Abstract
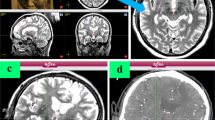
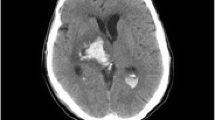
Deep brain stimulation (DBS) involves the implantation of electrodes into specific central brain structures for the treatment of Parkinson’s disease. Image guidance and robot-assisted techniques have been developed to assist in the accuracy of electrode placement. Traditional DBS is performed with the patient awake and utilizes microelectrode recording for feedback, which yields lengthy operating room times. Asleep DBS procedures use imaging techniques to verify electrode placement. The objective of this study is to demonstrate the validity of an asleep robot-assisted DBS procedure that utilizes intraoperative imaging techniques for precise electrode placement in a large, inclusive cohort. Preoperative magnetic resonance imaging (MRI) was used to plan the surgical procedure for the 128 patients that underwent asleep DBS. During the surgery, robot assistance was used during the implantation of the electrodes. To verify electrode placement, intraoperative CT scans were fused with the preoperative MRIs. The mean radial error of all final electrode placements is 0.85 ± 0.38 mm. MRI-CT fusion error is 0.64 ± 0.40 mm. The average operating room time for bilateral and unilateral implantations are 139.3 ± 34.7 and 115.4 ± 42.1 min, respectively. This study shows the validity of the presented asleep DBS procedure using robot assistance and intraoperative CT verification for accurate electrode placement with shorter operating room times.








Similar content being viewed by others
References
Abosch, A., L. Timmermann, S. Bartley, H. G. Rietkerk, D. Whiting, P. J. Connolly, D. Lanctin, and M. I. Hariz. An international survey of deep brain stimulation procedural steps. Stereotact. Funct. Neurosurg. 91:1–11, 2013.
American College of Radiology. MR accreditation program testing instructions. Revised 2018. https://www.acraccreditation.org/~/media/ACRAccreditation/Documents/MRI/MRAccreditationTestingInstructions.pdf
Barrett, J. F., and N. Keat. Artifacts in CT: recognition and avoidance. Radiographics. 24:1679–1691, 2012.
Bjerknes, S., I. M. Skogseid, T. Saehle, E. Dietrichs, and M. Toft. Surgical site infections after deep brain stimulation surgery: frequency, characteristics and management in a 10-year period. PLoS ONE 9:e105288, 2014.
Burchiel, K. J., S. McCartney, A. Lee, and A. M. Raslan. Accuracy of deep brain stimulation electrode placement using intraoperative computed tomography without microelectrode recording. J. Neurosurg. 119:301–306, 2013.
Chen, T., Z. Mirzadeh, K. M. Chapple, M. Lambert, H. A. Shill, G. Moguel-Cobos, A. I. Tröster, R. Dhall, and F. A. Ponce. Clinical outcomes following awake and asleep deep brain stimulation for Parkinson disease. J. Neurosurg. 2018. https://doi.org/10.3171/2017.8.JNS17883.
Faria, C., W. Erlhagen, M. Rito, E. de Momi, G. Ferrigno, and E. Bicho. Review of robotic technology for stereotactic neurosurgery. IEEE Rev. Biomed. Eng. 8:125–137, 2015.
Foltynie, T., L. Zrinzo, I. Martinez-Torres, E. Tripoliti, E. Petersen, E. Holl, I. Aviles-Olmos, M. Jahanshahi, M. Hariz, and P. Limousin. MRI-guided STN DBS in Parkinson’s disease without microelectrode recording: efficacy and safety. J. Neurol. Neurosurg. Psychiatry. 82:358–363, 2011.
Geevarghese, R., R. O’Gorman Tuura, D. E. Lumsden, M. Samuel, and K. Ashkan. Registration accuracy of CT/MRI fusion for localization of deep brain stimulation electrode position: an imaging study and systematic review. Stereotact. Funct. Neurosurg. 94:159–163, 2016.
Hariz, M. I. Safety and risk of microelectrode recording in surgery for movement disorders. Stereotact. Funct. Neurosurg. 78:146–157, 2002.
Hariz, G.-M., M. Lindberg, and A. T. Bergenheim. Impact of thalamic deep brain stimulation on disability and health-related quality of life in patients with essential tremor. J. Neurol. Neurosurg. Psychiatry. 72:47–52, 2002.
Herzog, J., J. Volkmann, P. Krack, F. Kopper, M. Pötter, D. Lorenz, M. Steinbach, S. Klebe, W. Hamel, B. Schrader, D. Weinert, D. Müller, H. M. Mehdorn, and G. Deuschl. Two-year follow-up of subthalamic deep brain stimulation in Parkinson’s disease. Mov. Disord. 18:1332–1337, 2003.
Ho, A. L., R. Ali, I. D. Connolly, J. M. Henderson, R. Dhall, S. C. Stein, and C. H. Halpern. Awake versus asleep deep brain stimulation for Parkinson’s disease: a critical comparison and meta-analysis. J. Neurol. Neurosurg. Psychiatry. 89:687–691, 2018.
Ho, A. L., A. V. Pendharkar, R. Brewster, D. L. Martinez, R. A. Jaffe, L. W. Xu, K. J. Miller, and C. H. Halpern. Frameless robot-assisted deep brain stimulation surgery: an initial experience. Oper. Neurosurg. (Hagerstown) 2019. https://doi.org/10.1093/ons/opy395.
Jacob, R. L., J. Geddes, S. McCartney, and K. J. Burchiel. Cost analysis of awake versus asleep deep brain stimulation: a single academic health center experience. J. Neurosurg. 124:1517–1523, 2016.
Jankovic, J. Parkinson’s disease: clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry. 79:368–376, 2008.
Joskowicz, L., R. Sharmir, M. Freiman, M. Shoham, E. Zehavi, F. Umansky, and Y. Shoshan. Image-guided system with miniature robot for precise positioning and targeting in keyhole neurosurgery. Comput. Aided Surg. 11:181–193, 2006.
Khoo, V. S., D. P. Dearnaley, D. J. Finnigan, A. Padhani, S. F. Tanner, and M. O. Leach. Magnetic resonance imaging (MRI): considerations and applications in radiotherapy treatment and planning. Radiother. Oncol. 42:1–15, 1997.
Krack, P., A. Batir, N. Van Blercom, S. Chabardes, V. Fraix, C. Ardouin, A. Koudsie, P. D. Limousin, A. Benazzouz, J. F. LeBas, A.-L. Benabid, and P. Pollak. Five-year follow-up of bilateral stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N. Engl. J. Med. 349:1925–1934, 2003.
Lefranc, M., C. Capel, A. S. Pruvot-Occean, A. Fichten, C. Desenclos, P. Toussaint, D. Le Gars, and J. Peltier. Frameless robotic stereotactic biopsies: a consecutive series of 100 cases. J. Neurosurg. 122:342–352, 2015.
Lefranc, M., Y. Zouitina, M. Tir, P. Merle, M. Ouendo, J.-M. Constans, O. Godefroy, J. Peltier, and P. Krystkowiak. Asleep robot-assisted surgery for the implantation of subthalamic electrodes provides the same clinical improvement and therapeutic window as awake surgery. World Neurosurg. 106:602–608, 2017.
McClelland, III, S., B. Ford, P. B. Senatus, L. M. Winfield, Y. E. Du, S. L. Pullman, Q. Yu, S. J. Frucht, G. M. McKhann, and R. R. Goodman. Subthalamic stimulation for Parkinson disease: determination of electrode location necessary for clinical efficacy. Neurosurg. Focus. 19:1–12, 2005.
Mirzadeh, Z., K. Chapple, M. Lambert, R. Dhall, and F. A. Ponce. Validation of CT-MRI fusion for intraoperative assessment of stereotactic accuracy in DBS surgery. Mov. Disord. 29:1788–1795, 2014.
Mirzadeh, Z., K. Chapple, M. Lambert, V. G. Evidente, P. Mahant, M. C. Ospina, J. Samanta, G. Moguel-Cobos, N. Salins, A. Lieberman, A. I. Tröster, R. Dhall, and F. A. Ponce. Parkinson’s disease outcomes after intraoperative CT-guided “asleep” deep brain stimulation in the globus pallidus internus. J. Neurosurg. 124:902–907, 2016.
Neudorfer, C., S. Hunsche, M. Hellmich, F. El Majdoub, and M. Maarouf. Comparative study of robot-assisted versus conventional frame-based deep brain stimulation stereotactic neurosurgery. Stereotact. Funct. Neurosurg. 96:327–334, 2018.
Ostrem, J. L., N. Ziman, N. B. Galifianakis, P. A. Starr, M. S. Luciano, M. Katz, C. A. Racine, A. J. Martin, L. C. Markun, and P. S. Larson. Clinical outcomes using ClearPoint interventional MRI for deep brain stimulation lead placement in Parkinson’s disease. J. Neurosurg. 124:908–916, 2016.
Rodriguez-Oroz, M. C., J. A. Obeso, A. E. Lang, J. L. Houeto, P. Pollak, S. Rehncrona, J. Kulisevsky, A. Albanese, J. Volkmann, M. I. Hariz, N. P. Quinn, J. D. Speelman, J. Guridi, I. Zamarbide, A. Gironell, J. Molet, B. Pascual-Sedano, B. Pidoux, A. M. Bonnet, Y. Agid, J. Xie, A. L. Benabid, A. M. Lozano, J. Saint-Cyr, L. Romito, M. F. Contarino, M. Scerrati, V. Fraix, and N. Van Blercom. Bilateral deep brain stimulation in Parkinson’s disease: a multicenter study with 4 years follow-up. Brain 128:2240–2249, 2005.
Schrader, B., W. Hamel, D. Weinert, and H. M. Mehdorn. Documentation of electrode localization. Mov. Disord. 17:S167–174, 2002.
Vadera, S., A. Chan, T. Lo, A. Gill, A. Morenkova, N. M. Phielipp, N. Hermanowicz, and F. P. Hsu. Frameless stereotactic robot-assisted subthalamic nucleus deep brain stimulation: case report. World Neurosurg. 97:762.e11–762.e14, 2015.
von Langsdorff, D., P. Paquis, and D. Fontaine. In vivo measurement of the frame-based application accuracy of the Neuromate neurosurgical robot. J. Neurosurg. 122:191–194, 2015.
Zimmermann, M., R. Krishnan, A. Raabe, and V. Seifert. Robot-assisted navigated neuroendoscopy. Neurosurgery. 51:1446–1452, 2002.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Joel Stitzel oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
VanSickle, D., Volk, V., Freeman, P. et al. Electrode Placement Accuracy in Robot-Assisted Asleep Deep Brain Stimulation. Ann Biomed Eng 47, 1212–1222 (2019). https://doi.org/10.1007/s10439-019-02230-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02230-3