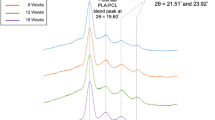
Abstract
Preterm premature rupture of membranes causes 40% of all preterm births, affecting 150000 women each year in the United States. Prenatal diagnostic procedures and surgical interventions increase incidence of adverse events, leading to iatrogenic membrane rupture after a fetoscopic procedure in 45% of cases. We propose an ultrathin, self-adherent, poly-l-lactic acid patch (“nanofilm”) as a reparative wound closure after endoscopic/fetoscopic procedures. These nanofilms are compatible with application in wet conditions and with minimally invasive instrumentation. Ex vivo studies to evaluate the nanofilm were conducted using human chorion–amnion (CA) membranes. A custom-built inflation device was used for mechanical characterization of CA membranes and for assessment of nanofilm adhesion and sealing of membrane defects up to 3 mm in size. These ex vivo tests demonstrated the ability of the nanofilm to seal human CA defects ranging in size from 1 to 3 mm in diameter. In vivo survival studies were conducted in 25 mid-gestational rabbits, defects were created by perforating the uterus and the CA membranes and subsequently using the nanofilm to seal these wounds. These in vivo studies confirmed the successful sealing of defects smaller than 3 mm observed ex vivo. Histological analysis of whole harvested uteri 7 days after surgery showed intact uterine walls in 59% of the nanofilm repaired fetuses, along with increased uterine size and intrauterine development in 63% of the cases. In summary, we have developed an ultrathin, self-adhesive nanofilm for repair of uterine membrane defects.






Similar content being viewed by others
References
Aagaard-Tillery, K. M., F. S. Nuthalapaty, P. S. Ramsey, and K. D. Ramin. Preterm premature rupture of membranes: perspectives surrounding controversies in management. Am. J. Perinatol. 22(6):287–297, 2005.
Adzick, N. S., E. A. Thom, C. Y. Spong, J. W. Brock, III, P. K. Burrows, M. P. Johnson, L. J. Howell, J. A. Farrell, M. E. Dabrowiak, L. N. Sutton, N. Gupta, N. B. Tulipan, M. E. D’Alton, and D. L. Farmer. A randomized trial of prenatal versus postnatal repair of myelomeningocele. N. Engl. J. Med. 364(11):993–1004, 2011.
Beaudoin, S., P. Barbet, and F. Bargy. Developmental stages in the rabbit embryo: guidelines to choose an appropriate experimental model. Fetal. Diagn. Ther. 18:422–427, 2003.
Beck, V., et al. Preterm prelabor rupture of membranes and fetal survival after minimally invasive fetal surgery: a systematic review of the literature. Fetal Diagn. Ther. 31:1–9, 2012.
Behzad, F., M. R. Dickinson, A. Charlton, and J. D. Aplin. Brief communication: sliding displacement of amnion and chorion following controlled laser wounding suggests a mechanism for short-term sealing of ruptured membranes. Placenta 15:775–778, 1994.
Benirschke, K., G. J. Burton, and R. N. Baergen. Anatomy and pathology of the placental membranes. Pathology of the human placenta6th, New York: Springer, 2012, pp. 249–307.
Bilic, G., et al. Injectable candidate sealants for fetal membrane repair: bonding and toxicity in vitro. Am. J. Obstet. Gynecol. 02:85.e1–85.e9, 2001.
Boulet, S. L., Q. Yang, C. Mai, et al. Trends in the postfortification prevalence of spina bifida and anencephaly in the United States. Birth Defects Res. A. Clin. Mol. Teratol 82(7):527–532, 2008.
Chua, W. K., and M. L. Oyen. Do we know the strength of the chorioamnion? A critical review and analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 1445:S128–S133, 2009.
Devlieger, R., L. K. Millar, G. Bryant-Greenwood, L. Lewi, and J. A. Deprest. Fetal membrane healing after spontaneous and iatrogenic membrane rupture: a review of current evidence. Am. J. Obstet. Gynecol. 195:1512–1520, 2006.
Fisk, N. M., D. Ronderos-Dumit, Y. Tannirandorn, U. Nicolini, D. Talbert, and C. H. Rodeck. Normal amniotic pressure throughout gestation. BJOG 99(1):18–22, 1992.
Fujino, K., M. Kinoshita, A. Saitoh, H. Yano, K. Nishikawa, T. Fujie, K. Iwaya, M. Kakihara, S. Takeoka, D. Saitoh, and Y. Tanaka. Novel technique of overlaying a poly-l-lactic acid nanosheet for adhesion prophylaxis and fixation of intraperitoneal onlay polypropylene mesh in a rabbit model. Surg. Endosc. 25(10):3428–3436, 2011.
Gratacós, E., J. Sanin-Blair, L. Lewi, N. Toran, G. Verbist, L. Cabero, and J. Deprest. A histological study of fetoscopic membrane defects to document membrane healing. Placenta 27:452–456, 2006.
Gratacos, E., H. Yamamoto, N. A. Papadopulos, T. Andriaenssens, T. Phlips, T. E. Lerut, and J. A. Deprest. The midgestational rabbit as a model for the creation of membrane defects after needle fetoscopy. Am. J. Obstet. Gynecol. 180(5):1263–1267, 1999.
Haller, C. M., W. Buerzle, C. E. Brubaker, P. B. Messersmith, E. Mazza, N. Ochsenbein-Koelble, R. Zimmermann, and M. Ehrbar. Mussle-mimetic tissue adhesive for fetal membrane repair: a standardized ex vivo evaluation using elastomeric membranes. Prenat. Diagn. 31:654–660, 2011.
Laufer, A., W. Z. Polishuk, J. Boxer, and R. Ganzfried. Studies of amniotic membranes. J. Reprod. Fertil. 12:99–105, 1966.
MacLachlan, T. B. A method for the investigation of the strength of the fetal membranes. Am. J. Obstet. Gynecol. 91:309–313, 1965.
Mercer, B. M. Preterm premature rupture of the membranes: diagnosis and management. Clin. Perinatol. 31(4):765–782, 2004.
Moore, R. M., J. M. Mansour, R. W. Redline, B. M. Mercer, and J. J. Moore. The Physiology of fetal membrane rupture: insight gained from the determination of Physical properties. Placenta 27:1037–1051, 2006.
Okamura, Y., K. Kabata, M. Kinoshita, D. Saitoh, and S. Takeoka. Free-standing biodegradable poly(lactic acid) nanosheet for sealing operations in surgery. Adv. Mater. 21(43):4388–4392, 2009.
Oyen, M. L., S. E. Calvin, and D. V. Landers. Premature rupture of fetal membrane: is the amnion the major determinant? Am. J. Obstet. Gynecol. 195:510–515, 2006.
Papadopulos, N. A., P. P. Van Ballaer, J. L. Ordonez, I. J. Laermans, K. Vandenberghe, T. E. Lerut, and J. A. Deprest. Fetal membrane closing techniques after hysteroamniotomy in the midgestational rabbit model. Am. J. Obstet. Gynecol. 178(5):938–942, 1998.
Peiro, J. L., C. G. Fontecha, R. Ruano, M. Esteves, C. Fonseca, M. Marotta, S. Haeri, and M. A. Belfort. Single Access Fetal Endoscopy (SAFE) for myelomeningocele in sheep model I: amniotic carbon dioxide gas approach. Surg. Endosc. 27:3835–3840, 2013.
Pensabene, V., S. Taccola, L. Ricotti, G. Ciofani, A. Menciassi, F. Perut, M. Salerno, P. Dario, and N. Baldini. Flexible polymeric ultrathin film for mesenchymal stem cell differentiation. Acta Biomater. 7:2883–2891, 2011.
Perrini, M., W. Burzle, C. Haller, N. Ochsenbein-Kolble, J. Deprest, R. Zimmermann, E. Mazza, and M. Ehrbar. Contractions, a risk for premature rupture of fetal membranes: a new protocol with cyclic biaxial tension. Med. Eng. Phys. 35(6):846–851, 2013.
Polishuk, W. Z., S. Kohane, and A. Hader. Fetal weight and membrane tensile strength. Am. J. Obstet. Gynecol. 20:204–250, 1964.
Polishuk, W. Z., S. Konhane, and A. Peranio. The physical properties of fetal membranes. Obstet. Gynecol. 20:204–210, 1962.
Ricotti, L., S. Taccola, V. Pensabene, V. Mattoli, T. Fujie, S. Takeoka, A. Menciassi, and P. Dario. Adhesion and proliferation of skeletal muscle cells on single layer poly(lactic acid) ultra-thin films. Biomed. Microdev. 12:809–819, 2010.
Schmidt, W. The amniotic fluid compartment: the fetal habitat. Adv. Anat. Embryol. Cell. Biol. 127:1–100, 1992.
Sydorak, R., A. Nijagal, and C. T. Albanese. Endoscopic techniques. Yonsei Med. J. 42(6):695–710, 2001.
Taccola, S., A. Desii, V. Pensabene, T. Fujie, A. Saito, S. Takeoka, P. Dario, A. Menciassi, and V. Mattoli. Free-standing poly(l-lactic acid) nanofilms loaded with superparamagnetic nanoparticles. Langmuir 27:5589–5595, 2011.
Wyatt-Ashmead, J., and A. Ashmead. Placental membrane bursting pressures. Mod. Pathol. 17:275, 2004.
Acknowledgments
The authors would like to acknowledge the staff at Division of Surgical Research, Vanderbilt University Medical Center for their support during animal surgery, Prof. Hernan Correa and Dr. Lacey Winchester from the Department of Pathology, Microbiology, and Immunology at Vanderbilt University Medical Center for their help for histological analysis. Special thanks are due to Dr. Kellie Boyle and the Translational Pathology Shared Resource at Vanderbilt University Medical Center. The authors are also grateful to Alicia K. Crum, Registered Diagnostic Medical Sonographer from the Department of fetal medicine for her support for ultrasound, Lynne Black and Prof. Kelly Bennett from the Division of Maternal Fetal Medicine for their help for human placenta collection and IRB approval, and Emily Guess and all the nurses at Labor&Delivery at Vanderbilt University Medical Center for tissues collections.
Disclosures
All authors disclosed no financial relationships relevant to this publication. This study was supported by an internal ViSE (Vanderbilt Initiative in Surgery and Engineering) grant and Vanderbilt Institute for Clinical and Translational Research (VICTR) within the project “Self-adhesive patch for fetal and obstetrical surgery and by CTSA award no. UL1TR000445 from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Jennifer West oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (WMV 17387 kb)
Rights and permissions
About this article
Cite this article
Pensabene, V., Patel, P.P., Williams, P. et al. Repairing Fetal Membranes with a Self-adhesive Ultrathin Polymeric Film: Evaluation in Mid-gestational Rabbit Model. Ann Biomed Eng 43, 1978–1988 (2015). https://doi.org/10.1007/s10439-014-1228-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-1228-9