Abstract
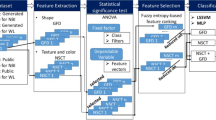
With the goal of enhancing the early diagnosis of colorectal cancer (CRC) polyps and reducing the risk of mortality in cancer patients, in this article, we present a unique diagnosis framework including a Vision-based Surface Tactile Sensor (VS-TS) and complementary Artificial Intelligence algorithms. Leveraging the morphological characteristics (i.e., shape and texture) and stiffness features of the CRC polyps, the proposed framework is able to reliably and sensitively identify their type and stage. To thoroughly characterize and identify the required VS-TS sensitivity for reliable identification of polyps, we first fabricated three different VS-TSs and qualitatively evaluated their performances on 48 different types of polyp phantoms fabricated based on four different types of realistic CRC polyps and three different materials. Next, to quantitatively compare the performance and sensitivity of the fabricated VS-TSs, we used Support Vector Machine (SVM) algorithm and employed various statistical metrics (i.e., accuracy, reliability, and sensitivity). Next, using the most sensitive VS-TS, we classified the type of tumors using the SVM algorithm and applied the t-Distributed Stochastic Neighbor Embedding algorithm to successfully identify the stiffness of classified polyp phantoms solely based on the output images of the VS-TS sensor. Results demonstrated that an SVM algorithm applied on the image outputs of a VS-TS with a Shore hardness of 00–40 scale is able to classify all types of polyps with > 90% accuracy, sensitivity, and reliability. We also repeated experiments on samples of ex-vivo lamb tripe tissues and successfully verified the high sensitivity and reliability of the proposed framework (i.e., > 94%).








Similar content being viewed by others
References
Abdelmoula, W. M., B. Balluff, S. Englert, J. Dijkstra, M. J. Reinders, A. Walch, L. A. McDonnell, and B. P. Lelieveldt. DData-driven identification of prognostic tumor subpopulations using spatially mapped t-SNE of mass spectrometry imaging data. Proc. Natl. Acad. Sci. U.S.A. 113:12244–12249, 2016.
Abdi, H., and L. J. Williams. Principal component analysis. Wiley Interdiscip. Rev.: Comput. Stat. 2:433–459, 2010.
Axon, A., M. Diebold, M. Fujino, R. Fujita, R. Genta, J.-J. Gonvers, M. Guelrud, H. Inoue, M. Jung, H. Kashida, et al. Update on the Paris classification of superficial neoplastic lesions in the digestive tract. Endoscopy 37:570–578, 2005.
Azer, S. A. Challenges facing the detection of colonic polyps: what can deep learning do? Medicina 55:473, 2019.
Bora, K., M. Bhuyan, K. Kasugai, S. Mallik, and Z. Zhao. Computational learning of features for automated colonic polyp classification. Sci. Rep. 11:1–16, 2021.
Chang, C.-C., and C.-J. Lin. LIBSVM: a library for support vector machines. ACM Trans. Intell. Syst. Technol. 2:1–27, 2011.
Dalal, N., and B. Triggs. Histograms of oriented gradients for human detection. In: 2005 IEEE Computer Society Conference on Computer Vision and Pattern Recognition (CVPR’05). IEEE, 2005, vol. 1, pp. 886–893.
Deptuła, P., D. Łysik, K. Pogoda, M. Cieśluk, A. Namiot, J. Mystkowska, G. Król, S. Głuszek, P. A. Janmey, and R. Bucki. Tissue rheology as a possible complementary procedure to advance histological diagnosis of colon cancer. ACS Biomater. Sci. Eng. 6:5620–5631, 2020.
Facciorusso, A., M. Antonino, M. Di Maso, M. Barone, and N. Muscatiello. Non-polypoid colorectal neoplasms: classification, therapy and follow-up. World J. Gastroenterol. 21:5149, 2015.
Heijnsdijk, E., M. Van Der Voort, H. De Visser, J. Dankelman, and D. Gouma. Inter- and intra-individual variabilities of perforation forces of human and pig bowel tissue. Surg. Endosc. Other Interv. Tech. 17:1923–1926, 2003.
Heo, H., Y. Jin, D. Yang, C. Wier, A. Minard, N. B. Dahotre, and A. Neogi. Manufacturing and characterization of hybrid bulk voxelated biomaterials printed by digital anatomy 3D printing. Polymers 13:123, 2020.
Kaltenbach, T., J. C. Anderson, C. A. Burke, J. A. Dominitz, S. Gupta, D. Lieberman, D. J. Robertson, A. Shaukat, S. Syngal, and D. K. Rex. Endoscopic removal of colorectal lesions-recommendations by the us multi-society task force on colorectal cancer. Gastroenterology 158:1095–1129, 2020.
Kara, O. C., N. Ikoma, and F. Alambeigi. HySenSe: a hyper-sensitive and high-fidelity vision-based tactile sensor. In: 2022 IEEE Sensors. IEEE, 2022, pp. 1–4.
Kawano, S., M. Kojima, Y. Higuchi, M. Sugimoto, K. Ikeda, N. Sakuyama, S. Takahashi, R. Hayashi, A. Ochiai, and N. Saito. Assessment of elasticity of colorectal cancer tissue, clinical utility, pathological and phenotypical relevance. Cancer Sci. 106:1232–1239, 2015.
Le, A., M. O. Salifu, and I. M. McFarlane. Artificial intelligence in colorectal polyp detection and characterization. Int. J. Clin. Res. Trials 6:157, 2021.
Lou, G.-C., J.-M. Yang, Q.-S. Xu, W. Huang, and S.-G. Shi. A retrospective study on endoscopic missing diagnosis of colorectal polyp and its related factors. Turk. J. Gastroenterol. 25:182–186, 2014.
Othman, W., and M. A. Qasaimeh. Tactile sensing for minimally invasive surgery: conventional methods and potential emerging tactile technologies. Front. Robot. AI 8:376, 2021.
Siegel, R. L., K. D. Miller, H. E. Fuchs, and A. Jemal. Cancer statistics, 2022. CA Cancer J. Clin. 72:7–33, 2022.
Sung, H., J. Ferlay, R. L. Siegel, M. Laversanne, I. Soerjomataram, A. Jemal, and F. Bray. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71:209–249, 2021.
Van der Maaten, L., and G. Hinton. Visualizing data using t-SNE. J. Mach. Learn. Res. 9:2579–2605, 2008.
Venkatayogi, N., Q. Hu, O. C. Kara, T. G. Mohanraj, S. F. Atashzar, and F. Alambeigi. Pit-pattern classification of colorectal cancer polyps using a hyper sensitive vision-based tactile sensor and dilated residual networks. arXiv preprint. 2022. http://arxiv.org/abs/2211.06814.
Venkatayogi, N., O. C. Kara, J. Bonyun, N. Ikoma, and F. Alambeigi. Classification of colorectal cancer polyps via transfer learning and vision-based tactile sensing. In: 2022 IEEE Sensors. IEEE, 2022, pp. 1–4.
Won, C.-H., J.-H. Lee, and F. Saleheen. Tactile sensing systems for tumor characterization: a review. IEEE Sens. J. 21:12578–12588, 2021.
Xi, Y., and P. Xu. Global colorectal cancer burden in 2020 and projections to 2040. Transl. Oncol. 14:101174, 2021.
Younas, F., M. Usman, and W. Q. Yan. A deep ensemble learning method for colorectal polyp classification with optimized network parameters. Appl. Intell. 53:2410–2433, 2022.
Yuan, W., S. Dong, and E. H. Adelson. GelSight: high-resolution robot tactile sensors for estimating geometry and force. Sensors 17:2762, 2017.
Zanotelli, M. R., and C. A. Reinhart-King. Mechanical forces in tumor angiogenesis. In: Biomechanics in Oncology. Advances in Experimental Medicine and Biology, 2018, vol. 1092, pp. 91-112.
Zhao, S., S. Wang, P. Pan, T. Xia, X. Chang, X. Yang, L. Guo, Q. Meng, F. Yang, W. Qian, et al. Magnitude, risk factors, and factors associated with adenoma miss rate of tandem colonoscopy: a systematic review and meta-analysis. Gastroenterology 156:1661–1674, 2019.
Acknowledgments
Research reported in this publication was supported by the UT Austin/MD Anderson Cancer Center Collaborative Pilot Projects Grants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Additional information
Associate Editor Andreas Anayiotos oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kara, O.C., Venkatayogi, N., Ikoma, N. et al. A Reliable and Sensitive Framework for Simultaneous Type and Stage Detection of Colorectal Cancer Polyps. Ann Biomed Eng 51, 1499–1512 (2023). https://doi.org/10.1007/s10439-023-03153-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-023-03153-w