Abstract
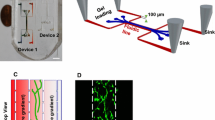
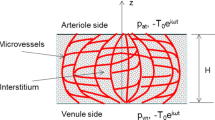
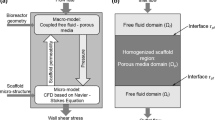
In vitro experiments have shown that subtle fluid flow environment plays a significant role in living biological tissues, while there is no in vivo practical dynamical measurement of the interstitial fluid flow velocity. On the basis of a new finding that capillaries and collagen fibrils in the interosseous membrane form a parallel array, we set up a porous media model simulating the flow field with FLUENT software, studied the shear stress on interstitial cells’ surface due to the interstitial fluid flow, and analyzed the effect of flow on protein space distribution around the cells. The numerical simulation results show that the parallel nature of capillaries could lead to directional interstitial fluid flow in the direction of capillaries. Interstitial fluid flow would induce shear stress on the membrane of interstitial cells, up to 30 Pa or so, which reaches or exceeds the threshold values of cells’ biological response observed in vitro. Interstitial fluid flow would induce nonuniform spacial distribution of secretion protein of mast cells. Shear tress on cells could be affected by capillary parameters such as the distance between the adjacent capillaries, blood pressure and the permeability coefficient of capillary’s wall. The interstitial pressure and the interstitial porosity could also affect the shear stress on cells. In conclusion, numerical simulation provides an effective way for in vivo dynamic interstitial velocity research, helps to set up the vivid subtle interstitial flow environment of cells, and is beneficial to understanding the physiological functions of interstitial fluid flow.
Similar content being viewed by others
References
Knothe Tate, M.L.: Interstitial fluid flow. In: Cowin, S.C. ed. Bone Mechanics Handbook, (2nd edn). CRC Press, Boca Raton, Florida, USA, 22, 1–29 (2002)
Tarbell, J.M., Weinbaum, S., Kamm, R.D.: Cellular fluid mechanics and mechanotransduction. Annals of Biomedical Engineering 33(12), 1719–1723 (2005)
Ng, C.P., Swartz, M.A.: Fibroblast alignment under interstitial fluid flow using a novel 3-D tissue culture model. Am J. Phys iol. Heart Circ. Physiol. 284(5), 1771–1777 (2003)
Casley-Smith, JR., Vincent, A.H.: The quantitative morphology if interstitial tissue channels in some tissues of the rat and rabbit. Tissue and Cell 10(3), 571–584 (1978)
Li, H.Y., Yang, J.F., Chen, M.: Visualized regional hypodermic migration channels of Interstitial fluid in human beings: Are these ancient meridians? The Journal of Alternative and Complementary Medicine 14(6), 621–628 (2008)
Tarbell J.M., Pahakis M.Y.: Mechanotransduction and the glycocalyx. Journal of Internal Medicine 259(4), 339–350 (2006)
Shigeru, T., Tarbell, J.M.: Flow through internal elastic lamina affects shear stress on smooth muscle cells (3D simulations). Am J. Physiol. Heart Circ. Physiol. 282(2), 576–584 (2002)
Fritton S.P., Weinbaum, S.: Fluid and solute transport in bone: flow-induced mechanotransduction. Annule Review of Fluid Mechanics 41, 347–374 (2009)
Riddle, R.C., Donahue, H.J.: From streaming potentials to shear stress: 25 years of bone cell. Journal of Orthopaedic Research 27(1), 143–149 (2009)
Butler, S.L., Kohles S.S., Thielke R.J., et al: Intersititial fluid flow in tendons or ligaments: a porous medium finite element simulation. Medical and Biological Engineering and Computing 35(6), 742–746 (1997)
Chen, C.T., Malkus, D.S., Vanderby, R.: A fiber matrix model for interstitial fluid flow and permeability in ligaments and tendons. Biorheology 35(2), 103–118 (1998)
Pedersen, J.A., Boschetti F., Swartz M.A.: Effects of extra-cellular fiber architecture on cell membrane shear stress in a 3D fibrous matrix. Journal of Biomechanics 40(7), 1484–1492 (2007)
Boardman, K.C., Swartz, M.A.: Interstitial flow as a guide for lymphangiogenesis. Circulation Research 92(7), 801–808 (2003)
Ng C.P., Hins, B., Swartz, M.A.: Interstitial fluid flow induces myofibroblast differentiation and collagen alignment in vitro. Journal of Cell Science 118(20), 4731–4739 (2005)
Vera, R.H., Genov, E., Alvarez, L., et al.: Interstitial fluid flow intensity modulates endothelial sprouting in restricted Src-activated cell clusters during capillary morphogenesis. Tissue Engineering, A 15(1), 175–185 (2009)
Hayward, L.N.M., Morgan, E.F.: Assessment of a mechano-regulation theory of skeletal tissue differentiation in an in vivo model of mechanically induced cartilage formation. Biomech Model Mechanobiol 8(6), 447–455 (2009)
Fei L., Chen H.S., Cai D.H., et al.: Researches and developments of meridian physical basic and function experiments. Chinese Science Bulletin 43(15), 658–672 (2000)
Zhang D., Ding G.H., Shen X.Y., et al.: Role of mast cells in acupuncture effect: a pilot study. Explore: the Journal of Science and Healing 4(3), 170–177 (2008)
Woo S.L.Y., Young E.P.: Structure and function of tendons and ligaments. In: Mow V.C., Hayes W.C. eds. Basic Orthopaedic Biomechanics. Raven Press, New York, 199–244 (1991)
Tsay, R.Y., Weinbaum S.: Viscous flow in a channel with periodic cross-bridging fibers: Exact solution and Brinkman approximation. Journal of Fluid Mechanics 226, 125–148 (1991)
Keener, J., Sneyd, J.: Mathematical Physiology. Springer-Verlag, New York, (1998)
Hu, X., Adamson, R.H., Liu, B., et al.: Starling forces that oppose filtration after tissue oncotic pressure is increased. Am. J. Physiol. Heart Circ. Physiol. 279(4), 1724–1736 (2009)
Gurdon, J.B., Bourillot, P.Y.: Morphogen gradient interpretation. Nature 413, 797–803 (2001)
Fu B.M.: Microvessel permeability and its regulation. Advances in Biomechanics. 231–247 (2001)
Ng, C.P., Helm, C.L.E., Swartz, M.A.: Interstitial flow differentially stimulates blood and lymphatic endothelial cell morphogenesis in vitro. Microvascular Research 68(3), 258–264 (2004)
Ng, C.P., Swartz, M.A.: Mechanisms of interstitial flow induced remodeling of fibroblast-collagen cultures. Annals of Biomedical Engineering 34(3), 446–454 (2006)
Alshihabi, S.N., Chang, Y.S., Frangos, J.A.: Shear stress induced release of PGE2 and PGI2 by vascular smooth muscle cells. Biochemical and Biophysical Research Communications 224(3), 808–814 (1996)
Wang L.N., Ding G.H., Gu Q.B., et al.: Single channel properties of a stretch-sensitive chloride channel in the human mast cell line HMC-1. European Biophysics Journal 39(5), 757–767 (2010)
Wang L.N.: Investigation f mechanical sensitive channels on mast cell. [Ph.D. Thesis]. Shanghai: Fudan University, China, 2010 (in Chinese)
Manes S. Gomez-Mouton C. Lacalle RA. et al.: Mastering time and space: immune cell polarization and chemotaxis. Semin. Immunol. 17(1), 77–86 (2005)
Schwank, G., Basler, K.: Regulation of organ growth by morphogen gradients. Cold Spring Harb Perspect Biol. 2, a001669 (2010)
Author information
Authors and Affiliations
Corresponding author
Additional information
The project was supported by Shanghai Leading Academic Discipline Project (B112 and T0302), Shanghai Science & Technology Development Foundation (09DZ1976600) and Shanghai Rising-Star Program (10QA1406100).
Rights and permissions
About this article
Cite this article
Yao, W., Ding, GH. Interstitial fluid flow: simulation of mechanical environment of cells in the interosseous membrane. Acta Mech Sin 27, 602–610 (2011). https://doi.org/10.1007/s10409-011-0439-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10409-011-0439-7