Abstract
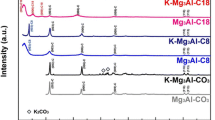
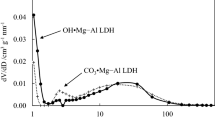
Potassium-promoted layered double hydroxide (LDH)-derived materials are suitable elevated temperature CO2 adsorbents for pre-combustion CO2 capture. A challenge for the commercialization of LDHs as efficient CO2 adsorbents is their low capacities (ca. 0.5–0.6 mmol/g@400 °C) due to their hydrogen-bonded stacked structure. In this study, the aqueous miscible organic solvent treatment (AMOST) was used to exfoliate Mg3Al–CO3 LDH into nanosheets with a flower-like morphology, resulting in high surface areas of 287 and 212 m2/g for CC1 (washed with ethanol) and CC2 (washed with acetone), respectively. The exfoliated LDH structure exposed more interlayered CO2 active sites and promoters for alkali metal modification. Six impregnation solvents, water, acetone, ethanediol, ethanol, DMAC, and methanol were screened to optimize the CO2 uptake of 20 wt% K2CO3-promoted CC1. K2CO3/CC1(ed) using ethanediol as the impregnation solvent reached a CO2 working capacity of 1.46 mmol/g at 400 °C in the first cycle and 1.23 mmol/g after 10 cycles, twice the capacity of the commercial K2CO3/MG70. Material characterization indicated that the unexpectedly high performance of K2CO3/CC1(ed) could be attributed to the uniform K+ dispersion on the surface of K2CO3/CC1(ed) rather than bulk phase formation and the release of the residual solvent during calcination that could generate more paths for CO2 diffusion.





Similar content being viewed by others
References
Bhatta, L.K.G., Subramanyam, S., Chengala, M.D., Olivera, S., Venkatesh, K.: Progress in hydrotalcite like compounds and metal-based oxides for CO2 capture: a review. J. Clean. Prod. 103, 171–196 (2015). https://doi.org/10.1016/j.jclepro.2014.12.059
Bui, M., Adjiman, C.S., Bardow, A., Anthony, E.J., Boston, A., Brown, S., Fennell, P.S., Fuss, S., Galindo, A., Hackett, L.A., Hallett, J.P., Herzog, H.J., Jackson, G., Kemper, J., Krevor, S., Maitland, B.M., Adjiman, C.S., Bardow, A., Anthony, E.J., Boston, A., Brown, S., Fennell, P.S., Fuss, S., Galindo, A., Hackett, L.A., Hallett, J.P., Herzog, H.J., Jackson, G., Kemper, J., Krevor, S., Maitland, G.C., Matuszewski, M., Metcalfe, I.S., Petit, C., Puxty, G., Reimer, J., Reiner, D.M., Rubin, E.S., Scott, S.A., Shah, N., Smit, B., Trusler, J.P.M., Webley, P., Wilcox, J., Mac Dowell, N.: Carbon capture and storage (CCS): the way forward. Energy Environ. Sci. 11(5), 1062–1176 (2018). https://doi.org/10.1039/c7ee02342a
Chen, C.P., Yang, M.S., Wang, Q., Buffet, J.C., O'Hare, D.: Synthesis and characterisation of aqueous miscible organic-layered double hydroxides. J. Mater. Chem. A 2(36), 15102–15110 (2014). https://doi.org/10.1039/c4ta02277g
Chen, C., Felton, R., Buffet, J.-C., O'Hare, D.: Core-shell SiO2@LDHs with tuneable size, composition and morphology. Chem. Commun. 51(16), 3462–3465 (2015a). https://doi.org/10.1039/c4cc10008e
Chen, C.P., Wangriya, A., Buffet, J.C., O'Hare, D.: Tuneable ultra high specific surface area Mg/Al-CO3 layered double hydroxides. Dalton Trans. 44(37), 16392–16398 (2015b). https://doi.org/10.1039/c5dt02641e
Chen, C.P., Byles, C.F.H., Buffet, J.C., Rees, N.H., Wu, Y., O'Hare, D.: Core-shell zeolite@aqueous miscible organic-layered double hydroxides. Chem. Sci. 7(2), 1457–1461 (2016). https://doi.org/10.1039/c5sc03208c
Chen, S., Yan, Q., Zhang, C., Wang, Q.: A novel highly active and sulfur resistant catalyst from Mn-Fe-Al layered double hydroxide for low temperature NH3-SCR. Catal. Today 327, 81–89 (2019). https://doi.org/10.1016/j.cattod.2018.06.006
Coenen, K., Gallucci, F., Cobden, P., van Dijk, E., Hensen, E., van Sint Annaland, M.: Chemisorption working capacity and kinetics of CO2 and H2O of hydrotalcite-based adsorbents for sorption-enhanced water-gas-shift applications. Chem. Eng. J. 293, 9–23 (2016)
Coenen, K., Gallucci, F., Hensen, E., Sint Annaland, M.: CO2 and H2O chemisorption mechanism on different potassium-promoted sorbents for SEWGS processes. J. CO2 Utiliz. 25, 180–193 (2018a). https://doi.org/10.1016/j.jcou.2018.04.002
Coenen, K., Gallucci, F., Cobden, P., van Dijk, E., Hensen, E., Annaland, M.V.: Influence of material composition on the CO2 and H2O adsorption capacities and kinetics of potassium-promoted sorbents. Chem. Eng. J. 334, 2115–2123 (2018). https://doi.org/10.1016/j.cej.2017.11.161
Du, H., Ebner, A.D., Ritter, J.A.: Temperature dependence of the nonequilibrium kinetic model that describes the adsorption and desorption behavior of CO2 in K-promoted HTlc. Ind. Eng. Chem. Res. 49(7), 3328–3336 (2010). https://doi.org/10.1021/ie901210y
Gao, Y., Zhang, Z., Wu, J., Yi, X., Zheng, A., Umar, A., O'Hare, D., Wang, Q.: Comprehensive investigation of CO2 adsorption on Mg-Al-CO3 LDH-derived mixed mental oxides. J. Mater. Chem. A 1(41), 12782–12790 (2013)
Garcia-Gallastegui, A., Iruretagoyena, D., Gouvea, V., Mokhtar, M., Asiri, A.M., Basahel, S.N., Al-Thabaiti, S.A., Alyoubi, A.O., Chadwick, D., Shaffer, M.S.P.: Graphene oxide as support for layered double hydroxides: enhancing the CO2 adsorption capacity. Chem. Mater. 24(23), 4531–4539 (2012). https://doi.org/10.1021/cm3018264
Hanif, A., Dasgupta, S., Divekar, S., Arya, A., Garg, M.O., Nanoti, A.: A study on high temperature CO2 capture by improved hydrotalcite sorbents. Chem. Eng. J. 236, 91–99 (2014). https://doi.org/10.1016/j.cej.2013.09.076
Hickman, G.E., Wright, C.M.R., Kilpatrick, A.F.R., Turner, Z.R., Buffet, J.-C., O’Hare, D.: Synthesis, characterisation and slurry phase ethylene polymerisation of rac-(PhBBI*)ZrCl2 immobilised on modified layered double hydroxides. Mol. Catal. 468, 139–147 (2019). https://doi.org/10.1016/j.mcat.2019.02.025
Ji, G., Yao, J.G., Clough, P.T., da Costa, J.C.D., Anthony, E.J., Fennell, P.S., Wang, W., Zhao, M.: Enhanced hydrogen production from thermochemical processes. Energy Environ. Sci. 11(10), 2647–2672 (2018). https://doi.org/10.1039/c8ee01393d
Kim, S., Lee, K.B.: Impregnation of hydrotalcite with NaNO3 for enhanced high-temperature CO2 sorption uptake. Chem. Eng. J. 356, 964–972 (2019). https://doi.org/10.1016/j.cej.2018.08.207
Kwok, W.L.J., Crivoi, D.G., Chen, C.P., Buffet, J.C., O'Hare, D.: Silica@layered double hydroxide core-shell hybrid materials. Dalton Trans. 47(1), 143–149 (2018). https://doi.org/10.1039/c7dt03861e
Liu, J., Wei, Y., Zhao, Y.: Trace carbon dioxide capture by metal-organic frameworks. ACS Sustain. Chem. Eng. 7(1), 82–93 (2019). https://doi.org/10.1021/acssuschemeng.8b05590
Marquez, C., Simonov, A., Wharmby, M.T., Van Goethem, C., Vankelecom, I., Bueken, B., Krajnc, A., Mali, G., De Vos, D., De Baerdemaeker, T.: Layered Zn2[Co(CN)6](CH3COO) double metal cyanide: a two-dimensional DMC phase with excellent catalytic performance. Chem. Sci. 10(18), 4868–4875 (2019). https://doi.org/10.1039/C9SC00527G
Mei, X., Yan, Q., Lu, P., Wang, J., Cui, Y., Nie, Y., Umar, A., Wang, Q.: Synthesis of Pt/K2CO3/MgAlOx–reduced graphene oxide hybrids as promising NOx storage–reduction catalysts with superior catalytic performance. Sci. Rep. 7(1), 42862 (2017). https://doi.org/10.1038/srep42862
Platonov, A.Y., Evdokimov, A.N., Kurzin, A.V., Maiyorova, H.D.: Solubility of potassium carbonate and potassium hydrocarbonate in methanol. J. Chem. Eng. Data 47(5), 1175–1176 (2002). https://doi.org/10.1021/je020012v
Rocha, C., Soria, M.A., Madeira, L.M.: Effect of interlayer anion on the CO2 capture capacity of hydrotalcite-based sorbents. Sep. Purif. Technol. 219, 290–302 (2019). https://doi.org/10.1016/j.seppur.2019.03.026
Shang, S., Hanif, A., Sun, M., Tian, Y., Ok, Y.S., Yu, I.K.M., Tsang, D.C.W., Gu, Q., Shang, J.: Novel M (Mg/Ni/Cu)-Al-CO3 layered double hydroxides synthesized by aqueous miscible organic solvent treatment (AMOST) method for CO2 capture. J. Hazard. Mater. 373, 285–293 (2019). https://doi.org/10.1016/j.jhazmat.2019.03.077
Silva, J.M., Trujillano, R., Rives, V., Soria, M.A., Madeira, L.M.: High temperature CO2 sorption over modified hydrotalcites. Chem. Eng. J. 325, 25–34 (2017). https://doi.org/10.1016/j.cej.2017.05.032
Theo, W.L., Lim, J.S., Hashim, H., Mustaffa, A.A., Ho, W.S.: Review of pre-combustion capture and ionic liquid in carbon capture and storage. Appl. Energy 183, 1633–1663 (2016). https://doi.org/10.1016/j.apenergy.2016.09.103
van Dijk, H.A.J., Walspurger, S., Cobden, P.D., van den Brink, R.W., de Vos, F.G.: Testing of hydrotalcite-based sorbents for CO2 and H2S capture for use in sorption enhanced water gas shift. Int. J. Greenhouse Gas Control 5(3), 505–511 (2011). https://doi.org/10.1016/j.ijggc.2010.04.011
Wang, Q., O'Hare, D.: Recent advances in the synthesis and application of layered double hydroxide (LDH) nanosheets. Chem. Rev. 112(7), 4124–4155 (2012). https://doi.org/10.1021/cr200434v
Wang, Q., O'Hare, D.: Large-scale synthesis of highly dispersed layered double hydroxide powders containing delaminated single layer nanosheets. Chem. Commun. 49(56), 6301–6303 (2013). https://doi.org/10.1039/c3cc42918k
Wang, Q., Tay, H.H., Ng, D.J.W., Chen, L., Liu, Y., Chang, J., Zhong, Z., Luo, J., Borgna, A.: The effect of trivalent cations on the performance of Mg-M-CO3 layered double hydroxides for high-temperature CO2 capture. ChemSusChem 3(8), 965–973 (2010). https://doi.org/10.1002/cssc.201000099
Wang, Q., Wu, Z., Tay, H.H., Chen, L., Liu, Y., Chang, J., Zhong, Z., Luo, J., Borgna, A.: High temperature adsorption of CO2 on Mg-Al hydrotalcite: effect of the charge compensating anions and the synthesis pH. Catal. Today 164(1), 198–203 (2011). https://doi.org/10.1016/j.cattod.2010.10.042
Wang, Q., Tay, H.H., Zhong, Z., Luo, J., Borgna, A.: Synthesis of high-temperature CO2 adsorbents from organo-layered double hydroxides with markedly improved CO2 capture capacity. Energy Environ. Sci. 5(6), 7526–7530 (2012). https://doi.org/10.1039/C2EE21409A
Wang, Y., Zhao, L., Otto, A., Robinius, M., Stolten, D.: A review of post-combustion CO2 capture technologies from coal-fired power plants. Energy Procedia 114, 650–665 (2017). https://doi.org/10.1016/j.egypro.2017.03.1209
Wang, J., Yang, Y., Jia, L., Yang, N., Guan, Q., Huang, L., Umar, A., Wang, Q., Ning, P.: The influence of the charge compensating anions of layered double hydroxides (LDHs) in LDH-NS/graphene oxide nanohybrid for CO2 capture. J. Nanosci. Nanotechnol. 18(4), 2956–2964 (2018). https://doi.org/10.1166/jnn.2018.14381
Wang, Z., Xu, S.-M., Xu, Y., Tan, L., Wang, X., Zhao, Y., Duan, H., Song, Y.-F.: Single Ru atoms with precise coordination on a monolayer layered double hydroxide for efficient electrooxidation catalysis. Chem. Sci. 10(2), 378–384 (2019). https://doi.org/10.1039/C8SC04480E
Yang, W.S., Kim, Y., Liu, P.K.T., Sahimi, M., Tsotsis, T.T.: A study by in situ techniques of the thermal evolution of the structure of a Mg-Al-CO3 layered double hydroxide. Chem. Eng. Sci. 57(15), 2945–2953 (2002). https://doi.org/10.1016/s0009-2509(02)00185-9
Yang, Z.-Z., Wei, J.-J., Zeng, G.-M., Zhang, H.-Q., Tan, X.-F., Ma, C., Li, X.-C., Li, Z.-H., Zhang, C.: A review on strategies to LDH-based materials to improve adsorption capacity and photoreduction efficiency for CO2. Coord. Chem. Rev. 386, 154–182 (2019)
Yao, Y., Yu, F., Li, J., Li, J., Li, Y., Wang, Z., Zhu, M., Shi, Y., Dai, B., Guo, X.: Two-dimensional NiAl layered double oxides as non-noble metal catalysts for enhanced CO methanation performance at low temperature. Fuel 255, 115770 (2019). https://doi.org/10.1016/j.fuel.2019.115770
Zhang, C., Gao, Y., Yan, Q., Wang, Q.: Fundamental investigation on layered double hydroxides derived mixed metal oxides for selective catalytic reduction of NOx by H2. Catal. Today (2019). https://doi.org/10.1016/j.cattod.2019.07.006
Zhang, S., Wang, Q., Puthiaraj, P., Ahn, W.-S.: MgFeAl layered double hydroxide prepared from recycled industrial solid wastes for CO2 fixation by cycloaddition to epoxides. J. CO2 Utiliz. 34, 395–403 (2019b). https://doi.org/10.1016/j.jcou.2019.07.035
Zheng, Y., Shi, Y., Li, S., Yang, Y., Cai, N.: Elevated temperature hydrogen/carbon dioxide separation process simulation by integrating elementary reaction model of hydrotalcite adsorbent. Int. J. Hydrogen Energy 39(8), 3771–3779 (2014). https://doi.org/10.1016/j.ijhydene.2013.12.167
Zhu, X., Chen, C., Suo, H., Wang, Q., Shi, Y., O'Hare, D., Cai, N.: Synthesis of elevated temperature CO2 adsorbents from aqueous miscible organic-layered double hydroxides. Energy 167, 960–969 (2019a). https://doi.org/10.1016/j.energy.2018.11.009
Zhu, X., Chen, C., Wang, Q., Shi, Y., O'Hare, D., Cai, N.: Roles for K2CO3 doping on elevated temperature CO2 adsorption of potassium promoted layered double oxides. Chem. Eng. J. 366, 181–191 (2019b). https://doi.org/10.1016/j.cej.2019.01.192
Acknowledgements
This research was financed by the National Key Research Development Program of China (Grant No. 2018YFC0810001) and the National Postdoctoral Program for Innovative Talent (Grant No. BX20190198).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, X., Chen, C., Shi, Y. et al. Aqueous miscible organic-layered double hydroxides with improved CO2 adsorption capacity. Adsorption 26, 1127–1135 (2020). https://doi.org/10.1007/s10450-020-00209-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00209-4