Abstract
The main objective of this work was to purify the exopolysaccharides (EPS) of Flammulina velutipes SF-06 and investigate the relationship between the different purified fractions and bioactive activity. Two fractions (EPS-1 and EPS-2) were separated and purified by DEAE-52 cellulose and Sephadex G-100 cellulose column chromatography. Monosaccharides composition analysis by gas chromatography indicated that EPS, EPS-1 and EPS-2 were heteropolysaccharides in which rhamnose was a major component. Fourier transform infrared (FT-IR) analysis detected furanose-ring in EPS-1 and EPS-2. All fractions possessed considerable antioxidant activity, while EPS-2 has stronger antioxidant activity than EPS and EPS-1 in vitro. The EPS also exhibited potent anti-aging activation in mice, such as increased catalase and total antioxidant capacity, and decreasing the malondialdehyde (MDA) content. Both the antioxidant in vitro and anti-aging in vivo potentials of EPS could be further utilized in the food industry.
Similar content being viewed by others
Introduction
Previous studies indicated that free radicals cause extensive damage to nucleic acid, protein and other biomolecules (Ames et al. 1993). Free radicals, especially reactive oxygen radicals, have physiological effects related to the pathogenesis of several human diseases including heart diseases, gastric problems, aging and certain types of cancer as well as the prevention of the food products from oxidative deterioration (Halliwell 1996). The antioxidant compounds, dietary free radical scavengers in the prevention of oxidative damage in living organism, play a vital role in curing and preventing undesirable physiological effects (Kohen and Nyska 2002). Therefore, new interest has been developed to employ antioxidants from natural sources by consumer preference. Polysaccharides extracted from edible mushrooms are one of the most important elements correlated with multiple bioactivities, such as antioxidant, reducing blood lipid, immunomodulatory and antitumor activities (Liu et al. 2014; Yang et al. 2012). Today an increasing number of consumers are interested in natural polysaccharides. Exopolysaccharides (EPS), a class of high-value biopolymers extracted from fermentation broth, have potential in many industrial applications. Furthermore, EPS is superior to polysaccharides from liquid-cultured mycelia and fruit body due to its higher productions with short time consuming, and less chance for contamination (Chen et al. 2008).
Flammulina velutipes, a commercially important edible mushroom, has been under a large-scale artificial cultivation and increasingly consumed in Asia because of its high nutritional value and delectable flavor (Fan et al. 2001; Smiderle et al. 2008; Yang et al. 2011). Several components isolated from F. velutipes, including a variety of polysaccharides and glycoproteins, have been reported to be bioactive (Lin et al. 2013). For example, bioactive polysaccharides isolated from F. velutipes fruiting body were noted to have immunomodulatory, anti-proliferation, anti-inflammatory and antitumor activities (Leung et al. 1997; Wu et al. 2010; Yang et al. 2012). Previous studies have shown that the bioactivities of polysaccharides can be affected by many factors including structure, molecular weight, monosaccharide components, even the extraction and isolation methods (Fan et al. 2012, 2014). However, there is still a lack of knowledge concerning EPS from submerged culture of F. velutipes. In particular, few reports have been documented concerning the purification, characterization and biological properties of EPS by F. velutipes.
In our work, EPS from fermentation broth of F. velutipes SF-06 were isolated and purified by DEAE-52 cellulose and then by Sephadex G-100 cellulose column chromatography. Preliminary structural characterization of the EPS and its fractions was determined by Fourier transform infrared (FT-IR) analysis and gas chromatography (GC). The antioxidant activities in vitro of the EPS and its fractions were evaluated and the anti-aging of the EPS was analyzed. In addition, the relationship between the different purified fractions and the antioxidant activity was investigated.
Materials and methods
Chemicals
l-Alanine and 1,1-diphenyl-2-picrylhydrazyl (DPPH) were from Sigma Chemicals Company (St. Louis, USA). T-AOC box was from Nanjing Jiancheng Bioengineering Institute. All the other chemicals were of analytical grade and purchased from local chemical suppliers in China.
Microorganism and culture conditions
Flammulina velutipes SF-06 was maintained on potato dextrose agar (PDA) plates at 4 °C by our laboratory and used in this experiment. Seeding cultivation in liquid media were cultured in a 1 L filter flask containing 500 mL of (g/L) potato (cut into piece) 200, glucose 10, KH2PO4 1.5 and MgSO4·7H2O 1 at 25 °C on a rotary shaker (160 rpm, Anting, Shanghai, China) for 7 days. Batch fermentation conditions of F. velutipes SF-06, including temperature, pH and air supply, were controlled automatically by a fermenter (100-L, Xianmin, China) for 7 days and the culture medium used the potato dextrose broth above.
Preparation and measurement of EPS
The fermentation broth of F. velutipes SF-06 was centrifuged at 3,000×g for 15 min, the supernatant liquid was mixed with 3 volumes of 95 % ethanol (v/v), stirred vigorously, and kept at 4 °C for 24 h. After centrifugation (3,000×g, 15 min), the precipitated EPS was dissolved in distilled water (60 °C), and the EPS content was determined at 490 nm by the phenol–sulfuric acid method (Dubois et al. 1956). After quick prefreezing (−70 °C, 24 h), the EPS solution was lyophilized by vacuum freeze drying (Labconco, USA).
Purification of EPS
The EPS was purified by DEAE-52 cellulose column chromatography according to Qiao et al. (2009) and Ye et al. (2008) with a sight modification. EPS (1.0 g) was dissolved in 15 mL of deionized water, and the sample was filtered through 0.22 µm membrane filter. Polysaccharides were fractionated by a DEAE-52 cellulose column (26 mm × 400 mm) and the column was stepwise eluted with distilled water and different concentrations of NaCl solutions (0.2, 0.5, and 1.0 mol/L NaCl) at a flow rate of 1.0 mL/min. The eluate was collected automatically (2 mL/tube) through fraction collector (BSZ-100, Huxi, Shanghai, China) and the carbohydrate content in each tube was analyzed spectrophotometrically using a phenol–sulfuric acid method (Dubois et al. 1956).
After dialyzing and concentrating, the eluted fractions were further loaded on a Sephadex G-100 column (26 mm × 600 mm), and eluted with deionized water at a flow rate of 0.1 mL/min. After dialysis of distilled water for 10 h, the obtained eluates were collected, concentrated and lyophilized for antioxidant experiments in vitro and FT-IR spectra analysis.
Ultraviolet (UV) and FT-IR spectra analysis
UV and FT-IR spectra were implemented as the descriptions (Zhao et al. 2010). The ultraviolet spectrum of the polysaccharide samples with a final concentration of 0.1 % was recorded with an ultraviolet spectrophotometer (Shanghai, China) at the range of 200–400 nm, and the FT-IR spectrum was scanned with a Nicolet FT-IR spectrometer (NICOLET NEXUS470, Thermo Nicolet Co., WI, USA). The polysaccharide (dried by freezing dried) was ground with potassium bromide (KBr) powder and pressed into pellet for FT-IR spectral measurement at the wavenumber range of 4,000–400 cm−1 (Kumar et al. 2004).
Monosaccharides composition
GC was used to determine the monosaccharides composition of the polysaccharide samples as described by Yang et al. (2012) with slight modifications. Monosaccharides composition was confirmed using the retention time of chromatographic peak. The percentage of different monosaccharide was determined according to the area normalization method. GC was performed using a Shimadzu GC-2010 equipped with a capillary column of Rtx-1 (30 m × 0.25 mm × 0.25 µm) by the method of our previous work (Zhang et al. 2014). Compared with standard spectra of known concentration, the formula was expressed as following according to the Eq. 1:
where A 1 and A 2 is peak area of are sample and standard peak areas, respectively; V is the sample volume constant volume (mL); M is weight the sample quality (g); C is the sample sugars concentration of mixed standard (mg/mL).
Antioxidant capacity in vitro
Reducing power assay
The reducing power of the polysaccharide samples was estimated according to Oyaizu (1986) with a slight modification. The reaction mixtures, mixing with the polysaccharide samples (1.0 mL, 100–1,000 mg/L), phosphate buffer (2.5 mL, pH 6.6, 0.2 mol/L) and potassium ferricyanide (1.0 mL, 1 %, w/v), were incubated at 50 °C for 20 min. The reaction was terminated via adding trichloroacetic acid (2.0 mL, 10 %, w/v). After centrifugation (1,200×g, 10 min), the supernatant was collected and incubated with ferric trichloride (1.2 mL, 0.1 %, w/v) for 15 min at 25 °C. The absorbance of the resulting solution was recorded at 700 nm.
Hydroxyl radical assay
Hydroxyl radical activity was determined as the descriptions of Smironff and Cumbes (1989) with slight modifications. The reaction mixture, including ferrous sulfate (1 mL, 9 mmol/L), salicylic acid (1 mL, 9 mmol/L), the polysaccharide samples (1 mL, 100–1,000 mg/L) and hydrogen peroxide (1 mL, 0.03 %, v/v), was incubated at 37 °C for 30 min. The absorbance of the solution was measured at 510 nm. IC50 values were calculated by probit regression and expressed as mg/L, indicating the concentration of the tested samples necessary to scavenge 50 % of the free radicals in the reaction mixture. The scavenging rate of hydroxyl radical was calculated as follows by the Eq. 2:
where A was the absorbance of the polysaccharide samples and A 0 was the absorbance of the blank (water instead of sample).
Superoxide radical assay
The superoxide radical activity of the polysaccharide samples was measured by the method of Stewar and Beewley (1980) with a slight modification. The reaction mixture, containing phosphate buffer saline (0.5 mL, 0.2 mol/L, pH 7.8), riboflavin (0.3 mL, 10 mmol/L), methionine (0.25 mL, 13 mmol/L), and the polysaccharide samples (1.0 mL, 100–1,000 mg/L), was illuminated with a fluorescent lamp at 25 °C for 30 min. The scavenging rate of superoxide anion generation was calculated with the following Eq. 3:
where A is the absorbance of the polysaccharide samples and A 0 is the absorbance of the blank (water instead of sample).
DPPH scavenging assay
The DPPH scavenging activity of the polysaccharide samples was measured by the previous method of Brand-Williams et al. (1995) and Kong et al. (2012) with some modifications. The reaction mixture contained ethanol (2 mL, 95 %, w/v), DPPH (0.1 μmol/L) and the polysaccharide samples (2 mL, 100–1,000 mg/L). After incubation at 25 °C for 15 min, the absorbance of the solution was determined at 517 nm. The scavenging rate of the polysaccharide samples was evaluated by the following formula Eq. 4:
where A is the absorbance of the tested sample and A 0 is the absorbance of the blank (water instead of sample).
Anti-aging of EPS in vivo
Animal experiments
Fifty male mice (Kunming strain) weighing 20 ± 2 g, providing by Center for Animal Testing of Shandong Lukang Drugs Group Limited (Jining, China), were housed in conventional cages with free access to water and standard food at 22 ± 1 °C with a 12 h light–dark cycle. Experiments were in accordance with the institutional ethical guidelines and under guidance of Shandong Agricultural University Committee. The aging group was established by intraperitoneal injection with d-galactose. After 3 days acclimatization period, animals were divided into five groups consisting of ten mice each. Mice in Groups I (low dose of EPS), II (middle dose of EPS) and III (high dose of EPS) were received doses of 200, 400 and 800 mg/kg body weight of mice by filling the stomach (0.01 mL/g body weight) with a syringe and d-galactose intraperitoneal injection (1,000 mg/kg) every day, respectively. Mice in Group IV (negative control) were treated with the same volume of saline instead of EPS. Mice in Group V (model control) were treated with the same volume of saline instead of EPS and d-galactose. After 20 days of treatment, mice were anaesthetized and blood was collected from the retrobulbar vein with a vacutainer and anticoagulated by heparin (stored at 4 °C). Heart, liver and kidney of mice were rapidly separated, weighed, and homogenized (1:9, w/v) immediately in phosphate buffer (0.2 mol/L, 4 °C, pH 7.4), respectively. The homogenates were centrifuged (6,000×g) at 4 °C for 20 min and the supernatants were reserved at −20 °C.
Measurement of biochemical parameters in vivo
The assay of Schlorff et al. (1999) with slight modifications was used to measure catalase (CAT). The reaction mixture, containing sodium–potassium phosphate (2 mL), hydrogen peroxide (65 μmol/L) and blood or tissue homogenate (0.5 mL), was reacted at 37 °C for 1 min. Ammonium molybdate (2 mL, 32.4 mmol/L) was added for termination reaction at 25 °C. The activity of CAT was expressed as μmol/L of H2O2 degraded per minute per milligram of tissue or per minute per milliliter of blood.
The content of malondialdehyde (MDA) was measured by 2-thiobarbituric acid (TBA) method with a slight modification and expressed as nanomole per milligram of protein (nmol/mg protein) in heart, liver and kidney or nanomole per milliliter (nmol/mL serums) in blood (Placer et al. 1966). The reaction mixture, mixing with blood samples or tissue homogenate (0.5 mL), distilled water (1.5 mL) and thiobarbituric acid (2 mL, 0.6 %, w/v), was heated in a boiling water for 15 min. After cooling rapidly, the mixture was centrifuged at 3,000 × g for 10 min, and the supernatant was used for the determination of MDA level.
The total antioxidant capacity (T-AOC) was measured according to the method of Benzie and Stain (1996) by T-AOC kit.
Statistical analysis
All the data were carried out in triplicates and recorded as mean ± standard deviation (S. D.).The results obtained were analyzed by one-way analysis of variance (ANOVA). A P value < 0.05 was considered statistically significant. Data were processed and analyzed using SPSS 16.0.
Results and discussion
Isolation and purification of EPS
The elution profile of EPS by DEAE-52 cellulose column chromatography was illustrated in Fig. 1. The first peak, eluted with distilled water, was ascribed to a neutral fraction (EPS-1), and the second peak eluted with 0.2–1 mol/L NaCl solutions was the acidic polysaccharide fraction (EPS-2) (Gan et al. 2011). The two fractions were further purified by Sephadex G-100 cellulose column. The purified subfractions revealed a single and relatively symmetrical peak (Fig. 2).
UV and FT-IR spectra of EPS-1 and EPS-2
No absorption was scanned for the UV spectra of EPS-1 and EPS-2 within 200–400 nm, indicating the absence of protein (280 nm) and nucleic acid (260 nm). The FT-IR spectra of two fractions were also analyzed (Fig. 3). It was observed that all samples exhibited typical major broad stretching peak appeared around 3,600–3,200 cm−1 for the –OH group (Cai et al. 2013). The relative stronger absorption peak at 1,650.74 cm−1 for N–H bending vibration might be related to the little content of protein in EPS-1 (Gan et al. 2011). The characteristic absorption peak at about 865.69 and 880.72 cm−1 suggested the presence of furan ring in EPS-1 and EPS-2 (Chen et al. 2013). The bands around 1,618.19 cm−1 was the C=O asymmetric stretching vibration absorption peak and the peaks at 1,618.19 and 1,457.17 cm−1 indicated the presence of –NH2 and –COOH groups in EPS-2 (Cerna et al. 2003). According to the results stated above, the EPS by F. velutipes SF-06 is novel in its structure, compared to the reports on Morchella conica of EPS (Su et al. 2013) and Boletus aereus (Zheng et al. 2014).
Monosaccharides composition
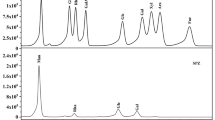
The monosaccharide composition of EPS and its subfractions were determined by GC. The monosaccharides in all samples were identified by comparing the retention times with those of standards (Fig. 4a). The content of monosaccharide is calculated by each peak area and sample sugars. As shown in Fig. 4b, EPS was mainly composed of rhamnose (Rha, 61.41 %), glucose (28.01 %, Glu) and galactose (Gal, 10.58 %) with the molar ratio of 46:23:8.7. As can be seen in Fig. 4c, the monosaccharides contents of EPS-1 were Rha (96.58 %) and Glu (3.42 %) with the molar ratio of 42.5:2.3. As shown in Fig. 4d, the monosaccharides contents of EPS-2 were Rha (94.39 %) and Gal (5.61 %) with the molar ratio of 44.5:2.9. Some reports showed that the monosaccharide composition of EPS of M. conica (Su et al. 2013), Pycnoporus sanguineus (Cao et al. 2014) and B. aereus (Zheng et al. 2014) was Man, Glu, Glu, respectively, which were differ from our results of the EPS by F. velutipes SF-06. According to the above results, the monosaccharide composition of EPS was also different from that of EPS-1 or EPS-2. In addition, the contents of Glu and Gal in EPS were relatively higher than those in EPS-1 and EPS-2. The result was different from the reported research by Pang et al. (2007) about monosaccharides composition analysis of polysaccharides isolated from F. velutipes mycelia. The possible reason may be related to the composition of different strains, culture medium and different conditions of fermentation, extraction and purification.
Evaluation of antioxidant activities in vitro
In mixture system, the mechanism for reducing properties of antioxidant substances in samples is to reduce ferricyanide complex to the ferrous form. Therefore, ferrous was determined at 700 nm by measuring the formation of Prussian blue (Xing et al. 2008). Higher absorbance value means stronger reducing power of the tested sample. The reducing power of EPS, EPS-1 and EPS-2 was 1.11 ± 0.04, 0.80 ± 0.01 and 1.45 ± 0.06 at 1,000 g/mL, respectively (Fig. 5a). The results suggested that EPS-2 had stronger reducing power in contrast to the weakest power of EPS and EPS-1. Compared with other reports, the reducing powers of EPS-2 at 1,000 g/mL was also higher than 0.5 of vitamin C (Ma et al. 2013), 0.1 of Jinqian mushroom (Liu et al. 2014), and 0.2 of Gomphidius rutilus (Gao et al. 2013), respectively.
Among the reactive oxygen species, the hydroxyl radical is the most reactive radicals and can easily cross cell membranes, readily react with most biomolecules, as well as induce tissue damage and cell death (Yuan et al. 2008). As shown in Fig. 5b, the scavenging rate of EPS-2 was 14.86 and 86.90 % higher than that of EPS and EPS-1 at a dosage of 1,000 mg/L, respectively. The scavenging rate of EPS-2 was also much higher than 35 % of Caripia montagnei (Castro et al. 2014) and 68 % of Agaricus blazei (Wu et al. 2014). The IC50 value of EPS-2 (465 mg/L) was lower than 5,230 mg/L of A. bisporus (Liu et al. 2013) and 719 mg/L of Phellinus nigricans (Wang et al. 2014a). The results showed that the EPS-2 of F. velutipes SF-06 significantly affects the scavenging of hydroxyl radical.
Superoxide anions radical was considered as a precursor of hydroxyl radicals. The superoxide anion radical was generated by auto oxidation of pyrogallol in an alkalescent condition (Wang et al. 2014b). As shown in Fig. 5c, the superoxide anions scavenging activities of EPS, EPS-1 and EPS-2 was correlated well with aggrandized concentrations. At a dosage of 1,000 mg/L, the scavenging of EPS, EPS-1 and EPS-2 were 58.32 ± 1.72, 43.62 ± 2.05, and 67.64 ± 1.89 %, respectively.
DPPH is a stable free radical that shows maximum absorbance at 517 nm in ethanol and can readily undergo scavenging by an antioxidant. DPPH free radical is widely used to evaluate the free radical scavenging activities of natural compounds (Leong and Shui 2002). As shown in Fig. 5d, the DPPH scavenging ability of EPS, EPS-1 and EPS-2 at a dosage of 1,000 mg/L were 59.55 ± 1.87, 37.38 ± 2.02, and 64.53 ± 3.02 %, respectively. The scavenging rate of the tested samples was much higher than 20 % of Boletus aereus EPS (Zheng et al. 2014). The DPPH scavenging results revealed that the EPS-2 probably contained substances that are proton donors and can react with free radicals to convert them to stable diamagnetic molecules.
Evaluation of anti-aging of EPS in vivo
CAT is the most important enzyme to provide homeostasis for hydrogen peroxide. The physiological variation of CAT concentration in different organs and tissues leads to different steady-state levels of hydrogen peroxide concentration for the same rate of hydrogen peroxide generation (Chance et al. 1979). As shown in Table 1, the maximum CAT activities of 10.12 ± 0.05 U/mg, 5.93 ± 0.06 U/mg and 0.43 ± 0.02 U/mL in heart, kidney and blood were observed in group III, which were 31.60 ± 3.26, 54.89 ± 2.10, and 59.43 ± 17.84 % higher than that in group IV, respectively. On the other hand, the maximum CAT activities of 4.37 ± 0.05 U/mg in liver were observed in group II, which was 95.58 ± 3.85 % higher than that in group IV. We can conclude that the activity of CAT in heart, kidney and blood was improved by high concentration of EPS.
MDA is considered as a main indicator of endogenous lipid peroxidation, reflecting the content of free radicals produced by lipid peroxidation (Niki et al. 2005). As shown in Table 1, the MDA contents of group III in heart, liver, kidney, and blood reached 1.04 ± 0.02, 0.62 ± 0.03, 7.07 ± 0.02, and 8.26 ± 0.04 nmol/mL, respectively, which were 47.36 ± 7.71, 56.39 ± 6.94, 24.01 ± 1.43, and 30.32 ± 5.01 % lower than that in group I, 25.59 ± 3.62, 8.30 ± 6.27, 5.56 ± 1.41, and 23.17 ± 0.50 % lower than that in group II, and 122.04 ± 6.93, 110.14 ± 12.83, 32.55 ± 0.44, and 43.56 ± 0.56 % lower than that in group V, respectively.
The T-AOC reflects or represents the capacity of the non-enzymatic antioxidant defense system (Li et al. 2013). As shown in Table 1, the maximum the T-AOC contents in liver, kidney and blood of group III were 1.14 ± 0.03, 0.54 ± 0.03 and 0.35 ± 0.01, which were 105.45 ± 4.78, 58.47 ± 13.28, and 87.79 ± 23.13 % higher than that in group IV, respectively. In addition, the maximum T-AOC content (0.66 ± 0.04) in heart was observed in group II, which was 5.26 ± 8.78 % higher than that in group IV. The results showed that the T-AOC activity could be improved in kidney and blood by high concentration of EPS.
Conclusion
Monosaccharide composition analysis revealed that the EPS is a heteropolysaccharide composed of rhamnose, glucose and galactose. FT-IR analysis indicated that furanose-ring was observed in EPS-1 and EPS-2. The antioxidant capacities analysis showed that the antioxidant ability of EPS-2 was stronger than that of EPS and EPS-1 in vitro. EPS demonstrated positive anti-aging potential in mice and may be useful as a natural antioxidant for food industry.
References
Ames BN, Shigenaga MK, Hagen TM (1993) Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci USA 90:7915–7922
Benzie IFF, Stain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “anti oxidant power”, the FRAP assay. Anal Biochem 239:70–76
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT 28:25–30
Cai WR, Xie LL, Chen Y, Zhang H (2013) Purification, characterization and anticoagulant activity of the polysaccharides from green tea. Carbohydr Polym 92:1086–1090
Cao J, Zhang HJ, Xu CP (2014) Culture characterization of exopolysaccharides with antioxidant activity produced by Pycnoporus sanguineus in stirred-tank and airlift reactors. J Taiwan Inst Chem Eng doi.org/10.1016/j.jtice.2014.05.005
Castroa AJG, Castro LSEPW, Santos MSN, Faustino MGC, Pinheiro TS, Dore CMPG, Basei IG, Leite EL (2014) Anti-inflamatory, anti-angiogenenic and antioxidant activities of polysaccharide-rich extract from fungi Caripia montagnei. Biomed Prev Nutr 4:121–129
Cerna M, Barros AS, Nunes A, Rocha SM, Delgadillo I, Copikova J et al (2003) Use of FT-IR spectroscopy as a tool for the analysis of polysaccharide food additives. Carbohydr Polym 51:383–389
Chance B, Sies H, Boveris A (1979) Hydroperoxide metabolism in mammalian organs. Physiol Rev 59:527
Chen W, Zhao Z, Chen SF, Li YQ (2008) Optimization for the production of exopolysaccharide from Fomes fomentarius in submerged culture and its antitumor effect in vitro. Bioresour Technol 99:3187–3194
Chen Y, Mao WJ, Wang BF, Zhou LN, Gu QQ, Chen YL, Zhao CQ, Li N, Wang CY, Shan JM, Yan MX, Lin C (2013) Preparation and characterization of an extracellular polysaccharide produced by the deep-sea fungus Penicillium griseofulvum. Bioresour Technol 132:178–181
Dubois M, Gillers KA, Hamilton JK (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 2:350–352
Fan LF, Ashok P, Carlos RS (2001) Production of Flammulina velutipes on coffee husk and coffee spent-ground. Braz Arch Biol Tech 44:205–212
Fan LP, Li JW, Deng KQ, Ai LZ (2012) Effects of drying methods on the antioxidant activities of polysaccharides extracted from Ganoderma lucidum. Carbohydr Polym 87:1849–1854
Fan JL, Wu ZW, Zhao TH, Sun Y, Ye H, Xu RJ, Zeng XX (2014) Characterization, antioxidant and hepatoprotective activities of polysaccharides from Ilex latifolia Thunb. Carbohydr Polym 110:990–997
Gan D, Ma L, Jiang C, Xu R, Zeng X (2011) Production, preliminary characterization and antitumor activity in vitro of polysaccharides from the mycelium of Pholiota dinghuensis Bi. Carbohydr Polym 84:997–1003
Gao CJ, Wang YH, Wang CY, Wang ZY (2013) Antioxidant and immunological activity in vitro of polysaccharides from Gomphidius rutilus mycelium. Carbohydr Polym 92:2187–2192
Halliwell B (1996) Antioxidants in human health and disease. Annu Rev Nutr 16:33–50
Kohen R, Nyska A (2002) Oxidation of biological systems: oxidative stress phenomena, antioxidants, redox reactions, and method for their quantification. Toxicol Pathol 30:620–650
Kong KW, Mat-Junit S, Aminudin N, Ismail A, Abdul-Aziz A (2012) Antioxidant activities and polyphenolics from the shoots of Barringtonia racemosa (L.) Spreng in a polar to apolar medium system. Food Chem 134:324–332
Kumar CG, Joo HS, Choi JW, Koo YM, Chang CS (2004) Purification and characterization of an extracellular polysaccharide from haloalkalophilic Bacillus sp. I-450. Enzyme Microb Tech 34:673–681
Leong LP, Shui G (2002) An investigation of antioxidant capacity of fruits in Singapore markets. Food Chem 76:69–75
Leung MYK, Fung KP, Choy YM (1997) The isolation and characterization of an immunomodulatory and anti-tumor polysaccharide preparation from Flammulina velutipes. Immunopharm 35:255–263
Li JE, Nie SP, Xie MY (2013) Chemical composition and antioxidant activities in immumosuppressed mice of polysaccharides isolated from Mosla chinensis Maxim cv. Jiangxiangru. Int Immunopharmacol 17:267–274
Lin JW, Jia J, Shen YH, Zhong M, Chen LJ, Li HG, Ma H, Guo ZF, Qi MF, Liu LX, Li TL (2013) Functional expression of FIP-fve, a fungal immunomodulatory protein from the edible mushroom Flammulina velutipes in Pichia pastoris GS115. J Biotechnol 168:527–533
Liu J, Jia L, Kan J, Jin CH (2013) In vitro and in vivo antioxidant activity of ethanolic extract of white button mushroom (Agaricus bisporus). Food Chem Toxicol 51:310–316
Liu Y, Du YQ, Wang JH, Zha XQ, Zhang JB (2014) Structural analysis and antioxidant activities of polysaccharide isolated from Jinqian mushroom. Int J Biol Macromol 64:63–68
Ma LS, Chen HX, Zhu WC, Wang ZS (2013) Effect of different drying methods on physicochemical properties and antioxidant activities of polysaccharides extracted from mushroom Inonotus obliquus. Food Res Int 50:633–640
Niki E, Yoshida Y, Saito Y (2005) Lipid peroxidation: mechanisms, inhibition, and biological effects. Biochem Biophys Res Commun 338:668–676
Oyaizu M (1986) Antioxidative activity of browning products of glucosamine fractionated by organic solvent and thin layer chromatography. Jpn J Nutr 35:771–775
Pang XB, Yao WB, Yang XB, Xie C, Liu D, Zhang J, Gao XD (2007) Purification, characterization and biological activity on hepatocytes of a polysaccharide from Flammulina velutipes mycelium. Carbohydr Polym 70:291–297
Placer ZA, Cushman LL, Johnson BC (1966) Estimation of product of lipid peroxidation, malondialdehyde in biochemical system. Anal Biochem 16:359–367
Qiao DL, Hu B, Gan D, Sun Y, Ye H, Zeng X (2009) Extraction optimized by using response surface methodology, purification and preliminary characterization of polysaccharides from Hyriopsis cumingii. Carbohydr Polym 76:422–429
Schlorff EC, Husain K, Somani SM (1999) Dose- and time-dependent effects of ethanol on plasma antioxidant system in rat. Alcohol 2:97
Smiderle FR, Carbonero ER, Sassaki GL, Gorin PAJ, Iacomini M (2008) Characterization of a heterogalactan: some nutritional values of the edible mushroom Flammulina velutipes. Food Chem 108:329–333
Smironff N, Cumbes QJ (1989) Hyroxyl radical scavenging activity of compatible solutes. Phytochemistry 28:1057–1060
Stewar RC, Beewley JD (1980) Lipid peroxidation associated with accelerated aging of soybean areas. Plant Physiol 65:245–248
Su CA, Xu XY, Liu DY, Wu M, Zeng FQ, Zeng MY, Wei W, Jiang N, Luo X (2013) Isolation and characterization of exopolysaccharide with immunomodulatory activity from fermentation broth of Morchella conica. DARU J Pharm Sci 21:5
Wang ZY, Zhou F, Quan Y (2014a) Antioxidant and immunological activity in vitro of polysaccharides from Phellinus nigricans mycelia. Int J Biol Macromol 64:139–143
Wang ZY, Wang CY, Quan Y (2014b) Extraction of polysaccharides from Phellinus nigricans mycelia and their antioxidant activities in vitro. Carbohydr Polym 99:110–115
Wu DM, Duan WQ, Liu Y, Cen Y (2010) Anti-inflammatory effect of the polysaccharides of golden needle mushroom in burned rats. Int J Biol Macromol 46:100–103
Wu SH, Li F, Jia SY, Ren HT, Gong GL, Wang YY, Lv ZS, Liu Y (2014) Drying effects on the antioxidant properties of polysaccharides obtained from Agaricus blazei Murrill. Carbohydr Polym 103:414–417
Xing R, Liu S, Guo Z, Yu H, Zhong Z, Ji X et al (2008) Relevance of molecular weight of chitosan-N-2-hydroxypropyl trimethyl ammonium chloride and their antioxidant activities. Eur J Med Chem 43:336–340
Yang WJ, Fang Y, Liang J, Hua QH (2011) Optimization of ultrasonic extraction of Flammulina velutipes polysaccharides and evaluation of its acetylcholinesterase inhibitory activity. Food Res Int 44:1269–1275
Yang WJ, Pei F, Shi Y, Zhao LY, Fang Y, Hu QH (2012) Purification, characterization and anti-proliferation activity of polysaccharides from Flammulina velutipes. Carbohydr Polym 88:474–480
Ye H, Wang K, Zhou C, Liu J, Zeng X (2008) Purification, antitumor and antioxidant activities in vitro of polysaccharides from the brown seaweed Sargassum pallidum. Food Chem 111:428–432
Yuan JF, Zhang ZQ, Fan ZC, Yang JX (2008) Antioxidant effects and cytotoxicity of three purified polysaccharides from Ligusticum chuanxiong Hort. Carbohydr Polym 74:822–827
Zhang JJ, Ma Z, Zheng L, Zhai GY, Wang LQ, Jia MS, Jia L (2014) Purification and antioxidant activities of intracellular zinc polysaccharides from Pleurotus cornucopiae SS-03. Carbohydr Polym 111:947–954
Zhao L, Dong Y, Chen G, Hu Q (2010) Extraction, purification, characterization and antitumor activity of polysaccharides from Ganoderma lucidum. Carbohydr Polym 80:783–789
Zheng JQ, Wang JZ, Shi CW, Mao DB, He PX, Xu CP (2014) Characterization and antioxidant activity for exopolysaccharide from submerged culture of Boletus aereus. Proc Biochem 49:1047–1053
Acknowledgments
The authors gratefully acknowledge the financial supports by Mushroom Technology System of Shandong Province (SDAIT-11-011-07).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, Z., Cui, F., Gao, X. et al. Purification, characterization, antioxidant activity and anti-aging of exopolysaccharides by Flammulina velutipes SF-06. Antonie van Leeuwenhoek 107, 73–82 (2015). https://doi.org/10.1007/s10482-014-0305-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-014-0305-2