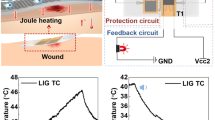
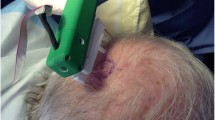
Abstract
Thermotherapy is considered to have potential beneficial effects when applied to wounds. Of particular relevance to this research are wounds that have dropped in temperature due to regional anaesthesia. This study is aimed at developing a normothermic system comprising of a heat patch controlled by external hardware. The study is divided into three parts: (i) the analyses of the skin temperature that form the foundation of the system; (ii) the development of an efficient wearable heat patch incorporating thermoelectric elements to electrical and thermal conductive textiles; and (iii) the hardware development to control the current flow to the thermoelectric elements thus managing the temperature of the heat patch and conserving current. It was observed that a distance of 3 cm between the thermoelectric elements provides ideal heat distribution relative to the surface area. The system allowed for an 80% reduction in current, while maintaining the temperature of the heat patch at the required thermophysiological skin temperature. Future studies will include development of a temperature sensor identifying the real-time temperature of the wound; and circuitry for switching the polarity of the thermoelectric elements. The cooling capabilities of the thermoelectric elements can be applied to wounds that have increased in temperature.









Similar content being viewed by others
References
Allen, T. K., and A. S. Habib. Inadvertent perioperative hypothermia induced by spinal anesthesia for cesarean delivery might be more significant than we think: are we doing enough to warm our parturients? Anesth. Analg. 126(1):7–9, 2018.
Allen, M. W., and D. J. Jacofsky. Normothermia in arthroplasty. J. Arthroplast. 32(7):2307–2314, 2017.
Altura, D. Insulator–conductor device for maintaining a wound near normal body temperature. US Patent No. US 6,613,953 B1, 2003.
Alvarez, O. M., R. S. Rogers, J. G. Booker, and M. Patel. Effect of noncontact normothermic wound therapy on the healing of neuropathic (diabetic) foot ulcers: an interim analysis of 20 patients. J. Foot Ankle Surg. 42(1):30–35, 2003.
Bach, A. J. E., I. B. Stewart, A. E. Disher, and J. T. Costello. A comparison between conductive and infrared devices for measuring mean skin temperature at rest, during exercise in the heat, and recovery. PLoS ONE 10(2):e0117907, 2015.
Banan, B., and W. Chapman. Promise of normothermia. Curr. Transplant. Rep. 4(1):42–51, 2017.
Buggy, D. J., and A. W. Crossley. Thermoregulation, mild perioperative hypothermia and postanaesthetic shivering. Br. J. Anaesth. 84(5):615–628, 2000.
Capon, A., and S. Mordon. Can thermal lasers promote skin wound healing? Am. J. Clin. Dermatol. 4(1):1–12, 2003.
Chanmugam, A., D. Langemo, K. Thomason, J. Haan, E. A. Altenburger, A. Tippet, L. Henderson, and T. A. Zortman. Relative temperature maximum in wound infection and inflammation as compared with a control subject using long-wave infrared thermography. Adv. Skin Wound Care 30(9):406–414, 2017.
Choi, J. K., K. Miki, S. Sagawa, and K. Shiraki. Evaluation of mean skin temperature formulas by infrared thermography. Int. J. Biometeorol. 41:68–75, 1997.
Ferreira, J. J. A., L. C. S. Mendonca, L. A. O. Nunes, A. C. C. A. Filho, J. R. Rebelatto, and T. F. Salvini. Exercise-associated thermographic changes in young and elderly subjects. Ann. Biomed. Eng. 36(8):1420–1427, 2008.
Flouris, A. D., and S. S. Cheung. Design and control optimization of microclimate liquid cooling systems underneath protective clothing. Ann. Biomed. Eng. 34(3):359–372, 2006.
Gurtner, G. C., S. Werner, Y. Barrandon, and M. T. Longaker. Wound repair and regeneration. Nature 453:314–321, 2008.
Jain, R. K. Temperature distributions in normal and neoplastic tissues during normothermia and hyperthermia. Ann. N. Y. Acad. Sci. 335:48–66, 1980.
Koek, M. B. G., T. E. M. Hopmans, L. C. Wille, S. E. Geerlings, M. C. Vos, B. H. B. van Benthem, and S. C. Greeff. Adhering to a national surgical care bundle reduces the risk of surgical site infections. PLoS ONE 12(9):e0184200, 2017.
Kurz, A. Thermal care in the perioperative period. Best Pract. Res. Clin. Anaesthesiol. 22(1):39–62, 2008.
Kurz, A., D. I. Sessler, and R. Lenhardt. Perioperative normothermia to reduce the incidence of surgical-wound infection and shorten hospitalization. Study of Wound Infection and Temperature Group. N. Engl. J. Med. 334(19):1209–1215, 1996.
Leijtens, B., M. Koeter, K. Kremers, and S. Koeter. High incidence of postoperative hypothermia in total knee and total hip arthroplasty: a prospective observational study. J. Arthroplast. 28(6):895–898, 2013.
Lin, Z. P., and S. Deng. A study on the thermal comfort in sleeping environments in the subtropics—developing a thermal comfort model for sleeping environments. Build. Environ. 43(1):70–81, 2008.
Matusiak, M. Investigation of the thermal insulationp of multilayer textiles. Fibers Text. East. Eur. 14(5):98–102, 2006.
Parish, O. L., N. Balachandran, and T. Quisenberry. Method and system for thermal and compression therapy relative to the prevention of deep vein thrombosis. WIPO Patent No. WO2007120639A3, 2008.
Ramos, G. V., C. M. Pinheiro, S. P. Messa, G. B. Delfino, R. C. Marqueti, T. F. Salvini, and J. L. Durigan. Cryotherapy reduces inflammatory response without altering muscle regeneration process and extracellular matrix remodeling of rat muscle. Sci. Rep. 6:18525, 2016.
Rivera, A. E., and J. M. Spencer. Clinical aspects of full-thickness wound healing. Clin. Dermatol. 25(1):39–48, 2007.
Romanovsky, A. A. Skin temperature: its role in thermoregulation. Acta Physiol. 210:498–507, 2014.
Sarver, D. C., K. B. Sugg, N. P. Disser, E. R. S. Enselman, T. M. Awan, and C. L. Mendias. Local cryotherapy minimally impacts the metabolome and transcriptome of human skeletal muscle. Sci. Rep. 7:2423, 2017.
Singh, S., A. Young, and C. E. McNaught. The physiology of wound healing. Surgery 35(9):473–477, 2017.
Teodorczyk, J. E., J. H. Heijmans, W. van Mook, D. Bergmans, and P. Roekaerts. Perioperative normothermia to reduce the incidence of surgical-wound infection and shorten hospitalization. Study of Wound Infection and Temperature Group. Open J. Anesthesiol. 2(3):65–69, 2012.
Thomas, R. T., M. R. Diebold, and L. M. Eggemeyer. A controlled, randomized, comparative study of a radiant heat bandage on the healing of stage 3–4 pressure ulcers: a pilot study. J. Am. Med. Dir. Assoc. 6(1):46–49, 2005.
Tyler, C. J. The effect of skin thermistor fixation method on weighted mean skin temperature. Physiol. Meas. 32:1541–1547, 2011.
Ud-Din, S., and A. Bayat. Non-invasive objective devices for monitoring the inflammatory, proliferative and remodelling phases of cutaneous wound healing and skin scarring. Exp. Dermatol. 25:579–585, 2016.
Whitney, J. D., G. Salvadalena, L. Higa, and M. Mich. Treatment of pressure ulcers with noncontact normothermic wound therapy: healing and warming effects. J. Wound Ostomy Cont. Nurs. 28(5):244–252, 2001.
Zaki, E., A. Salma, and S. Omar. Treatment of auricular keloids by triple combination therapy: surgical excision, platelet-rich plasma, and cryosurgery. J. Cosmet. Dermatol. 17:502–510, 2018.
Zhang, X. Smart Fibres, Fabrics and Clothing, Vol. 1. Cambridge: Woodhead Publishing Limited, pp. 34–57, 2001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Eiji Tanaka oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Logothetis, I., Gkoutzeli, D., Kagkas, D. et al. Thermoelectric Heat Patch for Clinical and Self-Management: Melanoma Excision Wound Care. Ann Biomed Eng 47, 537–548 (2019). https://doi.org/10.1007/s10439-018-02172-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-018-02172-2