Abstract
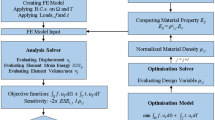
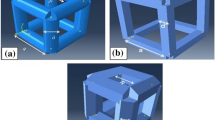
Computer-Aided Tissue Engineering (CATE) is based on a set of additive manufacturing techniques for the fabrication of patient-specific scaffolds, with geometries obtained from medical imaging. One of the main issues regarding the application of CATE concerns the definition of the internal architecture of the fabricated scaffolds, which, in turn, influences their porosity and mechanical strength. The present study envisages an innovative strategy for the fabrication of highly optimized structures, based on the a priori finite element analysis (FEA) of the physiological load set at the implant site. The resulting scaffold micro-architecture does not follow a regular geometrical pattern; on the contrary, it is based on the results of a numerical study. The algorithm was applied to a solid free-form fabrication process, using poly(ε-caprolactone) as the starting material for the processing of additive manufactured structures. A simple and intuitive geometry was chosen as a proof-of-principle application, on which finite element simulations and mechanical testing were performed. Then, to demonstrate the capability in creating mechanically biomimetic structures, the proximal femur subjected to physiological loading conditions was considered and a construct fitting a femur head portion was designed and manufactured.






Similar content being viewed by others
References
Abbah, S. A., C. X. Lam, D. W. Hutmacher, J. C. Goh, and H.-K. Wong. Biological performance of a polycaprolactone-based scaffold used as fusion cage device in a large animal model of spinal reconstructive surgery. Biomaterials 30:5086–5093, 2009.
Adachi, T., Y. Osako, M. Tanaka, M. Hojo, and S. J. Hollister. Framework for optimal design of porous scaffold microstructure by computational simulation of bone regeneration. Biomaterials 27:3964–3972, 2006.
Almeida, H. A., and P. J. Bartolo. Virtual topological optimisation of scaffolds for rapid prototyping. Med. Eng. Phys. 32:775–782, 2010.
Bartolo, P. J., C. K. Chua, H. A. Almeida, S. M. Chou, and A. S. C. Lim. Biomanufacturing for tissue engineering: present and future trends. Virtual Phys. Prototyp. 4:203–216, 2009.
Cahill, S., S. Lohfeld, and P. E. McHugh. Finite element predictions compared to experimental results for the effective modulus of bone tissue engineering scaffolds fabricated by selective laser sintering. J. Mater. Sci. Mater. Med. 20:1255–1262, 2009.
Cheah, C.-M., C.-K. Chua, K.-F. Leong, C.-H. Cheong, and M.-W. Naing. Automatic algorithm for generating complex polyhedral scaffolds for tissue engineering. Tissue Eng. 10:595–610, 2004.
Chen, Z., Z. Su, S. Ma, X. Wu, and Z. Luo. Biomimetic modeling and three-dimension reconstruction of the artificial bone. Comput. Methods Programs Biomed. 88:123–130, 2007.
Cheung, G., P. Zalzal, M. Bhandari, J. K. Spelt, and M. Papini. Finite element analysis of a femoral retrograde intramedullary nail subject to gait loading. Med. Eng. Phys. 26:93–108, 2004.
Eshraghi, S., and S. Das. Mechanical and microstructural properties of polycaprolactone scaffolds with one-dimensional, two-dimensional, and three-dimensional orthogonally oriented porous architectures produced by selective laser sintering. Acta Biomater. 6:2467–2476, 2010.
Gibson, L. J., and M. F. Ashby. Cellular Solids: Structure and Properties. Cambridge University Press: Cambridge, 532 pp, 1999.
Hobbie, R. K., and B. J. Roth. Intermediate Physics for Medicine and Biology. Springer: New York, 575 pp, 2007.
Hollister, S. J. Porous scaffold design for tissue engineering. Nat. Mater. 4:518–524, 2005.
Hutmacher, D. W., T. Schantz, I. Zein, K. W. Ng, S. H. Teoh, and K. C. Tan. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J. Biomed. Mater. Res. 55:203–216, 2001.
Hutmacher, D. W., M. Sittinger, and M. V. Risbud. Scaffold-based tissue engineering: rationale for computer-aided design and solid free-form fabrication systems. Trends Biotechnol. 22:354–362, 2004.
Jones, J. R., and L. L. Hench. Regeneration of trabecular bone using porous ceramics. Curr. Opin. Solid State Mater. Sci. 7:301–307, 2003.
Kalita, S. J., S. Bose, H. L. Hosick, and A. Bandyopadhyay. Development of controlled porosity polymer–ceramic composite scaffolds via fused deposition modeling. Mater. Sci. Eng. C. 23:611–620, 2003.
Keaveny, T. M., X. E. Guo, E. F. Wachtel, T. A. McMahon, and W. C. Hayes. Trabecular bone exhibits fully linear elastic behavior and yields at low strains. J. Biomech. 27:1127–1136, 1994.
Koch, J. C. The laws of bone architecture. Am. J. Anat. 21:177–298, 1917.
Lacroix, D., J. A. Planell, and P. J. Prendergast. Computer-aided design and finite-element modelling of biomaterial scaffolds for bone tissue engineering. Philos. Trans. A Math. Phys. Eng. Sci. 367:1993–2009, 2009.
Landau, D. L., and E. M. Lifshitz. Theory of Elasticity. Oxford: Butterworth Heinemann, 1986; (187 pp).
Lee, C. H., J. L. Cook, A. Mendelson, E. K. Moioli, H. Yao, and J. J. Mao. Regeneration of the articular surface of the rabbit synovial joint by cell homing: a proof of concept study. Lancet 376:440–448, 2010.
Lin, C. Y., N. Kikuchi, and S. J. Hollister. A novel method for biomaterial scaffold internal architecture design to match bone elastic properties with desired porosity. J. Biomech. 37:623–636, 2004.
McIntosh, L., J. M. Cordell, and A. J. Wagoner Johnson. Impact of bone geometry on effective properties of bone scaffolds. Acta Biomater. 5:680–692, 2009.
Melchels, F. P. W., K. Bertoldi, R. Gabbrielli, A. H. Velders, J. Feijen, and D. W. Grijpma. Mathematically defined tissue engineering scaffold architectures prepared by stereolithography. Biomaterials 31:6909–6916, 2010.
Olivares, A. L., E. Marsal, J. A. Planell, and D. Lacroix. Finite element study of scaffold architecture design and culture conditions for tissue engineering. Biomaterials 30:6142–6149, 2009.
Pálfi, P. Locally orthotropic femur model. J. Comput. Appl. Mech. 5:103–115, 2002.
Pandithevan, P., and G. Saravana Kumar. Reconstruction of subject-specific human femoral bone model with cortical porosity data using macro-CT. Virtual Phys. Prototyp. 4:115–129, 2009.
Pandithevan, P., and G. Saravana Kumar. Finite element analysis of a personalized femoral scaffold with designed microarchitecture. Proc. IMechE H J. Eng. Med. 224:877–889, 2010.
Papini, M., R. Zdero, E. H. Schemitsch, and P. Zalzal. The biomechanics of human femurs in axial and torsional loading: comparison of finite element analysis, human cadaveric femurs, and synthetic femurs. J. Biomech. Eng. 129:12–19, 2007.
Peltola, S. M., F. P. W. Melchels, D. W. Grijpma, and M. Kellomaki. A review of rapid prototyping techniques for tissue engineering purposes. Ann. Med. 40:268–280, 2008.
Rezwan, K., Q. Z. Chen, J. J. Blaker, and A. R. Boccaccini. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 27:3413–3431, 2006.
Sandino, C., J. A. Planell, and D. Lacroix. A finite element study of mechanical stimuli in scaffolds for bone tissue engineering. J. Biomech. 41:1005–1014, 2008.
Shao, X., J. C. Goh, D. W. Hutmacher, E. H. Lee, and G. Zigang. Repair of large articular osteochondral defects using hybrid scaffolds and bone marrow-derived mesenchymal stem cells in a rabbit model. Tissue Eng. 12:1539–1551, 2006.
Shipley, R. J., G. W. Jones, R. J. Dyson, B. G. Sengers, C. L. Bailey, C. J. Catt, C. P. Please, and J. Malda. Design criteria for a printed tissue engineering construct: a mathematical homogenization approach. J. Theor. Biol. 259:489–502, 2009.
Shor, L., S. Guceri, R. Chang, J. Gordon, Q. Kang, L. Hartsock, Y. An, and W. Sun. Precision extruding deposition (PED) fabrication of polycaprolactone (PCL) scaffolds for bone tissue engineering. Biofabrication 1:015003, 2009.
Simpson, R. L., F. E. Wiria, A. A. Amis, C. K. Chua, K. F. Leong, U. N. Hansen, M. Chandrasekaran, and M. W. Lee. Development of a 95/5 poly(l-lactide-co-glycolide)/hydroxylapatite and beta-tricalcium phosphate scaffold as bone replacement material via selective laser sintering. J. Biomed. Mater. Res. B Appl. Biomater. 84:17–25, 2008.
Sobral, J. M., S. G. Caridade, R. A. Sousa, J. F. Mano, and R. L. Reis. Three-dimensional plotted scaffolds with controlled pore size gradients: effect of scaffold geometry on mechanical performance and cell seeding efficiency. Acta Biomater. 7:1009–1018, 2011.
Starly, B., W. Lau, T. Bradbury, and W. Sun. Internal architecture design and freeform fabrication of tissue replacement structures. Computer-Aided Des. 38:115–124, 2006.
Sun, W., B. Starly, A. Darling, and C. Gomez. Computer aided tissue engineering application to biomimetic modeling and design of tissue scaffold. Biotechnol. Appl. Biochem. 39:49–58, 2004.
Swieszkowski, W., B. H. Tuan, K. J. Kurzydlowski, and D. W. Hutmacher. Repair and regeneration of osteochondral defects in the articular joints. Biomol. Eng. 24:489–495, 2007.
Tellis, B. C., J. A. Szivek, C. L. Bliss, D. S. Margolis, R. K. Vaidyanathan, and P. Calvert. Trabecular scaffolds created using micro CT guided fused deposition modeling. Mater. Sci. Eng. C Mater. Biol. Appl. 28:171–178, 2009.
Viceconti, M. A comparative study on different methods of automatic mesh generation on human femurs. Med. Eng. Phys. 20:1–10, 1998.
Williams, J. M., A. Adewunmi, R. M. Schek, C. L. Flanagan, P. H. Krebsbach, S. E. Feinberg, S. J. Hollister, and S. Das. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials 26:4817–4827, 2005.
Acknowledgment
This work was partially supported by University Campus Bio-Medico di Roma under the CIR Young Investigator Research Grant.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Thurmon E. Lockhart oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Rainer, A., Giannitelli, S.M., Accoto, D. et al. Load-Adaptive Scaffold Architecturing: A Bioinspired Approach to the Design of Porous Additively Manufactured Scaffolds with Optimized Mechanical Properties. Ann Biomed Eng 40, 966–975 (2012). https://doi.org/10.1007/s10439-011-0465-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-011-0465-4