Abstract
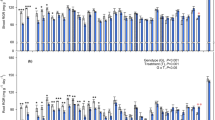
Nothofagus antarctica, is the most important species of the silvopastoral systems of southern Patagonia. However, there is limited information on the ecophysiological response mechanisms of seedlings for survival under different resource availability regimes, and whether such processes may differ between provenances. In this study, we evaluated the functional response to water shortage of seedlings of four N. antarctica provenances grown under controlled conditions. After an initial full irrigation period, seedlings were subjected to a water shortage cycle. Measurements of the diameter at the base of the stem, net photosynthesis (An), stomatal conductance to water vapour (gs), predawn water potential (Ψpd), osmotic potential at maximum and zero turgor (Π100; Π0), relative water content at zero turgor (RWC0) and maximum modulus of elasticity (Emax) were carried out during the experimental period. Differences between provenances concerning An and gs rates were recorded under full irrigation conditions, but were not detected in association with drought stress. All provenances had similar osmotic potential values (Π100 and Π0) at full irrigation, but water shortage led to significant osmotic potential differences between provenances. During the whole experimental period, no significant differences on RWC0 and Emax values were recorded among provenances. Under full irrigation conditions, all N. antarctica provenances had a similar physiological performance which suggests that environmental conditions may be the main driver for phenotypic differences in this species. From the osmotic adjustment results gathered, it is derived that a major osmoregulation capacity in response to water shortage is a common feature of all the N. antarctica provenances evaluated.




Similar content being viewed by others
References
Achten WMJ, Maes WH, Reubens B, Mathijs E, Singh VP, Verchot L, Muys B (2010) Biomass production and allocation in Jatro phacurcas L. Seedlings under different levels of drought stress. Biomass Bioener 34:667–676
Aranda I, Gil L, Pardos JA (2004) Osmotic adjustment in two temperate oak species [Quercus pyrenaica Willd and Quercus petraea (Matt.) Liebl] of the Iberian Peninsula in response to drought. Invest Agrar 13:339–345
Arend M, Kuster T, Gűnthardt-Goerg MS, Dobbertin M (2011) Provenance-specific growth responses to drought and air warming in three Eurpean oak species (Quercusrobur, Q. petraea and Q. pubescens). Tree Physiol 31:287–297
Atzmon N, Moshe Y, Schiller G (2004) Ecophysiological response to severe drought in Pinus halepensis Mill. Trees of two provenances. Plant Ecol 171:15–22
Bahamonde HA, Peri PL, Monelos LH, Martínez Pastur G (2011) Aspectos ecológicos de la regeneración por semillas en bosques nativos de Nothofagus antarctica en Patagonia Sur, Argentina. Bosque 32:20–29
Bahamonde HA, Peri PL, Monelos LH, Martínez Pastur G (2013) Regeneración por semillas en bosques nativos de Nothofagus antarctica bajo uso silvopastoril en Patagonia Sur, Argentina. Bosque 34:89–101
Bahamonde HA, Sánchez-Gómez D, Gyenge J, Peri PL, Aranda I (2014) Interaction of light and water stress on the ecophysiological response of Nothofagus antarctica (G. Forster) Oerst. In: Book of proceedings XII Portuguese-Spanish Symposium on Plant Water relations, pp 101–104
Bahamonde HA, Peri PL, Martínez Pastur G, Monelos L (2015) Litterfall and nutrients return in Nothofagus antarctica forests growing in a site quality gradient with different management uses in Southern Patagonia. Eur J For Res 134:113–124
Bahamonde HA, Lencinas MV, Martínez Pastur G, Monelos L, Soler R, Peri PL (2016a) Ten years of seed production and establishment of regeneration measurements in Nothofagus antarctica forests under different crown cover and quality sites, in Southern Patagonia. Agrofor Syst. https://doi.org/10.1007/s10457-016-9999-7
Bahamonde HA, Sosa Lovato S, Gesto E, Martínez Pastur G, Lencinas MV, Soler R, Peri PL (2016b) Variación temporal y espacial de la humedad de suelo y las raíces finas en bosques de ñire bajo uso silvopastoril en la Patagonia sur. Book of proceedings V Jornadas Forestales, Esquel, Argentina, 9-11 de Noviembre de 2016, pp 33–37
Bréda N, Huc R, Granier A, Dreyer E (2006) Temperate forest trees and stands under severe drought: a review of ecophysiological responses, adaptation processes and long-term consequences. Ann For Sci 63:625–644
Bucci SJ, Scholz FG, Campanello PI, Montti L, Jimenez-Castillo M, Rockwell FA, La Manna L, Guerra P, Lopez Bernal P, Troncoso O, Enricci J, Holbrook MN, Goldstein G (2013) Hydraulic differences along the water transport system of South American Nothofagus species: do leaves protect the stem functionality? Tree Physiol 32:880–893
Calabria C, Puntieri J (2008) Desarrollo foliar y caulinar de las unidades de alargamiento de Nothofagus dombeyi (Nothofagaceae) en condiciones de alta y baja luminosidad. Bol Soc Argent Bot 43:19–30. http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S1851-23722008000100002&lng=es&nrm=iso>. Accessed 15 Oct 2015
Collado L (2001) Los Bosques de Tierra del Fuego. Análisis de su estratificación mediante imágenes satelitales para el inventario forestal de la Provincia. Multequina 10:1–16
Corcuera L, Camarero JJ, Gil-Pelegrín E (2002) Functional groups in Quercus species derived from the analysis of pressure-volume curves. Trees Struct Funct 16(7):465–472. https://doi.org/10.1007/s00468-002-0187-1
Curt T, Coll L, Prévosto B, Balandier P, Kunstler G (2005) Plasticity in growth, biomass allocation and root morpfhology in beech seedlings as induced by irradiance and herbaceous competition. Ann For Sci 62:51–60
Dettmann S, Pérez CA, Thomas FM (2013) Xylem anatomy and calculated hydraulic conductance of four Nothofagus species with contrasting distribution in South-Central Chile. Trees 27:685–696
Flexas J, Escalona JM, Medrano H (1999) Water stress induces different levels of photosynthesis and electron transport rate regulation in grapevines. Plant Cell Environ 22:39–48
García-Plazaola JI, Becerril JM (2000) Effects of drought on photoprotective mechanisms in European beech (Fagus sylvatica L.) seedlings from different provenances. Trees 14:485–490
Gargaglione V, Peri PL, Rubio G (2013) Partición diferencial de nutrientes en árboles de Nothofagus antarctica creciendo en un gradiente de calidades de sitio en Patagonia Sur. Bosque 34:291–302
Golding AJ, Johnson GN (2003) Down-regulation of linear and activation of cycling electron transport during drought. Planta 218:107–114
Hovenden MJ, Brodribb T (2000) Altitude of origin influences stomatal conductance and therefore maximum assimilation rate in Southern Beech, Nothofagus cunninghamii. Aust J Plant Physiol 27:451–456
Hovenden MJ, Vander Schoor JK (2006) The response of leaf morphology to irradiance depends on altitude of origin in Nothofagus cunninghamii. New Phytol 169:291–297
Ladd B, Peri PL, Pepper DA, Silva LCR, Sheil D, Bonser SP, Laffan SW, Amelung W, Ekblad A, Eliasson P, Bahamonde H, Duarte-Guardia S, Bird M (2014) Carbon isotopic signatures of soil organic matter correlate with leaf area index across woody biomes. J Ecol 102(6):1606–1611
Lajtha K, Getz J (1993) Photosynthesis and wáter use efficiency in pinyon-juniper communities along an elevation gradient in northern New Mexico. Oecologia 94:95–101
Liu F, Stützel H (2004) Biomass partitioning, specific leaf area, and water use efficiency of vegetable amaranth (Amaranthus spp.) in response to drought stress. Sci Hortic 102:15–27
Ministry of environment and sustainable development of Argentina (2015) Third national communication about climate change. http://www.ambiente.gov.ar/archivos/web/ProyTerceraCNCC/file/02-Sumario.pdf. Accessed 2 Mar 2016
Nguyen-Queyrens A, Bouchet-Lannat F (2003) Osmotic adjustment in three-year-old seedlings of five provenances of maritime pine (Pinus pinaster) in response to drought. Tree Physiol 23:397–404
Ogaya R, Peñuelas J (2003) Comparative field study of Quercus ilex and Philly realatifolia: photosynthetic response to experimental drought conditions. Environ Exp Bot 50:137–148
Peri PL, Ormaechea SG (2013) Relevamiento de los bosques nativos de ñire (Nothofagus antarctica) en Santa Cruz: base para su conservación y manejo. Ediciones INTA web. http://inta.gob.ar/documentos/libro-201crelevamiento-de-los-bosques-nativos-de-nire-nothofagus-antarctica-en-santa-cruz-base-para-su-conservacion-y-manejo201d. Accessed 10 Oct 2015
Peri PL, Martínez Pastur G, Lencinas MV (2009) Photosynthetic response to different light intensities and wáter status of two main Nothofagus species of southern Patagonian Forest, Argentina. J For Sci 55:101–111
Peri PL, Gargaglione V, Martínez Pastur G, Lencinas MV (2010) Carbon accumulation along a stand development sequence of Nothofagus antarctica forests across a gradient in site quality in Southern Patagonia. Forest Ecol Manag 260:229–237
Peri PL, Bahamonde HA, Lencinas MV, Gargaglione V, Soler R, Ormaechea S, Pastur GM (2016) A review of silvopastoral systems in native forests of Nothofagus antarctica in southern Patagonia, Argentina. Agrofor Syst 90(6):933–960
Piper FI, Corcuera LJ, Alberdi M, Lusk C (2007) Differential photosynthetic and survival responses to soil drought in two evergreen Nothofagus species. Ann For Sci 64:447–452
Premoli AC, Brewer CA (2007) Environmentalv. genetically driven variation in ecophysiological traits of Nothofagus pumilio from contrasting elevations. Aust J Bot 55:585–591
Ramírez-Valiente JA, Sánchez-Gómez D, Aranda I, Valladares F (2010) Phenotypic plasticity and local adaptation in leaf ecophysiological traits of 13 contrasting cork oak populations under different water availabilities. Tree Physiol 30:618–627
Rasband WS (1997–2004) ImageJ. http://rsb.info.nih.gov/ij/ed. Bethesda, Maryland: National Institutes of Health
Read J, Farquhar G (1991) Comparative studies in Nothofagus (Fagaceae). I. Leaf carbon isotope discrimination. Funct Ecol 5:684–695
Read J, Hill RS, Hope GS (2010) Contrasting responses to water deficits of Nothofagus species from tropical New Guinea and high-latitude temperate forests: can rainfall regimes constrain latitudinal range? J Biogeogr 37:1962–1976
Sánchez-Gómez D, Robson TM, Gascó A, Gil-Pelegrín E, Aranda I (2013) Differences in the leaf functional traits of six beech (Fagus sylvatica) populations are reflected in their response to water limitation. Environ Exp Bot 87:110–119
Soler R, Martínez Pastur G, Peri PL, Lencinas MV, Pulido F (2013) Are silvopastoral systems compatible with forest regeneration? An integrative approach in southern Patagonia. Agrofor Syst 87:1213–1227
Tanai T (1986) Phytogeographic and phylogenetic history of the genus Nothofagus Bl. (Fagaceae) in the southern hemisphere. Journal of the Faculty of Science, Hokkaido University, series 4, 21:505–582
Valladares F, Sanchez-Gomez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94(6):1103–1116. https://doi.org/10.1111/j.1365-2745.2006.01176.x
Varela SA (2010) Respuestas morfológico-fisiológicas ante distintos tipos e intensidades de estrés en dos especies patagónicas del género Nothofagus con aptitud comercial. Master thesis, Universidad de Buenos Aires
Veblen TT, Donoso C, Kitzberger T, Rebertus AJ (1996) Ecology of Southern Chilean and Argentinean Nothofagus forests. In: Veblen TT, Hill RS, Read J (eds) The ecology and biogeography of Nothofagus forests. Yale University Press, New Haven, pp 293–353
Yin Ch, Peng Y, Zang R, Zhu Y, Li Ch (2005) Adaptative responses of Populus kangdingensis to drought stress. Physiol Plant 123:445–451
Acknowledgements
We are very grateful to Rosina Soler for helping with the collection of seeds and Victoria Fernández for helping with language revision.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahamonde, H.A., Sánchez-Gómez, D., Gyenge, J. et al. Thinking in the sustainability of Nothofagus antarctica silvopastoral systems, how differ the responses of seedlings from different provenances to water shortage?. Agroforest Syst 93, 689–701 (2019). https://doi.org/10.1007/s10457-017-0167-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-017-0167-5