Abstract
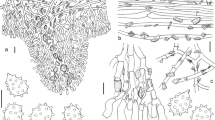
Seven new Entomocorticium species (Peniophoraceae) are described based on morphology and phylogenetic analyses. Along with the type species (E. dendroctoni), Entomocorticium comprises eight species of nutritional symbionts of pine bark beetles in North America. Entomocorticium cobbii is the mycangial associate of the southern pine beetle, Dendroctonus frontalis, and E. parmeteri is the mycangial associate of the western pine beetle, D. brevicomis. Entomocorticium whitneyi, E. portiae, E. kirisitsii, E. oberwinkleri and the previously described E. dendroctoni have been isolated from galleries of D. ponderosae and D. jeffreyi in western North America. Entomocorticium sullivanii forms an ambrosia-like layer of basidia and basidiospores in the pupal chambers of Ips avulsus in the southeastern USA. Entomocorticium is phylogenetically placed within Peniophora, a corticioid genus of wood decay fungi with wind-dispersed basidiospores. At least four species of Entomocorticium produce basidiospores on basidia with reduced sterigmata that apparently do not forcibly discharge basidiospores. Another basidiomycete, Gloeocystidium ipidophilum, was described from Ips typographus galleries in Europe, but it is phylogenetically and taxonomically placed in another genus of wood decay fungi as Cylindrobasidium ipidophilum (Physalacriaceae). The free-living wood-decay fungus Phlebiopsis gigantea (Phanerochaetaceae) has been occasionally associated with bark beetles but is unrelated to C. ipidophilum or Entomocorticium.






Similar content being viewed by others
References
Addison A, Powell JA, Six DL, Bentz BJ (2015) Integrating models to investigate critical phenological overlaps in complex ecological interactions: the mountain pine beetle-fungus symbiosis. J Theoretical Biol 368:55–66
Barras SJ, Perry TJ (1972) Fungal symbionts in the pro-thoracic mycangium of Dendroctonus frontalis. Z Angewandte Entomologie 71:95–104
Bracewell RR, Six DL (2014) Broadscale specificity in a bark beetle-fungal symbiosis: a spatio-temporal analysis of the mycangial fungi of the western pine beetle. Microb Ecol 68:859–870. https://doi.org/10.1007/s00248-014-0449-7
Bracewell RR, Six DL (2015) Experimental evidence of bark beetle adaptation to a fungal symbiont. Ecol Evol 5:5109–5119. https://doi.org/10.1002/ece3.1772
Bracewell RR, Vanderpool D, Good JM, Six DL (2018) Cascading speciation among mutualists and antagonists in a tree–beetle–fungi interaction. Royal Society Proc B 285:20180694. https://doi.org/10.1098/rspb.2018.0694
Bridges RL (1985) Relationship of symbiotic fungi to southern pine beetle population trends. In: Branham SJ and Thatcher RC (eds) Integrated Pest Management Research Symposium: the proceedings: USDA Forest Service, General Technical Report SO-56. USDA Forest Service, Asheville, NC. pp 127–135
Coppedge BR, Stephen FM, Felton GW (1995) Variation in female southern pine beetle size and lipid content in relation to fungal associates. Canad Entomol 127:145–154
Eriksson J, Ryvarden L (1975) The Corticiaceae of North Europe, vol 3. Corticium to Hyphoderma. Fungiflora, Norway, pp 446–545
Eriksson J, Hjortstam K, Ryvarden L (1978) The Corticiaceae of North Europe, vol 5. Mycoaciella to Phanerochaete. Fungiflora, Norway, pp 887–1047
Floudas D, Held BW, Riley R, Nagy LG, Koehler G, Ransdell AS, Younus H, Chow J, Chiniquy J, Lipzen A, Tritt A, Sun H, Haridas S, LaButti K, Ohm RA, Kües U, Blanchette RA, Grigoriev IV, Minto RE, Hibbett DS (2015) Evolution of novel wood decay mechanisms in Agaricales revealed by the genome sequences of Fistulina hepatica and Cylindrobasidium torrendii. Fungal Genet Biol 76:78–92. https://doi.org/10.1016/j.fgb.2015.02.002
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Happ GM, Happ CM, Barras SJ (1976a) Bark beetle-fungal symbiosis. II. Fine structure of a basidiomycetous ectosymbiont of the southern pine beetle. Can J Bot 54:1049–1062
Happ GM, Happ CM, French JRJ (1976b) Ultrastructure of the mesonotal mycangium of an ambrosia beetle, Xyleborus dispar (F.) (Coleoptera: Scolytidae). Intern J Insect Morphol Embryol 5:381–391
Hallenberg N, Larsson E, Mahlapuu M (1996) Phylogenetic studies in Peniophora. Mycol Res 100:179–187
Harrington TC (1980) Release of basidiospores from the pouch fungus, Cryptoporus volvatus. Mycologia 72:929–936
Harrington TC (2005) Ecology and evolution of mycophagous bark beetles and their fungal partners. In: Vega FE, Blackwell M (eds) Ecological and evolutionary advances in insect-fungal associations. Oxford University Press, Oxford, pp 257–291
Harrington TC, Aghayeva DN, Fraedrich SW (2010) New combinations in Raffaelea, Ambrosiella, and Hyalorhinocladiella, and four new species from the redbay ambrosia beetle, Xyleborus glabratus. Mycotaxon 111:337–361. https://doi.org/10.5248/111.337
Harrington TC, Furniss MM, Shaw CG (1981) Dissemination of hymenomycetes by Dendroctonus pseudotsugae (Coleoptera: Scolytidae). Phytopathology 71:551–554
Harrington TC, Zambino PJ (1990) Ceratocystiopsis ranaculosus, not Ceratocystis minor var. barrasii, is the mycangial fungus of the southern pine beetle. Mycotaxon 38:103–115
Hsiau PTW, Harrington TC (1997) Ceratocystiopsis brevicomi sp. nov., a mycangial fungus from Dendroctonus brevicomis (Coleoptera: Scolytidae). Mycologia 89:661–669. https://doi.org/10.2307/3761004
Hsiau PTW, Harrington TC (2003) Phylogenetics and adaptations of basidiomycetous fungi fed upon by bark beetles (Coleoptera: Scolytidae). Symbiosis 34:111–131
Jülich W (1984) Die Nichtblattterpilze, Gallerpilze und Bauchpilze: (Aphyllophorales, Heterobasidiomycetes, Gastromycetes. Gustav Fischer Verlag, Stuttgart – New York, pp 626
Jusino MA, Skelton J, Chen CC, Hulcr J, Smith ME (2020) Sexual reproduction and saprotrophic dominance by the ambrosial fungus Flavodon subulatus (= Flavodon ambrosius). Fungal Ecol. https://doi.org/10.1016/j.funeco.2020.100979
Kirisits T (2005) Gloeocystidium ipidophilum: Ein ungewöhnlicher Basidiomycete, der mit dem Buchdrucker assoziiert ist. Forstschutz Aktuell 34:14–17
Kirisits T (2010) Fungi isolated from Picea abies infested by the bark beetle Ips typographus in the Białowieża forest in north-eastern Poland. For Pathol 40:100–110. https://doi.org/10.1111/j.1439-0329.2009.00613.x
Larsson KH (2007) Re-thinking the classification of corticioid fungi. Mycol Res 111:1040–1063. https://doi.org/10.1016/j.mycres.2007.08.001
Leal-Dutra CA, Neves MA, Griffith GW, Reck MA, Clasen LA, Dentinger BTM (2018) Reclassification of Parapterulicium Corner (Pterulaceae, Agaricales), contributions to Lachnocladiaceae and Peniophoraceae (Russulales) and introduction of Baltazaria gen. nov. MyoKeys 37:39–56
Lee S, Kim JJ, Breuil C (2005) Diversity of fungi associated with the mountain pine beetle, Dendroctonus ponderosae, and infested lodgepole pines in British Columbia. Fungal Divers 22:91–105
Mayers CG, McNew DL, Harrington TC, Roeper RA, Fraedrich SW, Biedermann PHW, Castrillo LA, Reed SE (2015) Three genera in the Ceratocystidaceae are the respective symbionts of three independent lineages of ambrosia beetles with large, complex mycangia. Fungal Biol 119:1075–1092
Rayner RW (1970) A mycological colour chart. British Mycological Society Commonwealth Mycological Institute, Kew, Surrey
Rehner S, Samuels GJ (1994) Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol Res 98:625–634. https://doi.org/10.1016/S0953-7562(09)80409-7
Reijnders AFM (2000) A morphological analysis of the basic characters of the Gasteromycetes and their relation to other basidiomycetes. Mycol Res 104:900–910
Roe AD, Rice AV, Coltman DW, Cooke JEK, Sperling FAH (2011) Comparative phylogeography, genetic differentiation and contrasting reproductive modes in three fungal symbionts of a multipartite bark beetle symbiosis. Mol Ecol 20:584–600. https://doi.org/10.1111/j.1365-294X.2010.04953.x
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574. https://doi.org/10.1093/bioinformatics/btg180
Siemaszko W (1939) Zespoly grzybow towarzyszacych kornikom polskim. Planta Polonica 7:1–52
Six Dl (2019) A major symbiont shift supports a major niche shift in a clade of tree-killing bark beetles. Ecolog Entomol. https://doi.org/10.1111/een.12786
Six DL, Paine TD (1998) The effects of mycangial fungi on development and emergence of Dendroctonus ponderosae and D. jeffreyi. Environ Entomol 27:1393–1401
Slysh AR (1960) The genus Peniophora in New York State and adjacent regions. Technical Publication No. 83 of the State University College of Forestry at Syracuse University, p 95
Solheim H (1992) Fungal succession in sapwood of Norway spruce infested with Ips typographus. Eur J For Pathol 22:136–148
Stalpers JA (1978) Identification of wood-inhabiting Aphyllophorales in pure culture. Stud Mycol 16:248
Swofford DL (1992) PAUP: phylogenetic analysis using parsimony, version 3.1.1 edition. Illinois Natural History Survey, Champaign.
Tsuneda AS, Murakani S, Sigler L, Hiratsuka Y (1993) Schizolysis of dolipore-parenthesome septa in an arthroconidial fungus associated with Dendroctonus ponderosae and in similar anamorphic fungi. Can J Bot 71:1032–1038
Vanderpool D, Bracewell RR, McCutcheon JP (2017) Know your farmer: ancient origins and multiple independent domestications of ambrosia beetle fungal cultivars. Mol Ecol 27:2077–2094. https://doi.org/10.1111/mec.14394
Vainio EJ, Lipponen K, Hantula J (2008) Persistence of a biocontrol strain of Phlebiopsis gigantea in conifer stumps and its effects on within-species genetic diversity. For Pathol 31:285–295. https://doi.org/10.1046/j.1439-0329.2001.00249.x
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and application. Academic Press, San Diego, pp 315–322
Whitney HS, Cobb FW (1972) Non-staining fungi associated with the bark beetle Dendroctonus brevicomis (Coleoptera: Scolytidae) on Pinus ponderosa. Can J Bot 50:1943–1945
Whitney HS, Bandoni RJ, Oberwinkler F (1987) Entomocorticium dendroctoni gen. et sp. nov. (Basidiomycotina), a possible nutritional symbiote of the mountain pine beetle in lodgepole pine in British Columbia. Can J Bot 65:95–102. https://doi.org/10.1139/b87-013
Woodbridge B, Coley-Smith JR, Reid DA (1988) A new species of Cylindrobasidium parasitic on sclerotia of Typhula incarnata. Trans Brit Mycol Soc 91:166–169
Acknowledgements
Portia T. W. Hsiau, Thomas Kirisits, Martin MacKenzie, Karen Nakasone, Diana Six, Halvor Solheim, and Brian Sullivan kindly provided material used in this study. Stephen Fraedrich (US Forest Service, Athens, Georgia) and Rabiu Olatinwo (US Forest Service, Pineville, Louisiana) provided material for study and facilitated funding through a cooperative agreement with the US Forest Service. The authors especially thank K. H. Larsson for his helpful comments on the manuscript.
Funding
The work upon which this publication is based was funded in part through Joint Venture Agreement 16-JV-11330129-009 with the US Forest Service, Southern Research Station.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Harrington, T.C., Batzer, J.C. & McNew, D.L. Corticioid basidiomycetes associated with bark beetles, including seven new Entomocorticium species from North America and Cylindrobasidium ipidophilum, comb. nov. Antonie van Leeuwenhoek 114, 561–579 (2021). https://doi.org/10.1007/s10482-021-01541-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-021-01541-7