Abstract
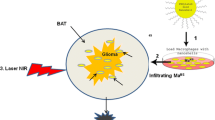
Site-specific delivery of nanoparticles poses a significant challenge, especially in the brain where the blood–brain barrier prevents the entry of most therapeutic compounds including nanoparticle-based anti-cancer agents. In this context, the use of macrophages as vectors for the delivery of gold–silica nanoshells to infiltrating gliomas will be reviewed in this article. Gold–silica nanoshells are readily phagocytosed by macrophages without any apparent toxic effects, and the results of in vitro studies have demonstrated the migratory potential of nanoshell-loaded macrophages in human glioma spheroids. Of particular interest is the observation that, after near-infrared exposure of spheroids containing nanoshell-loaded macrophages, sufficient heat was generated to suppress spheroid growth. Collectively, these findings demonstrate the potential of macrophages as nanoshell delivery vectors for photothermal therapy of gliomas, and they certainly provide the basis for future animal studies.









Similar content being viewed by others
References
American Cancer Society. Cancer Facts and Figures, 2010.
Baek, S. K., A. R. Makkouk, T. Krasieva, C. H. Sun, S. J. Madsen, and H. Hirschberg. Photothermal treatment of glioma: an in vitro study of macrophage-mediated delivery of gold nanoshells. J. Neurooncol. Epub ahead of print.
Ballabh, P., A. Braun, and M. Nedergaard. The blood–brain barrier: an overview: structure, regulation and clinical implications. Neurobiol. Dis. 16:1–13, 2004.
Beduneau, A., Z. Ma, C. B. Grotepas, A. Kabanov, B. E. Rabinow, N. Gong, R. L. Mosley, H. Dou, M. D. Boska, and H. E. Gendelman. Facilitated monocyte-macrophage uptake and tissue distribution of superparamagnetic iron-oxide nanoparticles. PLoS One 4:e4343, 2009.
Bernardi, R. J., A. R. Lowery, P. A. Thompson, S. M. Blaney, and J. L. West. Immunonanoshells for targeted photothermal ablation of medulloblastoma and glioma: an in vitro evaluation using human cell lines. J. Neurooncol. 86:165–172, 2008.
Carson, M. J., J. Crane, and A. X. Xie. Modeling CNS microglia: the quest to identify predictive models. Drug Discov. Today Dis. Models. 5:19–25, 2008.
Choi, M. R., K. J. Stanton-Maxey, J. K. Stanley, C. S. Levin, R. Bardhan, D. Akin, S. Badve, J. Sturgis, J. P. Robinson, R. Bashir, N. J. Halas, and S. E. Clare. A cellular Trojan Horse for delivery of therapeutic nanoparticles into tumors. Nano Lett. 7:3759–3765, 2007.
Cuenca, A. G., H. Jiang, S. N. Hochwald, M. Delano, W. G. Cance, and S. R. Grobmyer. Emerging implications of nanotechnology on cancer diagnostics and therapeutics. Cancer. 107:459–466, 2006.
Curley, S. A., P. Cherukuri, K. Briggs, C. R. Patra, M. Upton, E. Dolson, and P. Mukherjee. Nonivasive radiofrequency field-induced hyperthermic cytotoxicity in human cancer cells using cetuximab-targeted gold nanoparticles. J. Exp. Ther. Oncol. 7:313–326, 2008.
Cushing, H. Intracranial tumors: notes upon a series of two thousand verified cases with surgical mortality percentages pertaining thereto. Springfield, IL: Charles C. Thomas, 1932.
Day, E. S., P. A. Thompson, L. Zhang, N. A. Lewinski, N. Ahmed, R. A. Drezek, S. M. Blaney, and J. L. West. Nanoshell-mediated photothermal therapy improves survival in a murine glioma model. J. Neurooncol. Epub ahead of print.
Everts, M. Thermal scalpel to target cancer. Expert Rev. Med. Devices. 4:131–136, 2007.
Fleige, G., C. Nolte, M. Synowitz, F. Seeberger, H. Kettenmann, and C. Zimmer. Magnetic labeling of activated microglia in experimental gliomas. Neoplasia. 3:489–499, 2001.
Gobin, A. M., J. J. Moon, and J. L. West. EphrinA 1-targeted nanoshells for photothermal ablation of prostate cancer cells. Int. J. Nanomed. 3:351–358, 2008.
Goodman, T. T., J. Chen, K. Matveev, and S. H. Pun. Spatio-temporal modeling of nanoparticle delivery to multicellular tumor spheroids. Biotechnol. Bioeng. 101:388–399, 2008.
Halas, N. J. Tuning the optical resonant properties of nanoshells. MRS Bull. 30:362–367, 2005.
Hamoudeh, M., M. A. Kamleh, R. Diab, and H. Fessi. Radionuclides delivery systems for nuclear imaging and radiotherapy of cancer. Adv. Drug Deliv. Rev. 60:1329–1346, 2008.
Hickey, V. F. Leukocyte traffic in the central nervous system: the participants and their roles. Semin. Immunol. 11:125–137, 1999.
Hirsch, L. R., R. J. Stafford, J. A. Bankson, S. R. Sershen, B. Rivera, R. E. Price, J. D. Hazle, N. J. Halas, and J. L. West. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc. Natl. Acad. Sci. USA 100:13549–13554, 2003.
Hirschberg, H., S. K. Baek, Y. J. Kwon, C. H. Sun, and S. J. Madsen, Bypassing the blood–brain barrier: delivery of therapeutic agents by macrophages. Proc. SPIE. 7548:3Z-1, 2010.
Hsiao, J. K., H. H. Chu, Y. H. Wang, C. W. Lai, P. T. Chou, S. T. Hsieh, J. L. Wang, and H. M. Liu. Macrophage physiological function after superparamagnetic iron oxide labeling. NMR Biomed. 21:820–829, 2008.
Huang, X., I. H. El-Sayed, W. Qian, and M. A. El-Sayed. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 128:2115–2120, 2006.
Huang, X., P. K. Jain, I. H. El-Sayed, and M. A. El-Sayed. Gold nanoparticles: interesting optical properties and recent applications in cancer diagnostics and therapy. Nanomedicine 2:681–693, 2007.
Huang, X., P. K. Jain, I. H. El-Sayed, and M. A. El-Sayed. Plasmonic photothermal therapy (PPTT) using gold nanoparticles. Lasers Med. Sci. 23:217–228, 2008.
Huber, J., R. Egleton, and T. Davis. Molecular physiology and pathophysiology of tight junctions in the blood–brain barrier. Trends Neurosci. 24:719–725, 2001.
Ivascu, A., and M. Kubbies. Rapid generation of single-tumor spheroids for high-throughput cell function and toxicity analysis. J. Biomol. Screen. 11:922–932, 2006.
James, W. D., L. R. Hirsch, J. L. West, P. D. O’Neal, and J. D. Payne. Applications of INAA to the build-up and clearance of gold nanoshells in clinical studies in mice. J. Radioanal. Nucl. Chem. 271:455–459, 2007.
Kah, J. C., K. Y. Wong, K. G. Neoh, J. H. Song, J. W. Fu, S. Mhaisalkar, M. Olivo, and C. J. Sheppard. Critical parameters in the pegylation of gold nanoshells for biomedical applications: an in vitro macrophage study. J. Drug Target. 17:181–193, 2009.
Knowles, H. J., and A. L. Harris. Macrophages and the hypoxic tumour microenvironment. Front. Biosci. 12:4298–4314, 2007.
Liu, S. Y., Z. S. Liang, F. Gao, S. F. Luo, and G. Q. Lu. In vitro photothermal study of gold nanoshells functionalized with small targeting peptides to liver cancer cells. J. Mater. Sci. Mater. Med. 21:665–674, 2009.
Loo, C., L. Hirsch, M. H. Lee, E. Chang, J. West, N. Halas, and R. Drezek. Gold nanoshell bioconjugates for molecular imaging in living cells. Opt. Lett. 30:1012–1014, 2005.
Loo, C., A. Lin, L. Hirsch, M. H. Lee, J. Barton, N. Halas, J. West, and R. Drezek. Nanoshell-enabled photonics-based imaging and therapy of cancer. Technol. Cancer Res. Treat. 3:33–40, 2004.
Lowery, A. R., A. M. Gobin, E. S. Day, N. J. Halas, and J. L. West. Immunonanoshells for targeted photothermal ablation of tumor cells. Int. J. Nanomed. 1:149–154, 2006.
Madsen, S. J., E. Angell-Petersen, S. Spetalen, S. W. Carper, S. A. Ziegler, and H. Hirschberg. Photodynamic therapy of newly implanted glioma cells in the rat brain. Lasers Surg. Med. 38:540–548, 2006.
Madsen, S. J., C. H. Sun, B. J. Tromberg, V. Cristini, N. DeMagalhaes, and H. Hirschberg. Multicell tumor spheroids in photodynamic therapy. Lasers Surg. Med. 38:555–564, 2006.
Maeda, H. The enhanced permeability and retention effect in tumor vasculature: the key role of tumor-selective macromolecular drug targeting. Adv. Enzym. Regul. 41:189–207, 2001.
Martin, V., D. Liu, and C. Gomez-Manzano. Encountering and advancing through antiangiogenesis therapy for gliomas. Curr. Pharm. Des. 15:353–364, 2009.
Metz, S., G. Bonaterra, M. Rudelius, M. Settles, E. J. Rummeny, and H. E. Daldrup-Link. Capacity of human monocytes to phagocytose approved iron oxide MR contrast agents in vitro. Eur. Radiol. 14:1851–1858, 2004.
Murdcoch, C., and C. E. Lewis. Macrophage migration and gene expression in response to tumor hypoxia. Int. J. Cancer. 117:701–708, 2005.
O’Neal, D. P., L. R. Hirsch, N. J. Halas, J. D. Payne, and J. L. West. Photo-thermal tumor ablation in mice using near infrared-absorbing nanoparticles. Cancer Lett. 209:171–176, 2004.
Oldenburg, S. J., R. D. Averitt, and S. L. Westcott. Nanoengineering of optical resonances. Chem. Phys. Lett. 288:243–247, 1998.
Oldenburg, S. J., J. B. Jackson, S. L. Westcott, and N. J. Halas. Infrared extinction properties of gold nanoshells. Appl. Phys. Lett. 75:2897–2899, 1999.
Oude Engberink, R. D., E. L. Blezer, E. I. Hoff, S. M. van der Pol, A. van der Toorn, R. M. Dijkhuizen, and H. E. de Vries. MRI of monocyte infiltration in an animal model of neuroinflammation using SPIO-labeled monocytes or free USPIO. J. Cereb. Blood Flow Metab. 28:841–851, 2007.
Owen, M. R., H. M. Byrne, and C. E. Lewis. Mathematical modeling of the use of macrophages as vehicles for drug delivery to hypoxic tumour sites. J. Theor. Biol. 226:377–391, 2004.
Raynal, I., P. Prigent, S. Peyramaure, A. Najid, C. Rebuzzi, and C. Corot. Macrophage endocytosis of superparamagnetic iron oxide nanoparticles: mechanisms and comparison of ferumoxides and ferumoxtran-10. Invest. Radiol. 39:56–63, 2004.
Schwartz, J. A., A. M. Shetty, R. E. Price, R. J. Stafford, J. C. Wang, R. K. Uthamanthil, K. Pham, R. J. McNichols, C. L. Coleman, and J. D. Payne. Feasibility study of particle-assisted laser ablation of brain tumors in orthotopic canine model. Cancer Res. 69:1659–1667, 2009.
Stern, J. M., J. Stanfield, Y. Lotan, S. Park, J. T. Hsieh, and J. A. Cadeddu. Efficacy of laser-activated gold nanoshells in ablating prostate cancer cells in vitro. J. Endourol. 21:939–943, 2007.
Terentyuk, G. S., G. N. Maslyakova, L. V. Suleymanova, N. G. Khlebtsov, B. N. Khlebtsov, G. G. Akchurin, I. L. Maksimova, and V. V. Tuchin. Laser-induced tissue hyperthermia mediated by gold nanoparticles: toward cancer phototherapy. J. Biomed. Opt. 14:021016, 2009.
Valable, S., E. L. Barbier, M. Bernaudin, S. Roussel, C. Segebarth, E. Petit, and C. Remy. In vivo MRI tracking of exogenous monocytes/macrophages targeting brain tumors in a rat model of glioma. Neuroimage. 40:973–983, 2008.
Wallner, K. E., J. H. Galicich, G. Krol, E. Arbit, and M. G. Malkin. Patterns of failure following treatment for glioblastoma multiforme and anaplastic astrocytoma. Int. J. Radiat. Oncol. Biol. Phys. 16:1405–1409, 1989.
Wu, J., S. Yang, H. Luo, L. Zeng, L. Ye, and Y. Lu. Quantitative evaluation of monocyte transmigration into the brain following chemical opening of the blood-brain barrier in mice. Brain Res. 1098:79–85, 2006.
Acknowledgments
This study was partially supported by the Health Sciences System of the Nevada System of Higher Education through the Inter-Institutional Biomedical Research Activities Fund (IBRAF). This study was also supported by the Laser Microbeam and Medical Program (LAMMP) and the Chao Cancer Center Optical Biology Shared Resource at the University of California, Irvine. Henry Hirschberg is grateful for the support received from the Norwegian Radiumhospital Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Bahman Anvari oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Madsen, S.J., Baek, SK., Makkouk, A.R. et al. Macrophages as Cell-Based Delivery Systems for Nanoshells in Photothermal Therapy. Ann Biomed Eng 40, 507–515 (2012). https://doi.org/10.1007/s10439-011-0415-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-011-0415-1