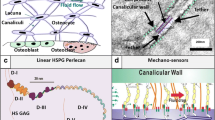
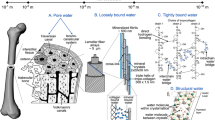
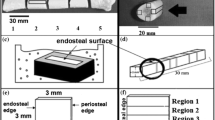
Abstract
In vivo the hydraulic permeability of cortical bone influences the transport of nutrients, waste products and signaling molecules, thus influencing the metabolic functions of osteocytes and osteoblasts. In the current study two hypotheses were tested: the presence of (1) lipids and (2) collagen matrix in the porous compartment of cortical bone restricts its permeability. Our approach was to measure the radial permeability of adult canine cortical bone before and after extracting lipids with acetone-methanol, and before and after digesting collagen with bacterial collagenase. Our results showed that the permeability of adult canine cortical bone was below 4.0 × 10−17 m2, a value consistent with prior knowledge. After extracting lipids, permeability increased to a median value of 8.6 × 10−16 m2. After further digesting with collagenase, permeability increased to a median value of 1.4 × 10−14 m2. We conclude that the presence of both lipids and collagen matrix within the porous compartment of cortical bone restricts its radial permeability. These novel findings suggest that the chemical composition of the tissue matrix within the porous compartment of cortical bone influences the transport and exchange of nutrients and waste products, and possibly influences the metabolic functions of osteocytes and osteoblasts.






Similar content being viewed by others
References
Beno, T., Y. Yoon, S. C. Cowin, and S. P. Fritton. Estimation of bone permeability using accurate microstructural measurements. J. Biomech. 39:2378–2387, 2006.
Berg, R. A. Determination of 3- and 4-hydroxyproline. Methods Enzymol. A 82:372–398, 1982.
Berthiaume, F., and J. A. Frangos. Fluid flow increases membrane permeability to merocyanine 540 in human endothelial cells. Biochim. Biophys. Acta 1191:209–218, 1994.
Black, J., R. Mattson, and E. Korostoff. Haversian osteons: size distribution, internal structure, and orientation. J. Biomed. Mater. Res. 8:299–319, 1974.
Buechner, P. M., R. S. Lakes, C. Swan, and R. A. Brand. A broadband visoelastic spectroscopic study of bovine bone: implications for fluid flow. Ann. Biomed. Eng. 29:719–728, 2001.
Burger, E. H., and J. Klein-Nulend. Mechanotransduction in bone-role of lacunocanalicular network. FASEB J. 13(suppl.):S101–S112, 1999.
Cooper, R. R., J. W. Milgram, and R. A. Robinson. Morphology of the osteon: an electron microscopic study. J. Bone Joint Surg. Am. 48:1239–1271, 1966.
Dillaman, R. M. Movement of ferritin in the 2-day-old chick femur. Anat. Rec. 209:445–453, 1984.
Dirksen, T. R., and G. V. Marinetti. Lipids of bovine Enamel and dentin and human bone. Calc. Tiss. Res. 6:1–10, 1970.
Hillsley, M. V., and J. A. Frangos. Review: bone tissue engineering: the role of interstitial fluid flow. Biotechnol. Bioeng. 43:573–581, 1994.
Irving, J. T., and R. E. Wuthier. Histochemistry and biochemistry of calcification with special reference to the role of lipids. Clin. Orthop. Rel. Res. 56:237–260, 1968.
Johnson, M. W. Behavior of fluid in stressed bone and cellular stimulation. Calcif. Tiss. Int. 36(Suppl 1):S72–S76, 1984.
Klein-Nulend, J., A. van der Plas, C. M. Semeins, N. E. Ajubi, J. A. Frangos, P. J. Nijweide, and E. H. Burger. Sensitivity of osteocytes to biomechanical stress in vitro. FASEB J. 9(5):441–445, 1995.
Knothe Tate, M. L., R. Steck, M. R. Forwood, and P. Niederer. In vivo demonstration of load-induced fluid flow in the rat tibia and its potential implications for processes associated with functional adaptation. J. Exp. Biol. 203(Pt 18):2737–4525, 2000.
Leach, A. A. The lipids of ox compact bone. Biochem. J. 69(3):429–432, 1958.
Li, G. P., J. T. Bronk, K. N. An, and P. J. Kelly. Permeability of cortical bone of canine tibia. Microvasc. Res. 34:302–310, 1987.
McCalden, R. W., J. A. McGeough, M. B. Barker, and C. M. Court-Brown. Age-related changes in tensile properties of cortical bone. The relative importance of changes in porosity, mineralization and microstructure. J. Bone Joint Surg. Am. 75(8):1193–1205, 1993.
Muellar, K. H., A. Trias, and R. D. Ray. Bone density and composition: age-related and pathological changes in water and mineral content. J. Bone Joint Surg. 48-A:140–148, 1966.
Pietrazk, W. S., and J. Woodell-May. The composition of human cortical allograft bone derived from FDA/AATB-screened donor. J. Craniofac. Surg. 16(4):579–585, 2005.
Robinson, L. G., and E. R. Pugh. The determination of serum cholesterol. US Armed Forces Med. J. 9(4):501–507, 1957.
Sander, E. A., and E. A. Nauman. Permeability of musculoskeletal tissues and scaffolding materials: experimental results and theoretical predictions. Crit. Rev. Biomed. Eng. 31(1):1–26, 2003.
Shimko, D. A., and E. A. Nauman. Development and characterization of a porous poly (methacrylate) scaffold with controllable modulus and permeability. J. Biomed. Mater. Res. B Appl. Biomater. 80B:360–369, 2007.
Simonds, W. F., G. Koski, R. A. Streaty, L. M. Hjelmeland, and W. A. Klee. Solubilization of active opiate receptors. Proc. Natl. Acad. Sci. USA 77(8):4623–4627, 1980.
Stegemann, H., and K. Stalder. Determination of hydroxyproline. Clin. Chim. Acta 18:267–273, 1967.
Takahashi, T., S. Ueda, K. Takahashi, and R. O. Scow. pH-dependent multilamellar structures in fetal mouse bone: possible involvement of fatty acids in bone mineralization. Am. J. Physiol. Cell Physiol. 266(35):C590–C600, 1995.
Toroian, D., J. Lim, and P. A. Price. The size exclusion characteristics of type I collagen-implications for the role of noncollagenous bone constituents. J. Biol. Chem. 282(31):22437–22447, 2007.
Wang, L., C. Ciani, S. B. Doty, and S. P. Fritton. Delineating bone’s interstitial fluid pathway in vivo. Bone 34:499–509, 2004.
Weinger, J. M., and M. E. Holtrop. An ultrastructural study of bone cells: the occurrence of microtubules, microfilaments and tight junctions. Calcif. Tiss. Res. 14:15–29, 1974.
Winet, H. A bone fluid flow hypothesis for muscle pump-driven capillary filtration: II proposed role for exercise in erodible scaffold implant incorporation. Eur. Cells Mater. 6:1–11, 2003.
Wuthier, R. E. Lipids of matrix vesicles. Fed. Proc. 35(2):117–121, 1976.
Zaffe, D. Some considerations on biomaterials and bone. Micron 36(7–8):583–592, 2005.
Acknowledgments
Funding for this project was provided in part by the Doctoral Dissertation Defense Program of Cleveland State University. We would like to acknowledge the Cleveland Clinic Musculoskeletal Core Center funded in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, No. 1 P30 AR-050953 for microscopic imaging services; the Kansas Lipidomics Research Center for lipid analyses (supported by NSF grants MCB 0455318 and DBI 0521587, NSF EPSCoR grant EPS-0236913 with matching support from the State of Kansas through Kansas Technology Enterprise Corporation and Kansas State University, and NIH Grant P20 RR16475 from the INBRE program of the National Center for Research Resources); and the Molecular Biology Proteomics Facility at the University of Oklahoma Health Sciences Center for amino acid and amino sugar analyses. We are also grateful to Dr. Matt R. Allen, Assistant Professor in the Department of Anatomy & Cell Biology at the Indiana University School of Medicine, for providing high resolution (3-μm) micro-computed tomography images of our bone wafers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Demin Wen and Caroline Androjna contributed equally as first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wen, D., Androjna, C., Vasanji, A. et al. Lipids and Collagen Matrix Restrict the Hydraulic Permeability Within the Porous Compartment of Adult Cortical Bone. Ann Biomed Eng 38, 558–569 (2010). https://doi.org/10.1007/s10439-009-9858-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-009-9858-z