Abstract
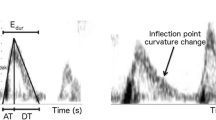
Impedance has been used in vascular biology to characterize the frequency dependent opposition the circulatory system presents to blood flow in response to a pulsatile pressure gradient. It has also been used to characterize diastolic function (DF) via the early, diastolic left ventricular (LV) pressure–flow relation. In a normal LV, early filling volume is accommodated primarily by wall-thinning and ascent of the mitral annulus relative to the spatially fixed apex (longitudinal chamber expansion). Simultaneously, the endocardial (transverse or short axis) dimension also increases while epicardial (transverse) external dimension remains essentially constant. To quantify these directional filling attributes, we compute longitudinal (Z L) and transverse (Z T) impedances during early rapid-filling (Doppler E-wave). Z L and Z T were calculated from 578 cardiac cycles of simultaneous transmitral flow and high fidelity LV pressure data in 17 subjects with normal LV function. Average Z L was 0.7 ± 0.4 mmHg s/cm4 and average Z T was 238 ± 316 mmHg s/cm2. Direct comparison, in the same units is achieved by computing Z T over the ≈10 cm2 cross-sectional area of LV (denoted ŽT) revealing that Z L is ≈34 times smaller than ŽT. This quantifies the physiologic preference for longitudinal LV volume accommodation. Lowest Z L and Z T values occurred in the first harmonic with monotonically increasing values with higher harmonics. We conclude that Z L and Z T characterize longitudinal and transverse chamber properties of DF and therefore, diastolic dysfunction can be viewed as a state of impedance mismatch.




Similar content being viewed by others
References
Appleton, C. P., M. S. Firstenberg, M. J. Garcia, and J. D. Thomas. The echo-Doppler evaluation of left ventricular diastolic function. Cardiol. Clin. 18:513–546, 2000.
Avolio, A. P., M. F. O’Rourke, K. Mang, P. T. Bason, and B. S. Gow. A comparative study of pulsatile arterial hemodynamics in rabbits and guinea pigs. Am. J. Physiol. 230:868–875, 1976.
Baumgartner, H., J. Hung, J. Bermejo, J. B. Chambers, A. Evangelista, B. P. Griffin, B. Iung, C. M. Otto, P. A. Pellikka, and M. Quiñones. Echocardiographic assessment of valve stenosis: EAE/ASE Recommendations for clinical practice. J. Am. Soc. Echocardiogr. 22(1):1–23, 2009.
Bowman, A. W., and S. J. Kovács. Assessment and consequences of the constant-volume attribute of the four-chambered heart. Am. J. Physiol. Heart Circ. Physiol. 285:H2027–H2033, 2003.
Buckberg, G., A. Mahajan, S. Saleh, J. I. Hoffman, and C. Coghlan. Structure and function relationships of the helical ventricular myocardial band. J. Thorac. Cardiovasc. Surg. 136:578–589, 89.e1–89.e11, 2008.
Bursi, F., S. A. Weston, M. M. Redfield, S. J. Jacobsen, S. Pakhomov, V. T. Nkomo, R. A. Meverden, and V. L. Roger. Systolic and diastolic heart failure in the community. J. Am. Med. Assoc. 296:2209–2216, 2006.
Carlsson, M., M. Ugander, E. Heiberg, and H. Arheden. The quantitative relationship between longitudinal and radial function in left, right, and total heart pumping in humans. Am. J. Physiol. Heart Circ. Physiol. 293:H636–H644, 2007.
Caro, C., T. Pedley, R. Schroter, and W. Seed. The Mechanics of the Circulation. London: Oxford University Press, 1978.
Chung, C. S., D. M. Ajo, and S. J. Kovács. Isovolumic pressure-to-early rapid filling decay rate relation: model-based derivation and validation via simultaneous catheterization echocardiography. J. Appl. Physiol. 100:528–534, 2006.
Courtois, M. R., S. J. Kovács, Jr., and P. A. Ludbrook. Physiologic early diastolic pressure gradient is lost during acute myocardial ischemia. Circulation 81:1688–1696, 1990.
Curi, M. A., C. L. Skelly, C. Quint, S. L. Meyerson, A. J. Farmer, U. M. Shakur, F. Loth, and L. B. Schwartz. Longitudinal impedance is independent of outflow resistance. J. Surg. Res. 108:191–197, 2002.
De Mey, S. K. Dumont, J. Geeraerts, P. Vandervoort, and P. Verdonck. Non-invasive assessment of left ventricular longitudinal impedance using color M-mode Doppler echocardiography. Comput. Cardiol. 27:17–20, 2000.
Eaton, G. M., R. J. Cody, and P. F. Binkley. Increased aortic impedance precedes peripheral vasoconstriction at the early stage of ventricular failure in the paced canine model. Circulation 88:2714–2721, 1993.
Ghosh, E., and S. J. Kovács. Longitudinal and transverse impedance can quantify left ventricular diastolic function. J. Am. Coll. Cardiol. 59(13s1):E1063–E1063, 2012.
Ghosh, E., and S. J. Kovács. Quantitative assessment of left ventricular diastolic function via longitudinal and transverse flow impedances. Conf. Proc. IEEE Eng. Med. Biol. Soc. 5595–5598, 2012.
Ghosh, E., L. Shmuylovich, and S. J. Kovács. Vortex formation time- to- left ventricular early rapid filling relation: model-based prediction with echocardiographic validation. J. Appl. Physiol. 109:1812–1819, 2010.
Greenberg, N. L., P. M. Vandervoort, M. S. Firstenberg, M. J. Garcia, and J. D. Thomas. Estimation of diastolic intraventricular pressure gradients by Doppler M-mode echocardiography. Am. J. Physiol. Heart Circ. Physiol. 280:H2507–H2515, 2001.
Hall, A. F., and S. J. Kovács. Automated method for characterization of diastolic transmitral Doppler velocity contours: early rapid filling. Ultrasound Med. Biol. 20:107–116, 1994.
Hall, A. F., S. P. Nudelman, and S. J. Kovács. Evaluation of model-based processing algorithms for averaged transmitral spectral Doppler images. Ultrasound Med. Biol. 24:55–66, 1998.
Katz, L. N. The role played by the ventricular relaxation process in filling the ventricle. Am. J. Physiol. 95:542–553, 1930.
Maeder, M. T., and D. M. Kaye. Heart failure with normal left ventricular ejection fraction. J. Am. Coll. Cardiol. 53:905–918, 2009.
Merillon, J. P., G. J. Fontenier, J. F. Lerallut, M. Y. Jaffrin, G. A. Motte, C. P. Genain, and R. R. Gourgon. Aortic input impedance in normal man and arterial hypertension: its modification during changes in aortic pressure. Cardiovasc. Res. 16:646–656, 1982.
Milde, J., M. Sessoms, J. Lisauskas, A. W. Bowman, M. Courtois, and S. J. Kovács. Ventricular diastolic impedance: a new index of global diastolic function. J. Am. Coll. Cardiol. 53, 2000.
Milde, J. M., M. W. Sessoms, J. B. Lisauskas, M. Courtois, J. Singh, and S. J. Kovács. Ventricular diastolic impedance Z(t): validation of a new, model-based image processing derived index of global diastolic function. In: Proceedings of the 1st Joint BMES/EMBS Conference, Vol. 1, 1999.
Milnor, W. R. Hemodynamics. Baltimore, MD: Williams & Wilkins, 1982.
Milnor, W. R. Arterial impedance as ventricular afterload. Circ. Res. 36:565–570, 1975.
Murgo, J. P., N. Westerhof, J. P. Giolma, and S. A. Altobelli. Aortic input impedance in normal man: relationship to pressure wave forms. Circulation 62:105–116, 1980.
Nagueh, S. F., C. P. Appleton, T. C. Gillebert, P. N. Marino, J. K. Oh, O. A. Smiseth, A. D. Waggoner, F. A. Flachskampf, P. A. Pellikka, and A. Evangelista. Recommendations for the evaluation of left ventricular diastolic function by echocardiography. Eur. J. Echocardiogr. 10:165–193, 2009.
Nichols, W., and M. O’Rourke. McDonald’s Blood Flow in Arteries. London: Hodder & Arnold, 2005.
O’Rourke, M. F. Pressure and flow waves in systemic arteries and the anatomical design of the arterial system. J. Appl. Physiol. 23:139–149, 1967.
O’Rourke, M. F., and M. Taylor. Vascular impedance of the femoral bed. Circ. Res. 18:126–139, 1966.
Pepine, C. J., and W. W. Nichols. Aortic input impedance in cardiovascular disease. Prog. Cardiovasc. Dis. 24:307–318, 1982.
Robinson, T. F., S. M. Factor, and E. H. Sonnenblick. The heart as a suction pump. Sci. Am. 254(6):84–91, 1986.
Shmuylovich, L., and S. J. Kovács. Stiffness and relaxation components of the exponential and logistic time constants may be used to derive a load-independent index of isovolumic pressure decay. Am. J. Physiol. Heart Circ. Physiol. 295:H2551–H2559, 2008.
Toger, J., M. Carlsson, G. Soderlind, H. Arheden, and E. Heiberg. Volume tracking: a new method for quantitative assessment and visualization of intracardiac blood flow from three-dimensional, time-resolved, three-component magnetic resonance velocity mapping. BMC Med. Imaging 11:10, 2011.
Westerhof, N., N. Stergiopulos, and M. I. M. Noble. Snapshots of Hemodynamics (2nd ed.). New York: Springer, 2010.
Wu, Y., A. W. Bowman, and S. J. Kovács. Frequency-based analysis of diastolic function: the early rapid filling phase generates negative intraventricular wave reflections. Cardiovasc. Eng. 5:1–12, 2005.
Wu, Y., and S. J. Kovács. Frequency-based analysis of the early, rapid-filling pressure–flow relation elucidates diastolic efficiency mechanisms. Am. J. Physiol. Heart Circ. Physiol. 291:H2942–H2949, 2006.
Yano, M., M. Kohno, S. Kobayashi, M. Obayashi, K. Seki, T. Ohkusa, T. Miura, T. Fujii, and M. Matsuzaki. Influence of timing and magnitude of arterial wave reflection on left ventricular relaxation. Am. J. Physiol. Heart Circ. Physiol. 280:H1846–H1852, 2001.
Yin, F. C., M. L. Weisfeldt, and W. R. Milnor. Role of aortic input impedance in the decreased cardiovascular response to exercise with aging in dogs. J. Clin. Invest. 68:28–38, 1981.
Acknowledgments
The assistance of the staff of the Cardiovascular Procedure Center at Barnes-Jewish Hospital at Washington University Medical Center and Peggy Brown for expert echocardiographic data acquisition is gratefully acknowledged. This work was supported in part by the Alan A. and Edith L. Wolff Charitable Trust (St. Louis, MO) and the Barnes-Jewish Hospital Foundation. E. Ghosh is a recipient of a Heartland Affiliate Predoctoral Fellowship Award from the American Heart Association.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Aleksander S. Popel oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Ghosh, E., Kovács, S.J. Early Left Ventricular Diastolic Function Quantitation Using Directional Impedances. Ann Biomed Eng 41, 1269–1278 (2013). https://doi.org/10.1007/s10439-013-0756-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-013-0756-z