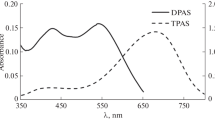
Abstract
Peroxidase-catalyzed oxidation of 2,2-azino-di(3-ethyl-benzthiazolydine-6-sulfonic acid) (ABTS) and 3,3′,5,5′-tetramethylbenzidine (TMB) is activated by tetrazole and its 5-substituted derivatives—5-amino-(AmT), 5-methyl-(MeT), 5-phenyl-(PhT), and 5-CF3-(CF3-T) tetrazoles. In phosphate-citrate or phosphate buffer (pH 6.4 or 7.2; 20°C), the activating effect of tetrazoles on TMB and ABTS oxidation decreased in the series AmT > MeT > T > PhT > CF3-T and T > AmT > MeT > PhT, respectively. The coefficient (degree) of activation (α), expressed in M−1, determined for both substrates and all activators, depended on substrate type, buffer nature, and pH (it increased as pH increased from 6.4 to 7.2). For TMB oxidation, good correlation between logα and the Hammet constants σmeta for m-substituents in the benzene series NH2, CH3, C6H5, and CF3 was found. It is suggested that AmT, MeT, and T can be used as activators of peroxidase-catalyzed oxidation of TMB and ABTS in enzyme immunoassay and designing peroxidase-based biosensors.
Similar content being viewed by others
Abbreviations
- A:
-
Activator
- ABTS:
-
diammonium salt of 2,2-azino-di(3-ethyl-benzthiazolydine-6-sulfonic acid)
- TMB:
-
3,3′,5,5′-tetramethylbenzidine
- T:
-
tetrazole
- AmT:
-
5-aminotetrazole
- MeT:
-
5-methyltetrazole
- PhT:
-
5-phenyltetrazole
- CF3-T:
-
5-CF3-tetrazole
- HP:
-
horseradish peroxidase
- PB:
-
0.06 M phosphate buffer
- PCB:
-
0.06 M phosphate-citrate buffer
- α:
-
coefficient (degree) of activation of peroxidase-catalyzed oxidation of ABTS and TMB
- σmeta :
-
the Hammet constant for meta-substituents in the benzene series
REFERENCES
Polac, J.M. and Van Noorden, S., An Introduction to Immunocytochemistry: Current Techniques and Problems, Oxford: Oxford Univ., 1984. Translated under the title Vvedenie v immunotsitokhimiyu: sovremenye metody i problemy, Moscow: Mir, 1987.
Egorov, A.M., Osipov, A.P., Dzantiev, B.B., and Gavrilova, E.M., Teoriya i praktika immunofermentnogo analiza (Theory and Practice of the Enzyme Immunoassay), Moscow: Vysshaya Shkola, 1991.
Rubtsova, M.Yu., Kovba, G.V., and Egorov, A.M., Biosensor and Bioelectronics, 1998, vol. 13, no.1, pp. 75–85.
Metelitza, D.I. and Karasyova, E.I., Biokhimiya, 2002, vol. 67, no.9, pp. 1265–1272.
Karasyova, E.I., Naumchik, I.V., and Metelitza, D.I., Bioorg. Khim., 2003, vol. 29, no.1, pp. 49–56.
Karasyova, E.I., Naumchik, I.V., and Metelitza, D.I., Biokhimiya, 2003, vol. 68, no.1, pp. 64–73.
Fridovich, I., J. Biol. Chem., 1963, vol. 238, no.12, pp. 3921–3928.
Claiborne, A. and Fridovich, I., Biochemistry, 1979, vol. 18, no.11, pp. 2327–2335.
Lebedeva, O.V., Ugarova, N.N., and Berezin, I.V., Biokhimiya, 1977, vol. 42, no.8, pp. 1372–1380.
Ugarova, N.N., Lebedeva, O.V., Kurilina, T.A., and Berezin, I.V., Biokhimiya, 1977, vol. 42, no.9, pp. 1577–1584.
Lebedeva, O.V., Dombrovskii, V.A., Ugarova, N.N., and Berezin, I.V., Biokhimiya, 1978, vol. 43, no.6, pp. 1024–1033.
Dolmanova, I.F., Shekhovtsova, T.N., and Kutcheryaeva, V.V., Talanta, 1987, vol. 34, no.1, pp. 201–205.
Metelitza, D.I., Savenkova, M.I., and Kurchenko, V.P., Prikl. Biokhim. Mikrobiol., 1987, vol. 23, no.1.
Spravochnik khimika (A Biochemist’s Reference Book), Nikol’skii, B.P, Ed., Leningrad: Khimiya, 1967, vol. 4, p. 919.
Gaponik, P.N., Ivashkevich, O.A., Krasitskii, V.A., Tuzik, A.A., and Lesnikovich, A.I., Zh. Obshch. Khim., 2002, vol. 72, no.9, pp. 1546–1551.
Finnegan, W.G., Henry, R.A., and Lofquist, R., J. Am. Chem. Soc., 1958, vol. 80, no.15, pp. 3908–3911.
Norris, W.P., J. Org. Chem., 1962, vol. 27, no.9, pp 3248–3251.
Re, R., Pellegrini, N., Pannala, A., Yahg, M., and Rice-Evans, C., Free Radical Biol. Med., 1999, vol. 26, nos.9–10, pp. 1231–1237.
Kratkaya khimicheskaya entsiklopediya (Abridged Chemical Encyclopedia), Moscow: Sovetskaya entsiklopediya, 1961, vol. 1, pp. 803–804.
Hammet, L., Physical Organic Chemistry: Reaction Rates, Equilibria and Mechanisms, New York: McGraw-Hill, 1970. Translated under the title Osnovy fizicheskoi organicheskoi khimii, Moscow: Mir, 1972, pp. 448–494.
Koldobskii, G.I., Ostrovskii, V.A., and Gidaspov, G.V., Khim. Geterotsikl. Soedin., 1980, no. 7, pp. 867–879.
Pal’m, V.A., Osnovy kolichestvennoi teorii organicheskikh reaktsii (Basics of Quantitative Theory of Organic Reactions), Leningrad: Khimiya, 1977, pp. 308–338.
Lopyrev, V.A., Larina, L.I., and Vakul’skaya, T.I., Usp. Khim., 1986, vol. 55, no.5, pp. 769–793.
Shchipanov, V.P., Khim. Geterotsikl. Soedin., 1983, no. 8, pp. 1130–1133.
Kim, B.B., Biotekhnologiya peroksidaz rastenii i gribov (Biotechnology of Peroxidases of Plants and Fungi), Itogi Nauki Tekh., Ser.: Biotekhnol., Moscow: VINITI, 1992, vol. 36, pp. 126–146.
Schuller, D.J., Ban, N., van Huystee, R.B., McPherson A., and Poulos, T.L., Structure, 1996, vol. 4, no.3, pp. 311–321.
Gajede, M., Henriksen, A., Schuller, D.J., Poulos, T.L., and Smith, A.T., Plant Peroxidases: Biochemistry and Physiology. Abst. IV Int. Symp., 1996, vol. 4, pp. 311–321.
Gazaryan, I.T., Uporov, I.V., Chubar’, T.A., Fechina, V.A., Mareeva, E.A., and Lagrimini, L.M., Biokhimiya, 1998, vol. 63, no.5, pp. 708–715.
Josephy, P.D., Eling, T., and Mason, R.P., J. Biol. Chem., 1982, vol. 257, no.7, pp. 3669–3675.
Rodrigez-Lopes, J.N., Gilabert, M.A., Tudela, J., Thorneley, R.N.F., and Garcia-Canovas, F., Biochemistry, 2000, vol. 39, no.43, pp. 13201–13209.
Kulys, Ju. and Ziemys, A., BMC Structural Biology, 2001, vol. 1, no.3.
Author information
Authors and Affiliations
Additional information
Translated from Prikladnaya Biokhimiya i Mikrobiologiya, Vol. 41, No. 2, 2005, pp. 148–157.
Original Russian Text Copyright © 2005 by Karasyova, Gaponik, Metelitza.
Rights and permissions
About this article
Cite this article
Karasyova, E.I., Gaponik, P.N. & Metelitza, D.I. Activation of peroxidase-catalyzed oxidation of chromogenic substrates by tetrazole and its 5-substituted derivatives. Appl Biochem Microbiol 41, 129–138 (2005). https://doi.org/10.1007/s10438-005-0022-8
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10438-005-0022-8