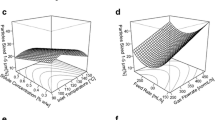
Abstract
Spray drying biologics into a powder can increase thermal stability and shelf-life relative to liquid formulations, potentially eliminating the need for cold chain infrastructure for distribution in developing countries. In this study, process modelling, microparticle engineering, and a supplemented phase diagram were used to design physically stable fully amorphous spray-dried powder capable of stabilizing biological material. A greater proportion of anti-Campylobacter bacteriophage CP30A remained biologically active after spray drying using excipient formulations containing trehalose and a high glass transition temperature amorphous shell former, either trileucine or pullulan, as compared to the commonly used crystalline shell former, leucine, or a low glass transition temperature amorphous shell former, pluronic F-68. Particle formation models suggest that the stabilization was achieved by protecting the bacteriophages against the main inactivating stress, desiccation, at the surface. The most promising formulation contained a combination of trileucine and trehalose for which the combined effects of feedstock preparation, spray drying, and 1-month dry room temperature storage resulted in a titer reduction of only 0.6 ± 0.1 log10(PFU mL−1). The proposed high glass transition temperature amorphous formulation platform may be advantageous for stabilizing biologics in other spray drying applications in the biomedical engineering industry.






Similar content being viewed by others
Notes
A droplet emitted from the atomizer is assumed to initially be well-mixed in terms of the excipients being evenly distributed within the droplet, while the phages are assumed to be randomly distributed. “A”: trileucine is surface-active and near saturation initially and forms an amorphous shell with small amounts of trehalose early, before all phages are present at the surface. The mainly trileucine shell folds, potentially since it is thin and rubbery; the mainly trehalose interior has yet to solidify. The shell potentially maintains the same surface area upon folding and prevents phages from reaching the surface. “B”: the crumpled appearance is controlled by the folded shell. The interior later solidifies into a glass from a highly viscous solution due to further desiccation. “C”: pullulan enriches at the surface due to high molecular mass and forms a viscous amorphous shell that contains small amounts of trehalose; the shell contracts, causing phages to recede with the surface, and desiccates until a glassy mixture of pullulan and trehalose is present at the surface. “D”: the surface is mainly pullulan, with some trehalose that may have prevented shell deformation. The interior is mainly trehalose with some pullulan. “E”: nucleation at the surface results in small leucine crystals that eventually become close enough and large enough to form a shell. Trehalose and remaining leucine eventually solidify. “F”: phages could be expelled from crystals, be damaged by inter-crystal forces, or inhibit crystallization. Small crystals are at the surface of the microparticles. The interior is primarily trehalose and may contain voids. “G”: pullulan enriches near the surface and solidifies there. The thin shell is moderately rigid due to a high glass transition temperature and may easily deform and contract as it is not hindered by a trehalose interior. “H”: there is no trehalose glass stabilizer or void space in the interior. “I”: surfactant may form a film that recedes but does not adequately stabilize the phages. “J”: small cohesive spherical microparticle with phages near or on the surface where they are not adequately stabilized by the low glass transition temperature surfactant. “K”: small cohesive solid spherical microparticle formed. There is a high chance that phages reside on the surface. “L”: same as “K” except that a very small microparticle results and there is a very high chance that phages reside on surface. Void space is not shown in the schematic and may be possible in many cases. Note that there is likely a radial distribution of each excipient within the drying droplet rather than complete separation of excipients.
References
Anandharamakrishnan, C., and S. Padma Ishwarya. Spray Drying Techniques for Food Ingredient Encapsulation. Chichester: Wiley, 2015.
Broadhead, J., S. K. Edmond-Rouan, and C. T. Rhodes. The spray drying of pharmaceuticals. Drug. Dev. Ind. Pharm. 18:1169–1206, 1992.
Carrigy, N. B., L. Liang, H. Wang, S. Kariuki, T. E. Nagel, I. F. Connerton, and R. Vehring. Spray-dried anti-Campylobacter bacteriophage CP30A powder suitable for global distribution without cold chain infrastructure. Int. J. Pharm. 569:118601, 2019.
Carrigy, N. B., M. Ordoubadi, Y. Liu, O. Melhem, D. Barona, H. Wang, L. Milburn, C. A. Ruzycki, W. H. Finlay, and R. Vehring. Amorphous pullulan trehalose microparticle platform for respiratory delivery. Int. J. Pharm. 563:156–168, 2019.
Carrigy, N. B., and R. Vehring. Engineering stable spray-dried biologic powder for inhalation. In: Pharmaceutical Inhalation Aerosol Technology (3rd ed.), edited by A. J. Hickey and S. da Rocha. Boca Raton: CRC Press, 2019, pp. 291–326.
Chen, T., A. Fowler, and M. Toner. Literature review: supplemented phase diagram of the trehalose-water binary mixture. Cryobiology 40:277–282, 2000.
di Stefano, F. Pullulan as release enhancer for controlled release capsular device: performance assessment and preparation methods. M.Sc. Thesis, Department of Chemistry, Materials and Chemical Engineering, Politecnico di Milano, Milan, Italy. 2017.
Feng, A. L., M. A. Boraey, M. A. Gwin, P. R. Finlay, P. J. Kuehl, and R. Vehring. Mechanistic models facilitate efficient development of leucine containing microparticles for pulmonary drug delivery. Int. J. Pharm. 409:156–163, 2011.
Grasmeijer, N., H. W. Frijlink, and W. L. J. Hinrichs. Model to predict inhomogeneous protein-sugar distribution in powders prepared by spray drying. J. Aerosol Sci. 101:22–33, 2016.
Hancock, B. C., S. L. Shamblin, and G. Zografi. Molecular mobility of amorphous pharmaceutical solids below their glass transition temperatures. Pharm. Res. 12:799–806, 1995.
Hasegawa, T., J. Umemura, and N. Yamada. Characterization of thin cast films of a trileucine-induced lipid by infrared multiple-angle incidence resolution spectrometry. J Mol. Struct. 735–736:63–67, 2005.
He, X., L. Pei, H. H. Y. Tong, and Y. Zheng. Comparison of spray freeze drying and the solvent evaporation method for preparing solid dispersions of baicalein with pluronic F68 to improve dissolution and oral bioavailability. AAPS PharmSciTech. 12:104–113, 2011.
Hoe, S., J. W. Ivey, M. A. Boraey, A. Shamsaddini-Shahrbabek, E. Javaheri, S. Matinkhoo, W. H. Finlay, and R. Vehring. Use of a fundamental approach to spray-drying formulation design to facilitate the development of multi-component dry powder aerosols for respiratory drug delivery. Pharm. Res. 31:449–465, 2014.
Hoe, S., D. D. Semler, A. D. Goudie, K. H. Lynch, S. Matinkhoo, W. H. Finlay, J. J. Dennis, and R. Vehring. Respirable bacteriophages for the treatment of bacterial lung infections. J. Aerosol Med. Pulm. Drug Deliv. 26:317–335, 2013.
Lechuga-Ballesteros, D., C. Charan, C. L. M. Stults, C. L. Stevenson, D. P. Miller, R. Vehring, V. Tep, and M. C. Kuo. Trileucine improves aerosol performance and stability of spray-dried powders for inhalation. J. Pharm. Sci. 97:287–302, 2008.
Leung, V., A. Szewczyk, J. Chau, Z. Hosseinidoust, L. Groves, H. Hawsawi, H. Anany, M. W. Griffiths, M. M. Ali, and C. D. M. Filipe. Long-term preservation of bacteriophage antimicrobials using sugar glasses. ACS Biomater. Sci. Eng. 4:3802–3808, 2018.
Masters, K. Spray Drying: An Introduction to Principles, Operational Practice and Applications. London: Leonard Hill Books, 1972.
Nishinari, K., K. Kohyama, P. A. Williams, G. O. Phillips, W. Burchard, and K. Ogino. Solution properties of pullulan. Macromolecules 24:5590–5593, 1991.
O’Reilly, C. E., P. Jaron, B. Ochieng, A. Nyaguara, J. E. Tate, M. B. Parsons, C. A. Bopp, K. A. Williams, J. Vinjé, E. Blanton, K. A. Wannemuehler, J. Vulule, K. F. Laserson, R. F. Breiman, D. R. Feikin, M. A. Widdowson, and E. Mintz. Risk factors for death among children less than 5 years old hospitalized with diarrhea in rural western Kenya, 2005–2007: a cohert study. PLoS Med. 9:e1001256, 2012.
Roe, K. D., and T. P. Labuza. Glass transition and crystallization of amorphous trehalose-sucrose mixtures. Int. J. Food Prop. 8:559–574, 2005.
Rumble, J. R. CRC Handbook of Chemistry and Physics (98th ed.). Boca Raton: CRC Press, 2018.
Schwartzbach, H. Achieving aseptic drying with spray drying technologies. Pharm. Technol. Eur. 23:90–92, 2011.
Siew, A. Exploring the use of aseptic spray drying in the manufacture of biopharmaceutical injectables. Pharm. Technol. 40:24–27, 2016.
Singh, R. S., and G. K. Saini. Biosynthesis of pullulan and its applications in food and pharmaceutical industry. In: Microorganisms in Sustainable Agriculture and Biotechnology, edited by T. Satyanarayana, B. N. Johri, and A. Prakash. Dordrecht: Springer, 2012, pp. 509–553.
Teekamp, N., Y. Tian, J. C. Visser, P. Olinga, H. W. Frijlink, H. J. Woerdenbag, and W. L. J. Hinrichs. Addition of pullulan to trehalose glasses improves the stability of β-galactosidase at high moisture contents. Carbohydr. Polym. 176:374–380, 2017.
Vandenheuvel, D., J. Meeus, R. Lavigne, and G. van den Mooter. Instability of bacteriophage in spray-dried trehalose powders is caused by crystallization of the matrix. Int. J. Pharm. 472:202–205, 2014.
Vehring, R. Pharmaceutical particle engineering via spray drying. Pharm. Res. 25:999–1022, 2008.
Vehring, R., W. R. Foss, and D. Lechuga-Ballesteros. Particle formation in spray drying. J. Aerosol Sci. 38:728–746, 2007.
Walsh, G. Biopharmaceutical benchmarks 2014. Nat. Biotechnol. 32:992–1000, 2014.
Walters, R. H., B. Bhatnagar, S. Tchessalov, K. I. Izutsu, K. Tsumoto, and S. Ohtake. Next generation drying technologies for pharmaceutical applications. J. Pharm. Sci. 103:2673–2695, 2014.
Wang, H., M. A. Boraey, L. Williams, D. Lechuga-Ballesteros, and R. Vehring. Low-frequency shift dispersive Raman spectroscopy for analysis of respirable dosage forms. Int. J. Pharm. 469:197–205, 2014.
Yamada, N., T. Komatsu, H. Yoshinaga, K. Yoshizawa, S. Edo, and M. Kunitake. Self-supporting elastic film without covalent linkages as a hierarchically integrated β-sheet assembly. Angew. Chem. Int. Ed. 42:5496–5499, 2003.
Acknowledgments
NC thanks the Killam Trusts, the Natural Sciences and Engineering Research Council of Canada, Alberta Innovates, and the University of Alberta for scholarship funding. This work was financially supported by the Biotechnology and Biological Sciences Research Council [Grant Number BB/P02355X/1] (United Kingdom). The funding source had no role in study design, collection, analysis, or interpretation of data, writing the article, or in decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Emmanuel Opara oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Carrigy, N.B., Liang, L., Wang, H. et al. Trileucine and Pullulan Improve Anti-Campylobacter Bacteriophage Stability in Engineered Spray-Dried Microparticles. Ann Biomed Eng 48, 1169–1180 (2020). https://doi.org/10.1007/s10439-019-02435-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02435-6