Abstract
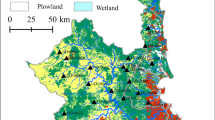
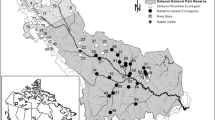
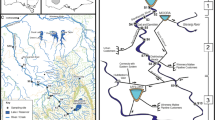
One of the main goals of ecology is to understand how the abiotic environment influences the biotic characteristics of the ecosystem. Various processes at multiple scales interact to affect the physical and chemical environments that are experienced by organisms, which ultimately influence community composition. We aimed to understand the processes that control benthic algae community composition within a watershed. We investigated the impact of both land cover and physiochemical variables on benthic algal community composition. We sampled benthic algae along with multiple habitat and water chemistry parameters within three microhabitats across eight sites along the mainstem of the Kiamichi River in southeastern Oklahoma. We used the benthic light availability model to assess the amount of light reaching the bottom of the stream. Additionally, we conducted a GIS analysis of the watershed to determine the land cover affecting each of these sites. Several of the in-stream site-scale variables that were measured (e.g., conductivity, pH and canopy cover) were strongly correlated with both position within the watershed and percent agriculture within the watershed. The physiochemical parameters that were correlated with watershed position and land cover were then used to understand the linkage with algae community composition. Algae genera composition was strongly correlated with both light reaching the bottom of the stream and conductivity. Our results suggest a hierarchy of factors that determine species composition and show the dependence of community composition on differing light regimes.






Similar content being viewed by others
References
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35:257–284. doi:10.1146/annurev.ecolsys.35.120202.110122
Atkinson CL, Vaughn CC, Forshay KJ, Cooper JT (2013) Aggregated filter-feeding consumers alter nutrient limitation: consequences for ecosystem and community dynamics. Ecology 94:1359–1369
Bergey EA, Weaver JE (2004) The influence of crevice size on the protection of epilithic algae from grazers. Freshw Biol 49:1014–1025
Biggs BJF (1995) The contribution of flood distrubance, catchment geology and land-use to the habitat template of periphyton in stream ecosystems. Freshw Biol 33:419–438
Biggs BJF, Smith RA (2002) Taxonomic richness of stream benthic algae: effects of flood disturbance and nutrients. Limnol Oceanogr 47:1175–1186
Burcher C, Valett H, Benfield E (2007) The land-cover cascade: relationships coupling land and water. Ecology 88:228–242
Cardinale BJ, Hillebrand H, Charles DF (2006) Geographic patterns of diversity in streams are predicted by a multivariate model of disturbance and productivity. J Ecol 94:609–618. doi:10.1111/j.1365-2745.2006.01107.x
Chazdon RL, Pearcy RW (1991) The importance of sunflecks for forest understory plants—photosynthetic machinery appears to be adapted to brief, unpredictable periods of radiation. Bioscience 41:760–766. doi:10.2307/1311725
Clarke KR (1993) Nonparametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143. doi:10.1111/j.1442-9993.1993.tb00438.x
Dillard GE (1999) Common freshwater algae of the United States: an illustrated key to the genera (excluding the diatoms). Gebrüder Borntraeger, Berlin
Dixit SS, Smol JP, Kingston JC, Charles DF (1992) Diatoms: powerful indicators of environmental change. Environ Sci Technol 26:23–33
Frazer GW, Canham CD, Lertzman KP (1999) Gap light analyzer (GLA), version 2.0: imaging software to extract canopy structure and gap light transmission indices from true-colour fisheye photographs, users manual and program documentation. Simon Fraser University, Burnaby, British Columbia and the Institute of Ecosystem Studies, Millbrook New York
Frissell CA, Liss WJ, Warren CE, Hurley MD (1986) A hierarchical framework for stream habitat classification—viewing streams in a watershed context. Environ Manag 10:199–214
Greenwood J, Lowe R (2006) The effects of ph on a periphyton community in an acidic wetland, USA. Hydrobiologia 561:71–82
Hansmann EW, Phinney HK (1973) Effects of logging on periphyton in coastal streams of oregon. Ecology 54:194–199. doi:10.2307/1934390
Hill BH, Herlihy AT, Kaufmannc PR, DeCelles SJ, Vander Borgh MA (2003) Assessment of streams of the eastern united states using a periphyton index of biotic integrity. Ecol Indic 2:325–338
Homer C, Huang C, Yang L, Wylie B, Coan M (2004) Development of a 2001 national land cover database for the United States. Photogramm Eng Remote Sens 70:829–840
Hynes HBN (1975) The stream and its valley. Verh Int Ver Limnol 19:1–15
Julian JP, Doyle MW, Powers SM, Stanley EH, Riggsbee JA (2008a) Optical water quality in rivers. Water Resour Res. doi:10.1029/2007wr006457
Julian JP, Doyle MW, Stanley EH (2008b) Empirical modeling of light availability in rivers. J Geophys Res Biogeosci. doi:10.1029/2007jg000601
Julian JP, Stanley EH, Doyle MW (2008c) Basin-scale consequences of agricultural land use on benthic light availability and primary production along a sixth-order temperate river. Ecosystems 11:1091–1105. doi:10.1007/s10021-008-9181-9
Kirk JTO (1994) Light and photosynthesis in aquatic ecosystems. Cambridge University Press, New York
Lawson LL (1999) Epilithic diatoms as indicators of stream condition in arid lotic ecosystems of the American southwest. Dissertation, University of Arizona
Likens GE, Bormann FH, Pierce RS, Reiners WA (1978) Recovery of a deforested ecosystem. Science 199:492–496. doi:10.1126/science.199.4328.492
Mallory MA, Richardson JS (2005) Complex interactions of light, nutrients and consumer density in a stream periphyton-grazer (tailed frog tadpoles) system. J Anim Ecol 74:1020–1028. doi:10.2307/3505251
Matthews WJ, Vaughn CC, Gido KB, Marsh-Matthews E (2005) Southern plains rivers. In: Benke AC, Cushing CE (eds) Rivers of North America. Academic Press, Burlington
Naymik J, Pan Y, Ford J (2005) Diatom assemblages as indicators of timber harvest effects in coastal oregon streams. J N Am Benthol Soc 24:569–584. doi:10.1899/03-054.1
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O'hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Wagner H (2011) Vegan: community ecology package. R package version 2.0-2. https://cran.r-project.org/web/packages/vegan/index.html
Pan Y, Stevenson RJ, Hill BH, Herlihy AT, Collins GB (1996) Using diatoms as indicators of ecological conditions in lotic systems: a regional assessment. J N Am Benthol Soc 15:481–495
Poff NL (1997) Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. J N Am Benthol Soc 16:391–409. doi:10.2307/1468026
Porter SD, Mueller DK, Spahr NE, Munn MD, Dubrovsky NM (2008) Efficacy of algal metrics for assessing nutrient and organic enrichment in flowing waters. Freshw Biol 53:1036–1054
Potapova MG, Charles DF (2002) Benthic diatoms in USA rivers: distributions along spatial and environmental gradients. J Biogeogr 29:167–187
Potapova MG, Charles DF (2003) Distribution of benthic diatoms in us rivers in relation to conductivity and ionic composition. Freshw Biol 48:1311–1328. doi:10.1046/j.1365-2427.2003.01080.x
Potapova MG, Charles DF (2005) Choice of substrate in algae-based water-quality assessment. J N Am Benthol Soc 24:415–427. doi:10.1899/03-111.1
Potapova MG, Charles DF (2007) Diatom metrics for monitoring eutrophication in rivers of the United States. Ecol Indic 7:48–70. doi:10.1016/j.ecolind.2005.10.001
Power ME, Stewart AJ (1987) Disturbance and recovery of an algal assemblage following flooding in an Oklahoma stream. Am Midl Nat 117:333–345. doi:10.2307/2425975
Pyne MI, Rader RB, Christensen WF (2007) Predicting local biological characteristics in streams: a comparison of landscape classifications. Freshw Biol 52:1302–1321. doi:10.1111/j.1365-2427.2007.01767.x
R Development Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Reavie ED, Jicha TM, Angradi TR, Bolgrien DW, Hill BH (2010) Algal assemblages for large river monitoring: comparison among biovolume, absolute and relative abundance metrics. Ecol Indic 10:167–177. doi:10.1016/j.ecolind.2009.04.009
Smucker NJ, Vis ML (2011) Diatom biomonitoring of streams: reliability of reference sites and the response of metrics to environmental variations across temporal scales. Ecol Indic 11:1647–1657. doi:10.1016/j.ecolind.2011.04.011
Snelder TH, Biggs BJF (2002) Multiscale river environment classification for water resources management. J Am Water Resour Assoc 38:1225–1239. doi:10.1111/j.1752-1688.2002.tb04344.x
Soininen J, Eloranta P (2004) Seasonal persistence and stability of diatom communities in rivers: are there habitat specific differences? Eur J Phycol 39:153–160. doi:10.1080/0967026042000201858
Stelzer RS, Lamberti GA (2001) Effects of N:P ratio and total nutrient concentration on stream periphyton community structure, biomass, and elemental composition. Limnol Oceanogr 46:356–367
Stevenson RJ (1997) Scale-dependent determinants and consequences of benthic algal heterogeneity. J N Am Benthol Soc 16:248–262. doi:10.2307/1468255
Stevenson RJ, Pan Y, Manoylov KM, Parker CA, Larsen DP, Herlihy AT (2008) Development of diatom indicators of ecological conditions for streams of the western us. J N Am Benthol Soc 27:1000–1016. doi:10.1899/08-040.1
Tabachnick BG, Fidell LS (2007) Using multivariate statistics. Pearson, Boston, MA
Taylor SL, Roberts SC, Walsh CJ, Hatt BE (2004) Catchment urbanisation and increased benthic algal biomass in streams: linking mechanisms to management. Freshw Biol 49:835–851. doi:10.1111/j.1365-2427.2004.01225.x
Tuchman M, Stevenson RJ (1980) Comparison of clay tile, sterilized rock, and natural substrate diatom communities in a small stream in southeastern Michigan, USA. Hydrobiologia 75:73–79. doi:10.1007/bf00006564
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Wang YK, Stevenson RJ, Metzmeier L (2005) Development and evaluation of a diatom-based Index of Biotic Integrity for the Interior Plateau Ecoregion, USA. J N Am Benthol Soc 24:990–1008
Weilhoefer CL, Pan Y (2006) Diatom assemblages and their associations with environmental variables in Oregon coast range streams, USA. Hydrobiologia 561:207–219. doi:10.1007/s10750-005-1615-1
Winemiller KO, Flecker AS, Hoeinghaus DJ (2010) Patch dynamics and environmental heterogeneity in lotic ecosystems. J N Am Benthol Soc 29:84–99. doi:10.1899/08-048.1
Winter JG, Duthie HC (2000) Stream biomonitoring at an agricultural test site using benthic algae. Can J Bot 78:1319–1325
Zampella RA, Laidig KJ, Lowe RL (2007) Distribution of diatoms in relation to land use and ph in blackwater coastal plain streams. Environ Manage 39:369–384
Acknowledgments
We thank Jason P. Julian for loaning us the fisheye lens and camera and associated equipment for canopy photographs. The Oklahoma Biological Survey provided support and transport to visit field sites. This manuscript benefited from reviews and comments by Elizabeth Bergey, Jason Julian, Caryn Vaughn and two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bas W. Ibelings.
Rights and permissions
About this article
Cite this article
Atkinson, C.L., Cooper, J.T. Benthic algal community composition across a watershed: coupling processes between land and water. Aquat Ecol 50, 315–326 (2016). https://doi.org/10.1007/s10452-016-9580-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-016-9580-5