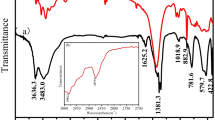
Abstract
Layered lithium–aluminum hydroxides have been used for the lithium adsorption for its high selectivity and cyclic stability. The adsorption performances of lithium and sodium in the mixing solution with abundant magnesium chloride were investigated using batch system at 303 K. The lithium adsorption capacity was increased from 5.02 to 5.69 mg/g (initial lithium concentration is 350 mg/L) with the increasing of initial chloride ions concentration from 200 to 350 mg/L, while the sodium decreased. Both the adsorption capacities of lithium and sodium were lower in Li–Na–MgCl2 system than that’s in Li–MgCl2 and Na–MgCl2 system, which indicated the competitive effects between lithium and sodium ions. In addition, the Langmuir model could describe the isotherms of lithium and sodium well. Then, the homogeneous surface diffusion model (HSDM) was applied to represent the lithium adsorption kinetics. The surface diffusion coefficients of lithium were dependent on the initial concentration and affected by sodium ions. The Biot numbers were ranged from 25 to 40 confirming that the intraparticle diffusion is the limiting step. Breakthrough curves of lithium and sodium demonstrated the lithium could be enriched and obtained with fixed bed adsorption process, and verified the competitive adsorption phenomenon.






Similar content being viewed by others
References
Besserguenev, A.V., Fogg, A.M., Francis, R.J., Price, S.J., O'Hare, D., Tolochko, V.P.I.: Synthesis and structure of the gibbsite intercalation compounds [LiAl2(OH)6]X {X = Cl, Br, NO3} and [LiAl2(OH)6]Cl·H2O using synchrotron X-ray and neutron powder diffraction. Chem. Mater. 9(1), 241–247 (1997)
Chabani, M., Amrane, A., Bensmaili, A.: Kinetic modelling of the adsorption of nitrates by ion exchange resin. Chem. Eng. J. 125(2), 111–117 (2006). https://doi.org/10.1016/j.cej.2006.08.014
Chitrakar, R., Makita, Y., Ooi, K., Sonoda, A.: Synthesis of iron-doped manganese oxides with an ion-sieve property: lithium adsorption from bolivian brine. Ind. Eng. Chem. Res. 53(9), 3682–3688 (2014). https://doi.org/10.1021/ie4043642
Choubey, P.K., Kim, M-s, Srivastava, R.R., Lee, J-c, Lee, J.-Y.: Advance review on the exploitation of the prominent energy-storage element: lithium. Part I: from mineral and brine resources. Miner. Eng. 89, 119–137 (2016). https://doi.org/10.1016/j.mineng.2016.01.010
Choy KKH, McKay G (2005) Sorption of cadmium, copper, and zinc ions onto bone char using Crank diffusion model. Chemosphere 60(8):1141–1150. doi:https://doi.org/10.1016/j.chemosphere.2004.12.041
Choy, K.K.H., Porter, J.F., McKay, G.: Intraparticle diffusion in single and multicomponent acid dye adsorption from wastewater onto carbon. Chem. Eng. J. 103(1–3), 133–145 (2004). https://doi.org/10.1016/j.cej.2004.05.012
Dotto, G.L., Buriol, C., Pinto, L.A.A.: Diffusional mass transfer model for the adsorption of food dyes on chitosan films. Chem. Eng. Res. Des. 92(11), 2324–2332 (2014). https://doi.org/10.1016/j.cherd.2014.03.013
Foo, K.Y., Hameed, B.H.: Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 156(1), 2–10 (2010)
Frohlich, A.C., Ocampo Perez, R., Diaz Blancas, V., Salau, N.P.G., Dotto, G.L.: Three-dimensional mass transfer modeling of ibuprofen adsorption on activated carbon prepared by sonication. Chem. Eng. J. 341, 65–74 (2018). https://doi.org/10.1016/j.cej.2018.02.020
Guo, X., Hu, S., Wang, C., Duan, H., Xiang, X.: Highly efficient separation of magnesium and lithium and high utilization of magnesium from salt lake brine by a reaction-coupled technology. Ind. Eng. Chem. Res. 57(19), 6618–6626 (2018). https://doi.org/10.1021/acs.iecr.8b01147
Hao, H., Liu, Z., Zhao, F., Geng, Y., Sarkis, J.: Material flow analysis of lithium in China. Resour. Policy 51, 100–106 (2017). https://doi.org/10.1016/j.resourpol.2016.12.005
Hong, H.J., Ryu, T., Park, I.S., Kim, M., Shin, J., Kim, B.G., Chung, K.S.: Highly porous and surface-expanded spinel hydrogen manganese oxide (HMO)/Al2O3 composite for effective lithium (Li) recovery from seawater. Chem. Eng. J. 337, 455–461 (2018). https://doi.org/10.1016/j.cej.2017.12.130
Hu, S., Sun, Y., Pu, M., Yun, R., Xiang, X.: Determination of boundary conditions for highly efficient separation of magnesium and lithium from salt lake brine by reaction-coupled separation technology. Sep. Purif. Technol. (2019). https://doi.org/10.1016/j.seppur.2019.115813
Ji, Z.Y., Yang, F.J., Zhao, Y.Y., Liu, J., Wang, N., Yuan, J.S.: Preparation of titanium–base lithium ionic sieve with sodium persulfate as eluent and its performance. Chem. Eng. J. 328, 768–775 (2017). https://doi.org/10.1016/j.cej.2017.07.047
Jiang, H., Yang, Y., Sun, S., Yu, J.: Adsorption of lithium ions on lithium–aluminum hydroxides: equilibrium and kinetics. Can. J. Chem. Eng. (2019). https://doi.org/10.1002/cjce.23640
Kotsupalo, N.P., Ryabtsev, A.D., Poroshina, I.A., Kurakov, A.A., Mamylova, E.V., Menzheres, L.T., Korchagin, M.A.: Effect of structure on the sorption properties of chlorine-containing form of double aluminum lithium hydroxide. Russ. J. Appl. Chem. 86(4), 482–487 (2013). https://doi.org/10.1134/s1070427213040046
Kozlova, S.G., Gabuda, S.P., Isupov, V.P., Chupakhina, L.: Using NMR in structural studies of aluminum hydroxide intercalation compounds with lithium salts. J. Struct. Chem. 44(2), 198–205 (2003)
Lawagon, C.P., Nisola, G.M., Mun, J., Tron, A., Torrejos, R.E.C., Seo, J.G., Kim, H., Chung, W.J.: Adsorptive Li + mining from liquid resources by H2TiO3: equilibrium, kinetics, thermodynamics, and mechanisms. J. Ind. Eng. Chem. 35, 347–356 (2016). https://doi.org/10.1016/j.jiec.2016.01.015
Lin X, Huang Q, Qi G, Xiong L, Huang C, Chen X, Li H, Chen X (2017) Adsorption behavior of levulinic acid onto microporous hyper-cross-linked polymers in aqueous solution: Equilibrium, thermodynamic, kinetic simulation and fixed-bed column studies. Chemosphere 171:231–239. doi:https://doi.org/10.1016/j.chemosphere.2016.12.084
Liu X, Zhong M, Chen X, Zhao Z (2018) Separating lithium and magnesium in brine by aluminum-based materials. Hydrometallurgy 176:73–77. doi:https://doi.org/10.1016/j.hydromet.2018.01.005
Martin, G., Rentsch, L., Höck, M., Bertau, M.: Lithium market research—global supply, future demand and price development. Energy Storage Mater. 6, 171–179 (2017). https://doi.org/10.1016/j.ensm.2016.11.004
Moazeni, M., Hajipour, H., Askari, M., Nusheh, M.: Hydrothermal synthesis and characterization of titanium dioxide nanotubes as novel lithium adsorbents. Mater. Res. Bull. 61, 70–75 (2015). https://doi.org/10.1016/j.materresbull.2014.09.069
Narins, T.P.: The battery business: Lithium availability and the growth of the global electric car industry. Extract. Ind. Soc. 4(2), 321–328 (2017). https://doi.org/10.1016/j.exis.2017.01.013
Ooi K, Sonoda A, Makita Y, Chitrakar R, Tasaki-Handa Y, Nakazato T (2017) Recovery of lithium from salt-brine eluates by direct crystallization as lithium sulfate. Hydrometallurgy 174:123–130. doi:https://doi.org/10.1016/j.hydromet.2017.10.007
Ponnusami, V., Rajan, K.S., Srivastava, S.N.: Application of film-pore diffusion model for methylene blue adsorption onto plant leaf powders. Chem. Eng. J. 163(3), 236–242 (2010). https://doi.org/10.1016/j.cej.2010.07.052
Ramakumar, S., Deviannapoorani, C., Dhivya, L., Shankar, L.S., Murugan, R.: Lithium garnets: synthesis, structure, Li + conductivity, Li + dynamics and applications. Prog. Mater. Sci. 88, 325–411 (2017). https://doi.org/10.1016/j.pmatsci.2017.04.007
Ramesh, C., Yoji, M., Kenta, O., Akinari, S.: Lithium recovery from salt lake brine by H2TiO3. Dalton Trans. 43(23), 8933–8939 (2014)
Ramos Suzaki, P.Y., Munaro, M.T., Triques, C.C., Kleinubing, S.J., Fagundes Klen, M.R., de Matos Jorge, L.M., Bergamasco, R.: Biosorption of binary heavy metal systems: phenomenological mathematical modeling. Chem. Eng. J. 313, 364–373 (2017). https://doi.org/10.1016/j.cej.2016.12.082
Rengaraj, S., Joo, C.K., Kim, Y., Yi, J.: Kinetics of removal of chromium from water and electronic process wastewater by ion exchange resins: 1200H, 1500H and IRN97H. J. Hazard. Mater. 102(2–3), 257–275 (2003). https://doi.org/10.1016/s0304-3894(03)00209-7
Ryu, T., Ryu, J.C., Shin, J., Lee, D.H., Kim, Y.H., Chung, K.-S.: Recovery of lithium by an electrostatic field-assisted desorption process. Ind. Eng. Chem. Res. 52(38), 13738–13742 (2013). https://doi.org/10.1021/ie401977s
Souza, P.R., Dotto, G.L., Salau, N.P.G.: Detailed numerical solution of pore volume and surface diffusion model in adsorption systems. Chem. Eng. Res. Des. 122, 298–307 (2017). https://doi.org/10.1016/j.cherd.2017.04.021
Souza, P.R., Dotto, G.L., Salau, N.P.G.: Statistical evaluation of pore volume and surface diffusion model in adsorption systems. J. Environ. Chem. Eng. 5(6), 5293–5297 (2017). https://doi.org/10.1016/j.jece.2017.10.012
Sun, Y., Guo, X., Hu, S., Xiang, X.: Highly efficient extraction of lithium from salt lake brine by LiAl-layered double hydroxides as lithium-ion-selective capturing material. J. Energy Chem. 34, 80–87 (2019). https://doi.org/10.1016/j.jechem.2018.09.022
Sun, Y., Yun, R., Zang, Y., Pu, M., Xiang, X.: Highly efficient lithium recovery from pre-synthesized chlorine-ion-intercalated LiAl-layered double hydroxides via a mild solution chemistry process. Materials (Basel, Switzerland) (2019). https://doi.org/10.3390/ma12121968
Vasiliu, S., Bunia, I., Racovita, S., Neagu, V.: Adsorption of cefotaxime sodium salt on polymer coated ion exchange resin microparticles: Kinetics, equilibrium and thermodynamic studies. Carbohydr. Polym. 85(2), 376–387 (2011). https://doi.org/10.1016/j.carbpol.2011.02.039
Wang S, Li P, Zhang X, Zheng S, Zhang Y (2017) Selective adsorption of lithium from high Mg-containing brines using HxTiO3 ion sieve. Hydrometallurgy 174:21–28. doi:https://doi.org/10.1016/j.hydromet.2017.09.009
Xiao, J.L., Sun, S.Y., Wang, J., Li, P., Yu, J.G.: Synthesis and adsorption properties of Li1.6Mn1.6O4 spinel. Ind. Eng. Chem. Res. 52(34), 11967–11973 (2013)
Yao, C., Chen, T.: A new simplified method for estimating film mass transfer and surface diffusion coefficients from batch adsorption kinetic data. Chem. Eng. J. 265, 93–99 (2015)
Zhang, Q., Sun, S., Li, S., Jiang, H., Yu, J.: Adsorption of lithium ions on novel nanocrystal MnO2. Chem. Eng. Sci. 62, 4869–4874 (2007). https://doi.org/10.1016/j.ces.2007.01.016
Zhang, Q.H., Li, S.P., Sun, S.Y., Yin, X.S., Yu, J.G.: Lithium selective adsorption on low-dimensional titania nanoribbons. Chem. Eng. Sci. 65(1), 165–168 (2010). https://doi.org/10.1016/j.ces.2009.06.001
Acknowledgement
The research was supported by the National Key R&D Program of China (2017YFB0603104) and NSFC (U1407120, U1707601, U1607112, 21776089, 51574126).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, H., Zhang, S., Yang, Y. et al. Synergic and competitive adsorption of Li–Na–MgCl2 onto lithium–aluminum hydroxides. Adsorption 26, 1039–1049 (2020). https://doi.org/10.1007/s10450-020-00208-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00208-5