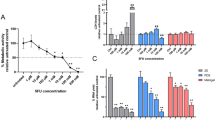
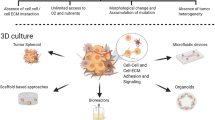
Abstract
Colorectal cancer is subject to a high rate of mutations, with late stage tumors often containing many mutations. These tumors are difficult to treat, and even with the recently implemented methods of personalized medicine at modern hospitals aiming to narrow treatments, a gap still exists. Proper modeling of these tumors may help to recommend optimal treatments for individual patients, preferably utilizing a model that maintains proper signaling in respect to the derived parent tissue. In this study, we utilized an extracellular matrix-derived hydrogel to create a 3D micro-tumor construct platform capable of both supporting cells for long time durations and for high throughput drug screening. Experiments with cell lines demonstrated long-term viability with maintenance of cell proliferation. Furthermore, studies with several chemotherapeutics utilizing different mechanisms of action displayed differences in efficacy in comparing 3D and 2D cultures. Finally, patient colorectal tumor tissue was acquired and employed to reconstruct micro-tumor constructs, providing a system for the testing of novel chemotherapeutics against tumors in a patient-specific manner. Collectively, the results describe a system capable of high throughput testing while maintaining important characteristics of the parent tissue.





Similar content being viewed by others
Abbreviations
- 2D:
-
Two dimensional
- 3D:
-
Three dimensional
- 5-FU:
-
5-Fluorouracil
- CK18:
-
Cytokeratin 18
- CRC:
-
Colorectal cancer
- DMEM:
-
Dulbecco’s minimum essential medium
- DMSO:
-
Dimethyl sulfoxide
- ECM:
-
Extracellular matrix
- EGFR:
-
Epidermal growth factor receptor
- FBS:
-
Fetal bovine serum
- HA:
-
Hyaluronic acid
- IHC:
-
Immunohistochemistry
- IRB:
-
Institutional Review Board
- Ki67:
-
Antigen Ki-67, a biomarker for proliferation
- µTCs:
-
Micro-tumor constructs
- PDMS:
-
Polydimethylsiloxane
- PEGDA:
-
Polyethylene glycol diacrylate
- RNA:
-
Ribonucleic acid
References
Ahmed, D., P. W. Eide, I. A. Eilertsen, S. A. Danielsen, M. Eknaes, M. Hektoen, G. E. Lind, and R. A. Lothe. Epigenetic and genetic features of 24 colon cancer cell lines. Oncogenesis 2:e71, 2013.
Ahronian, L. G., E. M. Sennott, E. M. Van Allen, N. Wagle, E. L. Kwak, J. E. Faris, J. T. Godfrey, K. Nishimura, K. D. Lynch, C. H. Mermel, E. L. Lockerman, A. Kalsy, J. M. Gurski, Jr, S. Bahl, K. Anderka, L. M. Green, N. J. Lennon, T. G. Huynh, M. Mino-Kenudson, G. Getz, D. Dias-Santagata, A. J. Iafrate, J. A. Engelman, L. A. Garraway, and R. B. Corcoran. Clinical acquired resistance to RAF inhibitor combinations in BRAF-mutant colorectal cancer through MAPK pathway alterations. Cancer Discov. 5:358–367, 2015.
Armaghany, T., J. D. Wilson, Q. Chu, and G. Mills. Genetic alterations in colorectal cancer. Gastrointest. Cancer Res. 5:19–27, 2012.
Berg, K. C. G., P. W. Eide, I. A. Eilertsen, B. Johannessen, J. Bruun, S. A. Danielsen, M. Bjornslett, L. A. Meza-Zepeda, M. Eknaes, G. E. Lind, O. Myklebost, R. I. Skotheim, A. Sveen, and R. A. Lothe. Multi-omics of 34 colorectal cancer cell lines—a resource for biomedical studies. Mol. Cancer 16:116, 2017.
Breslin, S., and L. O’Driscoll. The relevance of using 3D cell cultures, in addition to 2D monolayer cultures, when evaluating breast cancer drug sensitivity and resistance. Oncotarget 7:45745–45756, 2016.
Chin, L., J. N. Andersen, and P. A. Futreal. Cancer genomics: from discovery science to personalized medicine. Nat. Med. 17:297–303, 2011.
de Gramont, A. F. A., M. Seymour, M. Homerin, A. Hmissi, J. Cassidy, C. Boni, H. Cortes-Funes, A. Cervantes, G. Freyer, D. Papamichael, N. Le Bail, C. Louvet, D. Hendler, F. de Braud, C. Wilson, F. Morvan, and A. Bonetti. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J. Clin. Oncol. 18:2938–2947, 2000.
De Rosa, M., U. Pace, D. Rega, V. Costabile, F. Duraturo, P. Izzo, and P. Delrio. Genetics, diagnosis and management of colorectal cancer (Review). Oncol. Rep. 34:1087–1096, 2015.
Edmondson, R., J. J. Broglie, A. F. Adcock, and L. Yang. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay Drug Dev. Technol. 12:207–218, 2014.
Fleming, M., S. Ravula, S. F. Tatishchev, and H. L. Wang. Colorectal carcinoma: pathologic aspects. J. Gastrointest. Oncol. 3:153–173, 2012.
Gilbert, C. A., and A. H. Ross. Cancer stem cells: cell culture, markers, and targets for new therapies. J. Cell. Biochem. 108:1031–1038, 2009.
Gmeiner, W. H., W. Debinski, C. Milligan, D. Caudell, and T. S. Pardee. The applications of the novel polymeric fluoropyrimidine F10 in cancer treatment: current evidence. Future Oncol. 12:2009–2020, 2016.
Greystoke, A., M. Ayub, D. G. Rothwell, D. Morris, D. Burt, C. L. Hodgkinson, C. J. Morrow, N. Smith, K. Aung, J. Valle, L. Carter, F. Blackhall, C. Dive, and G. Brady. Development of a circulating miRNA assay to monitor tumor burden: from mouse to man. Mol. Oncol. 10:282–291, 2016.
Hamburg, M. A., and F. S. Collins. The path to personalized medicine. N. Engl. J. Med. 363:301–304, 2010.
Hu, T., Z. Li, C. Y. Gao, and C. H. Cho. Mechanisms of drug resistance in colon cancer and its therapeutic strategies. World J. Gastroenterol. 22:6876–6889, 2016.
Institute N. S. Cancer Stat Facts: Colon and Rectum Cancer. Bethesda: NIH, 2017.
Kapalczynska, M., T. Kolenda, W. Przybyla, M. Zajaczkowska, A. Teresiak, V. Filas, M. Ibbs, R. Blizniak, L. Luczewski, and K. Lamperska. 2D and 3D cell cultures—a comparison of different types of cancer cell cultures. Arch. Med. Sci. 14:910–919, 2018.
Karlsson, H., M. Fryknas, R. Larsson, and P. Nygren. Loss of cancer drug activity in colon cancer HCT-116 cells during spheroid formation in a new 3-D spheroid cell culture system. Exp. Cell Res. 318:1577–1585, 2012.
Katt, M. E., A. L. Placone, A. D. Wong, Z. S. Xu, and P. C. Searson. In Vitro tumor models: advantages, disadvantages, variables, and selecting the right platform. Front. Bioeng. Biotechnol. 4:12, 2016.
Langhans, S. A. Three-dimensional in vitro cell culture models in drug discovery and drug repositioning. Front. Pharmacol. 9:6, 2018.
Loessner, D., K. S. Stok, M. P. Lutolf, D. W. Hutmacher, J. A. Clements, and S. C. Rizzi. Bioengineered 3D platform to explore cell-ECM interactions and drug resistance of epithelial ovarian cancer cells. Biomaterials 31:8494–8506, 2010.
Luca, A. C., S. Mersch, R. Deenen, S. Schmidt, I. Messner, K. L. Schafer, S. E. Baldus, W. Huckenbeck, R. P. Piekorz, W. T. Knoefel, A. Krieg, and N. H. Stoecklein. Impact of the 3D microenvironment on phenotype, gene expression, and EGFR inhibition of colorectal cancer cell lines. PLoS ONE 8:e59689, 2013.
Mazzocchi, A. R., S. A. P. Rajan, K. I. Votanopoulos, A. R. Hall, and A. Skardal. In vitro patient-derived 3D mesothelioma tumor organoids facilitate patient-centric therapeutic screening. Sci. Rep. 8:2886, 2018.
Mazzocchi, A. R., S. Soker, and A. Skardal. Biofabrication technologies for developing in vitro tumor models. In: Tumor Organoids, edited by S. Soker, and A. Skardal. Berlin, Germany: Springer Nature, 2017, pp. 51–70.
Miserocchi, G., L. Mercatali, C. Liverani, A. De Vita, C. Spadazzi, F. Pieri, A. Bongiovanni, F. Recine, D. Amadori, and T. Ibrahim. Management and potentialities of primary cancer cultures in preclinical and translational studies. J. Transl. Med. 15:229, 2017.
Parasuraman, S. Toxicological screening. J. Pharmacol. Pharmacother. 2:74–79, 2011.
Riedl, A., M. Schlederer, K. Pudelko, M. Stadler, S. Walter, D. Unterleuthner, C. Unger, N. Kramer, M. Hengstschlager, L. Kenner, D. Pfeiffer, G. Krupitza, and H. Dolznig. Comparison of cancer cells in 2D vs 3D culture reveals differences in AKT-mTOR-S6K signaling and drug responses. J. Cell Sci. 130:203–218, 2017.
Sameer, A. S. Colorectal cancer: molecular mutations and polymorphisms. Front. Oncol. 3:114, 2013.
Scholzen, T., and J. Gerdes. The Ki-67 protein: from the known and the unknown. J. Cell. Physiol. 182:311–322, 2000.
Siegel, R. L., K. D. Miller, S. A. Fedewa, D. J. Ahnen, R. G. S. Meester, A. Barzi, and A. Jemal. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 67:177–193, 2017.
Skardal, A. Biopolymers for in vitro tissue model biofabrication. In: Biopolymers for Medical Applications, edited by J. M. Ruso, and P. V. Messina. Boca Raton, FL: CRC Press, 2016.
Skardal, A., M. Devarasetty, S. Forsythe, A. Atala, and S. Soker. A reductionist metastasis-on-a-chip platform for in vitro tumor progression modeling and drug screening. Biotechnol. Bioeng. 113:2020–2032, 2016.
Skardal, A., M. Devarasetty, C. Rodman, A. Atala, and S. Soker. Liver-tumor hybrid organoids for modeling tumor growth and drug response in vitro. Ann. Biomed. Eng. 43:2361–2373, 2015.
Skardal, A., L. Smith, S. Bharadwaj, A. Atala, S. Soker, and Y. Zhang. Tissue specific synthetic ECM hydrogels for 3-D in vitro maintenance of hepatocyte function. Biomaterials 33:4565–4575, 2012.
Votanopoulos, K. I., A. Mazzocchi, H. Sivakumar, S. Forsythe, J. Aleman, E. A. Levine, and A. Skardal. Appendiceal cancer patient-specific tumor organoid model for predicting chemotherapy efficacy prior to initiation of treatment: a feasibility study. Ann. Surg. Oncol. 26:139–147, 2019.
Acknowledgments
We wish to thank Libby McWilliams (Procurement Manager), Kathleen Cummings (Protocol and Data Manager) and the Wake Forest Advanced Tumor Bank Shared Resource. AS acknowledges funding through the Wake Forest Clinical and Translational Science Institute Open Pilot Program, supported by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health, through Grant Award Number UL1TR001420. AS and KV acknowledge funding through the Comprehensive Cancer Center at Wake Forest Baptist Medical Center’s Clinical Research Associate Director Pilot Funds, and services from the Tumor Tissue and Pathology Shared Resource supported by the Comprehensive Cancer Center at Wake Forest Baptist Medical Center’s NCI Cancer Center Support Grant P30CA012197.
Conflict of interest
Dr. Skardal is an inventor on several patents associated with this work related to the generation of patient-derived tumor models for drug screening and personalized medicine.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Jane Grande-Allen oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Forsythe, S., Mehta, N., Devarasetty, M. et al. Development of a Colorectal Cancer 3D Micro-tumor Construct Platform From Cell Lines and Patient Tumor Biospecimens for Standard-of-Care and Experimental Drug Screening. Ann Biomed Eng 48, 940–952 (2020). https://doi.org/10.1007/s10439-019-02269-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02269-2