Abstract
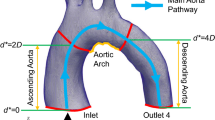
Congenital heart disease is the leading cause of infant death in the United States with over 36,000 newborns affected each year. Despite this growing problem there are few mechanical circulatory support devices designed specifically for pediatric and neonate patients. Previous research has been done investigating pediatric ventricular assist devices (PVADs) assuming blood to be a Newtonian fluid in computational fluid dynamics (CFD) simulations, ignoring its viscoelastic and shear-thinning properties. In contrast to adult VADs, PVADs may be more susceptible to hemolysis and thrombosis due to altered flow into the aorta, and therefore, a more accurate blood model should be used. A CFD solver that incorporates a modified Oldroyd-B model designed specifically for pediatric blood is used to investigate important hemodynamic parameters in a pediatric aortic model under pulsatile flow conditions. These results are compared to Newtonian blood simulations at three physiological pediatric hematocrits. Minor differences are seen in both velocity and wall shear stress (WSS) during early stages of the cardiac cycle between the Newtonian and viscoelastic models. During diastole, significant differences are seen in the velocities in the descending aorta (up to 12%) and in the aortic branches (up to 30%) between the two models. Additionally, peak WSS differences are seen between the models throughout the cardiac cycle. At the onset of diastole, peak WSS differences of 43% are seen between the Newtonian and viscoelastic model and between the 20 and 60% hematocrit viscoelastic models at peak systole of 41%.












Similar content being viewed by others
References
Almond, C., D. Morales, E. Blackstone, M. Turrentine, M. Imamura, M. Massicotte, L. Jordan, et al. Berlin heart excor pediatric ventricular assist device for bridge to heart transplantation in us children. Circulation 127(16):1702–1711, 2013. doi:10.1161/CIRCULATIONAHA.
Anand, M., and K. Rajagopal. A shear-thinning viscoelastic fluid model for describing the flow of blood. Int. J. Cardiovasc. Med. Sci. 4(2):59–68, 2004.
Bachmann, C., G. Hugo, G. Rosenberg, S. Deutsch, A. Fontaine, and J. Tarbell. Fluid dynamics of a pediatric ventricular assist device. Artif Organs. 24(5):362–372, 2000.
Baldwin, J., H. Borovetz, and B. Duncan. The national heart, lung, and blood institute pediatric circulatory support program. Circulation 113(1):147–155, 2006. doi:10.1161/CIRCULATIONAHA.105.571422.
Canter, C., R. Shaddy, and D. Bernstein. Indications for heart transplantation in pediatric heart disease: a scientific statement from the american heart association council on cardiovascular disease in the young; the councils on clinical cardiology, cardiovascular nursing, and cardiovascular surgery and anesthesia: and the quality of care and outcomes research interdisciplinary working group. Circulation 115(5):658–676, 2007.
Chandran, K. B. Flow dynamics in the human aorta. J. Biomech. Eng. 115(4B):611–616, 1993.
Chien, S., S. Usami, R. Dellenback, and M. Gregersen. Blood viscosity: influence of erythrocyte deformation. Science 157:829–831, 1967. doi:10.1126/science.157.3790.829.
Chien, S., S. Usami, R. Dellenback, M. Gregersen, L. Nanninga, and M. Guest. Blood viscosity: influence of erythrocyte aggregation. Science 157:829–831, 1967. doi:10.1126/science.157.3790.829.
Deville, O., and T. Gatski. Mathematical Modeling for Complex Fluids and Flows. Springer Science & Business Media, 2012. doi:10.1007/978-3-642-25295-2.
Favero, J., A. Secchi, N. Cardozo, and H. Jasak. Viscoelastic flow analysis using the software OpenFOAM and differential constitutive equations. J. Non-Newton Fluid 165(23–24):1555–1716, 2010.
Fogel, M., P. Weinberg, J. Rychik, A. Hubbard, M. Jacobs, T. Spray, and J. Haselgrove. Caval contribution to flow in the branch pulmonary arteries of fontan patients with a novel application of magnetic resonance presaturation pulse. Circulation 99(9):1215–1221, 1999.
Gao, F., M. Watanabe, and T. Matsuzawa. Stress analysis in a layered aortic arch model under pulsatil blood flow. Biomed. Eng. Online. 5(25):1–11, 2006. doi:10.1186/1475-925X-5-25.
Giesekus, H. A simple constitutive equation for polymer fluids based on the concept of deformation-dependant tensorial mobility. J. Non-Newton Fluid. 11(1):69–109, 1982. doi:10.1016/0377-0257(82)85016-7.
Gijsen, F., F. van de Vosse, and J. Janssen. The influence of the non-newtonian properties of blood on the flow in large arteries: unsteady flow in a 90 degree curved tube. J. Biomech. 32(7):705–713, 1999.
Gijsen, F., F. van de Vosse, and J. Janssen. The influence of the non-newtonian properties of blood on the flow in larger arteries: steady flow in a carotid bifurcation model. J. Biomech. 32(6):601–608, 1999.
Hakim, S., J. Morshedian, M. Narenji, and P. Nia. Rheological Modelling of Caspian Pony Blood. Iran. Polym. J. 10(5):293–303, 2001.
Issa, R. Solution of the implicitly discretised fluid flow equations by operator-splitting. J. Comput. Phys. 62(1):1–263, 1986.
Johnston, B., P. Johnston, S. Corney, and D. Kilpatrick. Non-Newtonian blood flow in human right coronary arteries: steady state simulations. J. Biomech. 37(5):709–720, 2004.
Kent, A., Z. Kecskes, B. Shadbolt, and M. Falk. Blood pressure in the first year of life in healthy infants born at term. Pediatr. Nephrol. 22(10):1743–1749, 2007; (Epub 2007).
Ku, D. Blood flow in arteries. Annu. Rev. Fluid Mech. 29(1):399–434, 1997. doi:10.1146/annurev.fluid.29.1.
Leonov, A. Nonequilibrium thermodynamics and rheology of viscoelastic polymer media. Rheol. Acta. 15(2):85–98, 1976.
Linderkamp, O., H. Versmold, K. Riegel, and K. Betke. Contributions of red cells and plasma to blood viscosity in preterm and full-term infants and adults. Pediatrics 74(1):45–51, 1984.
Liu, X., Y. Fan, X. Deng, and F. Zhan. Effect of non-newtonian and pulsatile blood flow on mass transport in the human aorta. J. Biomech. 44(6):1123–1131, 2011. doi:10.1016/j.jbiomech.2011.01.024.
Long, J., A. Undar, K. Manning, and S. Deutsch. Viscoelasticity of pediatric blood and its implications for the testing of a pulsatile pediatric blood pump. ASAIO J. 51(5):563–566, 2005.
Machii, M., and A. Becker. Morphologic features of the normal aortic arch in neonates, infants, and children pertinent to growth. Ann. Thorac. Surg. 64(2):511–515, 1997.
Mah, D., T. Singh, R. Thiagarajan, K. Gauvreau, G. Piercey, E. Blume, F. Fynn-Thompson, and C. Almond. Incidence and risk factors for mortality in infants awaiting heart transplantation in the USA. J. Heart Lung Transpl. 28(12):1292–1298, 2009. doi:10.1016/j.healun.2009.06.013.
Nakamura, M., and T. Sawada. Numerical study on the flow of a non-newtonian fluid through an axisymmetric stenosis. J. Biomech. Eng. 110(2):137–143, 1988.
Pantalos, G., G. Giridharan, J. Colyer, M. Mitchel, J. Speakman, C. Lucci, G. Johnson, M. Gartner, and S. Koenig. Effect of continuous and pulsatile left ventricular assist on pulsatility in a pediatric animal model of left ventricular dysfunction: pilot observations. ASAIO J. 53(3):385–391, 2007.
Pekkan, K., O. Dur, K. Sundareswaran, K. Kanter, M. Fogel, A. Yoganathan, and A. Undar. Neonatal aortic arch hemodynamics and perfusion during cardiopulmonary bypass. J. Biomech. Eng. 130(6):061012, 2008. doi:10.1115/1.2978988.
Pekkan, K., D. Frakes, D. De Zelicourt, C. Lucas, J. Parks, and A. Yoganathan. Coupling pediatric ventricle assist devices to the fontan circulation: simulations with a lumped-parameter model. ASAIO J. 51(5):618–628, 2005.
Phillips, W., and S. Deutsch. Towards a constitutive equation for blood. Biorheology 12(6):383–389, 1975.
Rajagopal, K., and A. Srinivasa. A thermodynamic framework for rate-type fluid models. J. Non-Newton Fluid 88(3):207–227, 2000.
Reinhart, W., S. Danoff, R. King, and S. Chien. Rheology of fetal and maternal blood. Pediatr. Res. 19(1):147–153, 1985.
Shahcheraghi, N., H. Dwyer, A. Cheer, I. Barakat, and T. Rutaganira. Unsteady and three-dimensional simulation of blood flow in the human aortic arch. J. Biomech. Eng. 124(4):378–387, 2002.
Thurston, G. Viscoelasticity of human blood. Biophys. J. 12(9):1205–1217, 1972.
Tu, C., and M. Deville. Pulsatile flow of non-newtonian fluids through arterial stenoses. J. Biomech. 29(7):899–908, 1996.
Yang, N., S. Deutsch, E. Paterson, and K. Manning. Comparative study of continuous and pulsatile left ventricular assist devices on hemodynamics of a pediatric end-to-side anastomotic graft. Cardiovasc. Eng. Technol. 1(1):88–103, 2010. doi:10.1007/s13239-010-0006-6.
Yang, N., S. Deutsch, E. Paterson, and K. Manning. Hemodynamics of an end-to-side anastomotic graft for a pulsatile pediatric ventricular assist device. J. Biomech. Eng. 132(3):031009, 2010. doi:10.1115/1.4000872.
Yeleswarapu, K. Evaluation of continuum models for characterizing the constitutive behavior of blood. Pittsburgh: Springer, 1996.
Acknowledgements
We would like to acknowledge the National Institutes of Health for their support of this project through NIH NHLBI HL108123. Bryan C Good, Steven Deutsch, and Keefe B. Manning declare that they have no conflict of interest. No human or animal studies were carried out by the authors for this article. We also thank Ajit Yoganathan, PhD and Christopher M. Haggerty, PhD from the Department of Biomedical Engineering at the Georgia Institute of Technology for providing the pediatric aortic model.
Disclosures
The authors have no disclosures.
Conflict of interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Diego Gallo oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Good, B.C., Deutsch, S. & Manning, K.B. Hemodynamics in a Pediatric Ascending Aorta Using a Viscoelastic Pediatric Blood Model. Ann Biomed Eng 44, 1019–1035 (2016). https://doi.org/10.1007/s10439-015-1370-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-015-1370-z