DP, Prabha R, Gupta VK, Verma MK (2018) Metatranscriptome analysis deciphers multifunctional genes and enzymes linked with the degradation of aromatic compounds and pesticides in the wheat rhizosphere. Front Microbiol 9:1331. https://doi.org/10.3389/fmicb.2018.01331
Sun Y, Zhao Q, Zhi D, Wang Z, Wang Y, Xie Q, Wu Z, Wang X, Li Y, Yu L, Yang H, Zhou J, Li H (2017) Sporosarcina terrae sp. nov., isolated from orchard soil. Int J Syst Evol Microbiol 67:2104–2108. https://doi.org/10.1099/ijsem.0.001835
Talwar C, Nagar S, Lal R, Negi RK (2018) Fish gut microbiome: current approaches and future perspectives. Indian J Microbiol 58:397–414. https://doi.org/10.1007/s12088-018-0760-y
Tamaoka J (1986) Analysis of bacterial menaquinone mixtures by reverse-phase high-performance liquid chromatography. Method Enzymol 123:31–36. https://doi.org/10.1016/S0076-6879(86)23028-1
Tarnecki AM, Brennan NP, Schloesser RW et al (2019) Shifts in the skin-associated microbiota of hatchery-reared common snook Centropomus undecimalis during acclimation to the wild. Microb Ecol 77:770–781. https://doi.org/10.1007/s00248-018-1252-7
Tominaga T, An SY, Oyaizu H, Yokota A (2009) Sporosarcina luteola sp. nov. isolated from soy sauce production equipment in Japan. J Gen Appl Microbiol 55:217–223. https://doi.org/10.2323/jgam.55.217
Vandamme P, Pot B, Gillis M, De Vos P, Kersters K, Swings J (1996) Polyphasic taxonomy, a consensus approach to bacterial systematics. Microbiol Rev 60:407–438. https://doi.org/10.1128/mr.60.2.407-438.1996
Wackett LP (2017) Microbial β-lactone natural products. Microb Biotechnol 10:218–220
Wolfgang WJ, Coorevits A, Cole JA, De Vos P, Dickinson MC, Hannett GE, Jose R, Nazarian E, Schumann P, Landschoot AV, Wirth SV, Musser KA (2012) Sporosarcina newyorkensis sp. nov. from clinical specimens and raw cow’s milk. Int J Syst Evol Microbiol 62:322–329. https://doi.org/10.1099/ijs.0.030080-0
Xu L, Dong Z, Fang L, Luo Y, Wei Z, Guo H, Zhang G, Gu YQ, Coleman-Derr D, Xia Q, Wang Y (2019) OrthoVenn2: a web server for whole-genome comparison and annotation of orthologous clusters across multiple species. Nucleic Acid Res 47:W52–W58. https://doi.org/10.1093/nar/gkz333
Yaakop AS, Chan KG, Ee R, Lim YL, Lee SK, Manan FA, Goh KM (2016) Characterization of the mechanism of prolonged adaptation to osmotic stress of Jeotgalibacillus malaysiensis via genome and transcriptome sequencing analyses. Sci Rep 6:33660. https://doi.org/10.1038/srep33660
Ye Y, Doak TG (2009) A parsimony approach to biological pathway reconstruction/inference for genomes and metagenomes. PLoS Comput Biol 5(8):e1000465
Yoon JH, Lee KC, Weiss N, Kho YH, Kang KH, Park YH (2001) Sporosarcina aquimarina sp. nov., a bacterium isolated from seawater in Korea, and transfer of Bacillus globisporus (Larkin and Stokes 1967), Bacillus psychrophilus (Nakamura 1984) and Bacillus pasteurii (Chester 1898) to the genus Sporosarcina as Sporosarcina globispora comb. nov., Sporosarcina psychrophila comb. nov. and Sporosarcina pasteurii comb. nov., and emended description of the genus Sporosarcina. Int J Syst Evol Microbiol 51:1079–1086. https://doi.org/10.1099/00207713-51-3-1079
Yu Y, Xin YH, Liu HC, Chen B, Sheng J, Chi ZM, Zhou PJ, Zhang DC (2008) Sporosarcina antarctica sp. nov., a psychrophilic bacterium isolated from the Antarctic. Int J Syst Evol Microbiol 58:2114–2117. https://doi.org/10.1099/ijs.0.65838-0
Yukgehnaish K, Kumar P, Sivachandran P, Marimuthu K, Arshad A, Paray BA, Arockiaraj J (2020) Gut microbiota metagenomics in aquaculture: factors influencing gut microbiome and its physiological role in fish. Rev Aquac 12:1903–1927. https://doi.org/10.1111/raq.12416
Zhang G, Ren H, Chen X, Zhang Y, Yang Y, Wang S, Jiang Y (2014) Sporosarcina siberiensis sp. nov., isolated from the east Siberian Sea. Antonie Van Leeuwenhoek 106:489–495. https://doi.org/10.1007/s10482-014-0217-1
Zhang H, Yohe T, Huang L, Entwistle S, Wu P, Yang Z, Busk PK, Xu Y, Yin Y (2018) dbCAN2: a meta server for automated carbohydrate-active enzyme annotation. Nucleic Acid Res 46:95–101. https://doi.org/10.1093/nar/gky418
Funding
This work was supported by grants received from the Indian Council of Agricultural Research—National Bureau of Agriculturally Important Microorganisms funded project (ICAR-NBAIM) [grant number NBAIM/AMAAS/2017–20/GF/1a/512], and Faculty Research Program (FRP)-Institute of Eminence (IoE), University of Delhi. MB, MS, SC, SN acknowledge Council of Scientific and Industrial research (CSIR) and SY thanks UGC for providing the research fellowship.
Author information
Authors and Affiliations
Contributions
R.K.N. conceived the idea. M.B. isolated the strain. M.B., H.K. and M.S. performed the biochemical and physiological tests. M.S. designed the experiments. M.S., S.Y., S.M. performed phylogenetic and genome analysis. S.C. performed the chemotaxonomic analysis under the guidance of SK. M.B. wrote the manuscript and M.S. edited the manuscript. H.K., M.S., S.N., S.C., S.K. and R.K.N. critically reviewed the manuscript.
Corresponding author
Ethics declarations
Competing of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bharti, M., Khurana, H., Sharma, M. et al. Taxonomic and genomic characterization of Sporosarcina cyprini sp. nov., moderately tolerant of Cr+6 and Cd+2 isolated from the gut of invasive fish Cyprinus carpio var. communis (Linn., 1758). Antonie van Leeuwenhoek 116, 193–206 (2023). https://doi.org/10.1007/s10482-022-01794-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-022-01794-w