Abstract
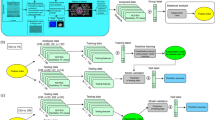
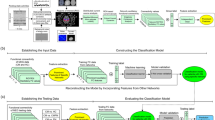
Advanced analyses of electroencephalography (EEG) are rapidly becoming an important tool in understanding the brain’s processing of pain. To date, it appears that none have been explored as a way of distinguishing between migraine patients with aura (MWA) vs. those without aura (MWoA). In this work, we apply a mixture of predictive, e.g., classification methods and attribute-selection techniques, and traditional explanatory, e.g., statistical, analyses on functional connectivity measures extracted from EEG signal acquired from at-rest participants (N = 52) during their interictal period and tested them against the distinction between MWA and MWoA. We show that a functional connectivity metric of EEG data obtained during resting state can serve as a sole biomarker to differentiate between MWA and MWoA. Using the proposed analysis, we not only have been able to present high classification results (average classification of 84.62%) but also to discuss the underlying neurophysiological mechanisms upon which our technique is based. Additionally, a more traditional statistical analysis on the selected features reveals that MWoA patients show higher than average connectivity in the Theta band (p = 0.03) at rest than MWAs. We propose that our data-driven analysis pipeline can be used for resting-EEG analysis in any clinical context.




Similar content being viewed by others
Notes
There are 262 possible connections, out of which “self-loops” need to be removed (26 possible loops) and only half of possible connections need to be selected, thus the number need to be divided by 2 (this due to the fact that the measured connectivity is undirected).
For the Bonferroni correction, we assumed the most stringent case, i.e. the tested hypotheses are independent, and thus for the required confidence interval of α = 5% and the number of tested hypotheses n = 5 the corrected confidence value would be α/n resulting in 1% (i.e. the required corrected p value should be p ≤ 0.01).
References
Bellman, R. E. Adaptive Control Processes: A Guided Tour. Princeton: Princeton University Press, 1961.
Brighina, F., G. Cosentino, and B. Fierro. Is lack of habituation a biomarker of migraine? A critical perspective. J. Headache Pain 16(S1):A13, 2015.
Buono, V. L., et al. Functional connectivity and cognitive impairment in migraine with and without aura. J. Headache Pain 18(1):72, 2017.
Carter, G., C. Knapp, and A. Nuttall. Estimation of the magnitude-squared coherence function via overlapped fast Fourier transform processing. IEEE Trans. Audio Electroacoust. 21(4):337–344, 1973.
Celka, P. Statistical analysis of the phase-locking value. IEEE Signal Process. Lett. 14(9):577–580, 2007.
Chang, C.-C., and C.-J. Lin. LIBSVM: a library for support vector machines. ACM Trans. Intell. Syst. Technol. 2(3):27, 2011.
Charles, A., and J. M. Hansen. Migraine aura: new ideas about cause, classification, and clinical significance. Curr. Opin. Neurol. 28(3):255–260, 2015.
Cucchiara, B., R. Datta, G. K. Aguirre, K. E. Idoko, and J. Detre. Measurement of visual sensitivity in migraine: validation of two scales and correlation with visual cortex activation. Cephalalgia 35(7):585–592, 2015.
Damoiseaux, J. S., et al. Consistent resting-state networks across healthy subjects. PNAS 103(37):13848–13853, 2006.
Datta, R., G. K. Aguirre, S. Hu, J. A. Detre, and B. Cucchiara. Interictal cortical hyperresponsiveness in migraine is directly related to the presence of aura. Cephalalgia 33(6):365–374, 2013.
de Tommaso, M., S. Stramaglia, D. Marinazzo, G. Trotta, and M. Pellicoro. Functional and effective connectivity in EEG alpha and beta bands during intermittent flash stimulation in migraine with and without aura. Cephalalgia 33(11):938–947, 2013.
de Tommaso, M., G. Trotta, E. Vecchio, K. Ricci, R. Siugzdaite, and S. Stramaglia. Brain networking analysis in migraine with and without aura. J. Headache Pain 18(1):98, 2017.
Frid, A. Differences in phase synchrony of brain regions between regular and dyslexic readers. In: 2014 IEEE 28th Convention of Electrical Electronics Engineers in Israel (IEEEI), 2014, pp. 1–4.
Frid, A., and Z. Breznitz. An SVM based algorithm for analysis and discrimination of dyslexic readers from regular readers using ERPs. In: 2012 IEEE 27th Convention of Electrical Electronics Engineers in Israel (IEEEI), 2012, pp. 1–4.
Frid, A., and L. M. Manevitz. Analyzing Cognitive Processes from Complex Neuro-physiologically Based Data. In: AMAI, 2019.
Granovsky, Y., M. Shor, A. Shifrin, E. Sprecher, D. Yarnitsky, and T. Bar-Shalita. Assessment of responsiveness to everyday non-noxious stimuli in pain-free migraineurs with versus without aura. J. Pain 19(8):943–951, 2018.
Hesse, W., E. Möller, M. Arnold, and B. Schack. The use of time-variant EEG Granger causality for inspecting directed interdependencies of neural assemblies. J. Neurosci. Methods 124(1):27–44, 2003.
Hougaard, A., F. M. Amin, S. Magon, T. Sprenger, E. Rostrup, and M. Ashina. No abnormalities of intrinsic brain connectivity in the interictal phase of migraine with aura. Eur. J. Neurol. 22(4):702-e46, 2015.
Kay, S. M. Modern Spectral Estimation: Theory and Application/Book and Disk. Upper Saddle River: PTR Prentice Hall, 1988.
Kira, K., and L. A. Rendell. A practical approach to feature selection. In: Proceedings of the Ninth International Workshop on Machine Learning, San Francisco, CA, USA, 1992, pp. 249–256.
Lauritzen, M. Pathophysiology of the migraine aura. The spreading depression theory. Brain 117(Pt 1):199–210, 1994.
LeCun, Y., Y. Bengio, and G. Hinton. Deep learning. Nature 521(7553):436–444, 2015.
Lev, R., Y. Granovsky, and D. Yarnitsky. Enhanced pain expectation in migraine: EEG-based evidence for impaired prefrontal function. Headache 53(7):1054–1070, 2013.
Mendonça-de-Souza, M., et al. Resilience in migraine brains: decrease of coherence after photic stimulation. Front. Hum. Neurosci. 6:207, 2012.
Nawa, N. E., and H. Ando. Classification of self-driven mental tasks from whole-brain activity patterns. PLoS ONE 9(5):e97296, 2014.
Rabiner, L. R., and B. Gold. Theory and Application of Digital Signal Processing, F First (Edition ed.). Englewood Cliffs, NJ: Prentice Hall, 1975.
Raichle, M. E., A. M. MacLeod, A. Z. Snyder, W. J. Powers, D. A. Gusnard, and G. L. Shulman. A default mode of brain function. Proc. Natl. Acad. Sci. 98(2):676–682, 2001.
Russell, M. B., and J. Olesen. A nosographic analysis of the migraine aura in a general population. Brain 119(Pt 2):355–361, 1996.
Sand, T., N. Zhitniy, L. R. White, and L. J. Stovner. Visual evoked potential latency, amplitude and habituation in migraine: a longitudinal study. Clin. Neurophysiol. 119(5):1020–1027, 2008.
Tfelt-Hansen, P. C. History of migraine with aura and cortical spreading depression from 1941 and onwards. Cephalalgia 30(7):780–792, 2010.
Welch, P. The use of fast Fourier transform for the estimation of power spectra: a method based on time averaging over short, modified periodograms. IEEE Trans. Audio Electroacoust. 15(2):70–73, 1967.
Wilkins, L. W. Visual cortex hyperexcitability in migraine in response to sound-induced flash illusions. Neurology 86(12):1172, 2016.
Acknowledgments
We thank the Migraine Research Foundation, USA, for supporting the research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Michael Gower oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Frid, A., Shor, M., Shifrin, A. et al. A Biomarker for Discriminating Between Migraine With and Without Aura: Machine Learning on Functional Connectivity on Resting-State EEGs. Ann Biomed Eng 48, 403–412 (2020). https://doi.org/10.1007/s10439-019-02357-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02357-3