Abstract
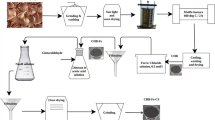
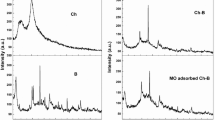
To anchor chelating groups with nitrogen and sulfur atoms on tannin, a novel adsorbent (tannin-thiosemicarbazide-formaldehyde resin) was prepared through Mannich reaction by using blank wattle tannin, thiosemicarbazide and formaldehyde as raw materials. The adsorption behaviour of Ag+ from aqueous solution on the resin was evaluated via batch adsorption experiments. Fourier transform infrared spectroscopy (FT–IR) and elemental analysis were applied to verify the successful immobilization. The surface morphology, thermal stability and pore structure of the resin were also characterized. The results showed that the adsorption isotherm of Ag+ for the resin was described well by the Freundlich model. Ag+ adsorption equilibrium was achieved within 180 min, and the kinetic data were better fitted by the pseudo-second-order kinetic equation than by the pseudo-first-order and intraparticle diffusion equations. The adsorption capacity first increased and then stabilized with increasing pH (ranging from 1.0 to 7.0), and the resin exhibited high selectivity towards Ag+ in relation to Pb2+, Cd2+, Ni2+ and Ca2+. After three regeneration and reuse cycles, the adsorption capacity reached 1.68 mmol/g (84.0% of removal efficiency). Based on the experimental results and findings from various characterization techniques, the mechanism of Ag+ adsorption onto the resin could be attributed to inner-sphere complexation and chelation between Ag+ and multiple electron-rich atoms ( N, O, and S), in which S atoms played the most important role.











Similar content being viewed by others
References
Nakhjiri, A.T., et al.: Recovery of precious metals from industrial wastewater towards resource recovery and environmental sustainability: A critical review. Desalination 527, 115510 (2022)
Biswas, F.B., et al.: Selective recovery of silver and palladium from acidic waste solutions using dithiocarbamate-functionalized cellulose. Chem. Eng. J. 407, 127225 (2021)
Pilśniak-Rabiega, M., Wolska, J.: Silver(I) recovery on thiomorpholine - modified functional polymer. Physicochem. Probl. Miner. Process. 58, 156609 (2022)
Vasileiadis, S., et al.: Silver toxicity thresholds for multiple soil microbial biomarkers. Environ. Sci. Technol. 52, 8745–8755 (2018)
Eckelman, M.J., Graedel, T.E.: Silver emissions and their environmental impacts: a multilevel assessment. Environ. Sci. Technol. 41, 6283–6289 (2007)
Tortella, G.R., et al.: Silver nanoparticles: toxicity in model organisms as an overview of its hazard for human health and the environment. J. Hazard. Mater. 390, 121974 (2020)
Kalčíková, G., et al.: An environmental concentration of aged microplastics with adsorbed silver signifcantly affects aquatic organisms. Water Res. 175, 115644 (2020)
Birloaga, I., Vegliò, F.: Overview on hydrometallurgical procedures for silver recovery from various wastes. J. Environ. Chem. Eng. 6, 2932–2938 (2018)
Zimmermann, P., et al.: Selective recovery of silver ions from copper-contaminated effluents using electrodialysis. Desalination 572, 117108 (2024)
Mitov, M., et al.: Silver recovery by microbial electrochemical snorkel and microbial fuel cell. Electrochim. Acta 408, 139941 (2022)
Wang, Z., et al.: Recovery of silver from dilute effluents via electrodeposition and redox replacement. J. Electrochem. Soc. 166, 266–274 (2019)
John, M., et al.: Recovery of Ag and Au from synthetic and industrial wastewater by 2-step ferritization and Lt-delafossite process via precipitation. J. Water Process. Eng. 30, 100532 (2019)
Cho, S.Y., Kim, T.Y., Sun, P.P.: Recovery of silver from leachate of silicon solar cells by solvent extraction with TOPO. Sep. Purif. Technol. 215, 516–520 (2019)
Virolainen, S., et al.: Ion exchange recovery of silver from concentrated base metal-chloride solutions. Hydrometallurgy 152, 100–106 (2015)
Yi, M., et al.: In-situ silver recovery for biofouling mitigation with catechol-assisted nanofiltration membrane. Desalination 547, 116233 (2023)
Huang, X., et al.: Fabrication of polyvinylidene fluoride and acylthiourea composite membrane and its adsorption performance and mechanism on silver ions. Sep. Purif. Technol. 315, 123675 (2023)
Shao, P., et al.: Mixed-valence molybdenum oxide as a recyclable sorbent for silver removal and recovery from wastewater. Nat. Commun. 14, 1365 (2023)
Abd El-Ghaffar, M.A., et al.: Extraction and separation studies of silver(I) and copper(II) from their aqueous solution using chemically modified melamine resins. Hydrometallurgy 96, 27–34 (2009)
Huang, Y., et al.: Anion-synergistic adsorption enhances the selective removal of silver ions from complex wastewater by chitosan-coated magnetic silica core-shell nanoparticles. J. Clean. Prod. 339, 130777 (2022)
Wang, P., et al.: Magnetic mesoporous calcium carbonate-based nanocomposites for the removal of toxic Pb(II) and Cd(II) ions from water. ACS Appl. Nano Mater. 3, 1272–1281 (2020)
Fan, J., et al.: Selective adsorption and recovery of silver from acidic solution using biomass-derived sulfur-doped porous carbon. ACS Appl. Mater. Interfaces 15, 40088–40099 (2023)
Ghomi, G.A., et al.: Biosorpion for sustainable recovery of precious metals from wastewater. J. Environ. Chem. Eng. 8, 103996 (2020)
Elwakeel, K.Z., et al.: 2-Mercaptobenzimidazole derivative of chitosan for silver sorption – Contribution of magnetite incorporation and sonication effects on enhanced metal recovery. Chem. Eng. J. 403, 126265 (2021)
Arbenz, A., Averous, L.: Chemical modification of tannins to elaborate aromatic biobased macromolecular architectures. Green Chem. 17, 2626–2646 (2015)
Kavitha, V.U., Kandasubramanian, B.: Tannins for wastewater treatment. SN Appl. Sci. 2, 1081 (2020)
Bacelo, H.A.M., Santos, S.C.R., Botelho, C.M.S.: Tannin-based biosorbents for environmental applications – A review. Chem. Eng. J. 303, 575–587 (2016)
Kim, Y.H., Nakano, Y.: Adsorption mechanism of palladium by redox within condensed-tannin gel. Water Res. 39, 1324–1330 (2005)
Ogata, T., Nakano, Y.: Mechanisms of gold recoveryfrom aqueous solutions using a novel tannin gel adsorbent synthesized from natural condensed tannin. Water Res. 39, 4281–4286 (2005)
Sun, X., et al.: Adsorptive removal of Cu(II) from aqueous solutions using collagen-tannin resin. J. Hazard. Mater. 186, 1058–1063 (2011)
Huang, X., Liao, X.P., Shi, B.: Hg(II) removal from aqueous solution by bayberry tannin-immobilized collagen fiber. J. Hazard. Mater. 170, 1141–1148 (2009)
Huang, X., et al.: Adsorptive recovery of Au3+ from aqueous solutions using bayberry tannin-immobilized mesoporous silica. J. Hazard. Mater. 183, 793–798 (2010)
Wang, Y., et al.: Enhanced adsorption of Pb(II) ions from aqueous solution by persimmon tannin-activated carbon composites. J. Wuhan Univ. Technol. Mater. Sci. Ed. 28, 650–657 (2013)
Xu, Q., et al.: Adsorption of Cu (II), Pb (II) and Cr (VI) from aqueous solutions using black wattle tannin-immobilized nanocellulose. J. Hazard. Mater. 339, 91–99 (2017)
Fan, R., Min, H., Hong, X.: Plant tannin immobilized Fe3O4@SiO2 microspheres: a novel and green magnetic bio-sorbent with superior adsorption capacities for gold (III) and palladium(II). J. Hazard. Mater. 364, 780–790 (2019)
Liu, F., et al.: Au(III) adsorption and reduction to gold particles on cost-effective tannin acid immobilized dialdehyde corn starch. Chem. Eng. J. 370, 228–236 (2019)
Wang, Z., et al.: Equilibrium, kinetics and mechanism of Au3+, Pd2+ and Ag+ ions adsorption from aqueous solutions by graphene oxide functionalized persimmon tannin. Mat. Sci. Eng. C-Mater. 79, 227–236 (2017)
Pearson, R.G.: Hard and soft acids and bases. J. Am. Chem. Soc. 85, 3533–3539 (1963)
Gurung, M., et al.: Persimmon tannin-based new sorption material for resource recycling and recovery of precious metals. Chem. Eng. J. 228, 405–414 (2013)
Gurung, M., et al.: Selective recovery of precious metals from acidic leach liquor of circuit boards of spent mobile phones using chemically modified persimmon tannin gel. Ind. Eng. Chem. Res. 51, 11901–11913 (2012)
Gurung, M., et al.: N-aminoguanidine modified persimmon tannin: a new sustainable material for selective adsorption, preconcentration and recovery of precious metals from acidic chloride solution. Bioresour. Technol. 129, 108–117 (2013)
Xiong, Y.C., et al.: Selective recovery of precious metals by persimmon waste chemically modified with dimethylamine. Bioresour. Technol. 100, 4083–4089 (2009)
Li, X., et al.: Chitosan modification persimmon tannin bioadsorbent for highly efficiency removal of Pb(II) from aqueous environment: the adsorption equilibrium, kinetics and thermodynamics. Environ. Technol. 40, 112–124 (2019)
Ricci, A., et al.: Application of fourier transform infrared (FTIR) spectroscopy in the characterization of tannins. Appl. Spectrosc. Rev. 50, 407–442 (2015)
Wang, F., et al.: Superior Au-adsorption performance of aminothiourea-modified waste cellulosic biomass. J. Cent. South Univ. 25, 2992–3003 (2018)
Liu, P., et al.: Adsorption of silver ion from the aqueous solution using a polyvinylidene fluoride functional membrane bearing thiourea groups. J. Water Process Eng. 34, 101184 (2020)
Lin, G., et al.: Synthesis and evaluation of thiosemicarbazide functionalized corn bract for selective and efficient adsorption of Au(III) from aqueous solutions. J. Mol. Liq. 258, 235–243 (2018)
Zhou, L., Liu, J., Liu, Z.: Adsorption of platinum(IV) and palladium(II) from aqueous solution by thiourea-modified chitosan microspheres. J. Hazard. Mater. 172, 439–446 (2009)
Xiong, Y., et al.: Selective recovery of Ag(I) coordination anion from simulate nickel electrolyte using corn stalk based adsorbent modified by ammonia–thiosemicarbazide. J. Hazard. Mater. 301, 277–285 (2016)
Deng, S., et al.: Polyacrylonitrile-based fiber modified with thiosemicarbazide by microwave irradiation and its adsorption behavior for Cd(II) and Pb(II). J. Hazard. Mater. 307, 64–72 (2016)
Chen, Z., et al.: Synthesis of silica supported thiosemicarbazide for Cu(II) and Zn(II) adsorption from ethanol: A comparison with aqueous solution. Fuel 286, 119287 (2021)
Wang, Z.H., et al.: Enhanced electrochemical performance of porous activated carbon by forming composite with graphene as high-performance supercapacitor electrode material. J. Nanopart. Res. 19, 77–86 (2017)
Liu, D., et al.: Organic-inorganic hybrid mesoporous titanium silica material as bi-functional heterogeneous catalyst for the CO2 cycloaddition. Fuel 244, 196–206 (2019)
Dong, Z., et al.: Performance and mechanism of selective adsorption of silver to L-cysteine functionalized cellulose microsphere. Cellulose 27, 3249–3261 (2020)
El-Ghaffar, M.A.A., Abdel-Wahab, Z.H., Elwakeel, K.Z.: Extraction and separation studies of silver(I) and copper(II) from their aqueous solution using chemically modified melamine resins. Hydrometallurgy 96, 27–34 (2009)
Du, J.F., et al.: Facile fabrication of sodium styrene sulfonate-grafted ethylene-vinyl alcohol copolymer as adsorbent for ammonium removal from aqueous solution. Environ. Sci. Pollut. R. 25, 27235–27244 (2018)
Elshehy, E.A., et al.: Selective recovery of silver(I) ions from E-waste using cubically multithiolated cage mesoporous monoliths. Eur. J. Inorg. Chem. 2017, 4823–4833 (2017)
Condomitti, U., et al.: Silver recovery using electrochemically active magnetite coated carbon particles. Hydrometallurgy 147, 241–245 (2014)
Abd El-Ghaffar, M.A., et al.: Adsorption of silver(I) on synthetic chelating polymer derived from 3-amino-1,2,4-triazole-5-thiol and glutaraldehyde. Chem. Eng. J. 151, 30–38 (2009)
Zhang, M., et al.: Ion-imprinted chitosan gel beads for selective adsorption of Ag+ from aqueous solutions. Carbohydr. Polym. 130, 206–212 (2015)
Tran, H.N., et al.: Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: A comparison study. J. Environ. Chem. Eng. 4(3), 2671–3268 (2016)
Goldberg, S.: Inconsistency in the triple layer model description of ionic strength dependent boron adsorption. J. Colloid Interf. Sci. 285, 509–517 (2005)
Al-Degs, Y.S., et al.: Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon. Dyes Pigments 77, 16–23 (2008)
An, F.Q., et al.: Selective adsorption of AuCl4- on chemically modified D301 resin with containing N/S functional polymer. J. Environ. Chem. Eng. 5, 10–15 (2017)
Villalobos, L.F., Yapici, T., Peinemann, K.V.: Poly-thiosemicarbazide membrane for gold recovery. Sep. Purif. Technol. 136, 94–104 (2014)
Fu, L., et al.: Selective adsorption of Ag+ by silica nanoparticles modified with 3-amino-5-mercapto-1,2,4-triazole from aqueous solutions. J. Mol. Liq. 241, 292–300 (2017)
Rahman, M.M., et al.: Optimization, kinetic and thermodynamic studies for removal of Brilliant Red (X-3B) using tannin gel. J. Environ. Chem. Eng. 2(1), 76–83 (2014)
Acknowledgements
This work was supported by the Science and Technology Support Program of Sichuan Province (No. 2019YJ0399).
Funding
This work was supported by the Science and Technology Support Program of Sichuan Province (No. 2019YJ0399).
Author information
Authors and Affiliations
Contributions
X.B. Sun developed the concept, designed the experiments and drafted the manuscript. S.Y. Yin performed the experiments and acquired the data. Y.H. You reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, X., Yin, S. & You, Y. Synthesis and evaluation of tannin-thiosemicarbazide-formaldehyde resin for selective adsorption of silver ions from aqueous solutions. Adsorption (2024). https://doi.org/10.1007/s10450-024-00492-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10450-024-00492-5