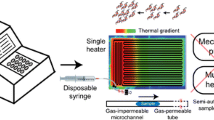
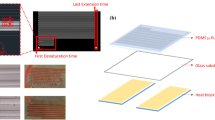
Abstract
Polymerase chain reaction (PCR) is commonly used to amplify and quantify the nucleic acid segments typically using a benchtop thermocycler. To automate, integrate, and miniaturize the PCR process, several strategies have been studied in a microfluidic environment. Among them, continuous-flow-based microfluidic PCR allows fast thermal cycling using minuscule volume, in a minimal reaction time with multiplexing. The objective was to develop a microfluidic device with a soft-lithographically fabricated continuous-flow serpentine microchannel for DNA amplification executed on a separately designed portable, easy-to-use, low-cost, automated, and miniaturized temperature controller platform (TCP). Direct laser writer (DLW) was used for developing a master on glass using a dry-film photoresist (DFR). Further, a PDMS-based microfluidic device, with dimensions 30 (L) × 0.32 mm2 (W) × 35 µm (H), was developed which was bonded on glass using oxygen plasma. The portable device exhibits key features of live data streaming using an IoT platform enabling easy data accessing, monitoring and storage onto the smartphone. The temperature sensitivity of the device was ± 0.5 °C and the maximum achievable temperature was 300 °C. The microfluidic device was placed on TCP. A 20 µL of reaction volume was introduced using an automated syringe pump at various flow rates. As a proof-of-concept, the rat GAPDH gene of the 594-base pair was successfully amplified on the proposed platform which was validated using the gel electrophoresis method. Finally, the results obtained from the proposed device were compared with the conventional thermocycler which showed promising performance and novelty exists in the significant reduction of required amplification time with good device efficiency and low-power consumption.











Similar content being viewed by others
References
Azizipour N, Avazpour R, Rosenzweig DH, Sawan M, Ajji A (2020) Evolution of biochip technology: a review from lab-on-a-chip to organ-on-a-chip. Micromachines 11(6):1–15. https://doi.org/10.3390/mi11060599
Beaulieu LY, Logan ER, Gering KL, Dahn JR (2017) An automated system for performing continuous viscosity versus temperature measurements of fluids using an Ostwald viscometer. Rev Sci Instrum. https://doi.org/10.1063/1.4990134
Bhaiyya M, Pattnaik PK, Goel S (2021) Electrochemiluminescence sensing of vitamin B 12 using laser-induced graphene based bipolar and single electrodes in a 3D-printed portable system. Microfluid Nanofluid 25(41):1–8. https://doi.org/10.1007/s10404-021-02442-x
Bruijns B, van Asten A, Tiggelaar R, Gardeniers H (2016) Microfluidic devices for forensic DNA analysis: a review. Biosensors 6(3):1–35. https://doi.org/10.3390/bios6030041
Cho CH, Cho W, Ahn Y, Hwang SY (2007) PDMS–glass serpentine microchannel chip for time domain PCR with bubble suppression in sample injection. J Micromech Microeng 17(9):1810–1817. https://doi.org/10.1088/0960-1317/17/9/009
Cui P, Wang S (2019) Application of microfluidic chip technology in pharmaceutical analysis: a review. J Pharm Anal 9(4):238–247. https://doi.org/10.1016/j.jpha.2018.12.001
Cui F, Rhee M, Singh A, Tripathi A (2015) Microfluidic sample preparation for medical diagnostics. Annu Rev Biomed Eng 17:267–286. https://doi.org/10.1146/annurev-bioeng-071114-040538
Ding X, Srinivasan B, Tung S (2015) Development and applications of portable biosensors. J Lab Autom 20(4):365–389. https://doi.org/10.1177/2211068215581349
Dungchai W, Chailapakul O, Henry CS (2009) Electrochemical detection for paper-based microfluidics. Anal Chem 81(14):5821–5826. https://doi.org/10.1021/ac9007573
Erickson D, Li D (2004) Integrated microfluidic devices. Anal Chim Acta 507(1):11–26. https://doi.org/10.1016/j.aca.2003.09.019
Etchart I, Chen H, Dryden P, Jundt J, Harrison C, Hsu K, Marty F, Mercier B (2008) MEMS sensors for density-viscosity sensing in a low-flow microfluidic environment. Sens Actuators A Phys 141(2):266–275. https://doi.org/10.1016/j.sna.2007.08.007
Fernández-Carballo BL, McGuiness I, McBeth C, Kalashnikov M, Borrós S, Sharon A, Sauer-Budge AF (2016) Low-cost, real-time, continuous flow PCR system for pathogen detection. Biomed Microdevices 18(2):1–10. https://doi.org/10.1007/s10544-016-0060-4
Giordano BC, Ferrance J, Swedberg S, Hühmer AFR, Landers JP (2001) Polymerase chain reaction in polymeric microchips: DNA amplification in less than 240 seconds. Anal Biochem 291(1):124–132. https://doi.org/10.1006/abio.2000.4974
Hartig SM (2013) Basic image analysis and manipulation in imageJ. Curr Protoc Mol Biol. https://doi.org/10.1002/0471142727.mb1415s102
Hu J, Cui X, Gong Y, Xu X, Gao B, Wen T, Lu TJ, Xu F (2016) Portable microfluidic and smartphone-based devices for monitoring of cardiovascular diseases at the point of care. Biotechnol Adv 34(3):305–320. https://doi.org/10.1016/j.biotechadv.2016.02.008
Kopp MU, Luechinger MB, Manz A (1998) Continuous flow PCR on a chip. Micro Total Anal Syst 98:7–10. https://doi.org/10.1007/978-94-011-5286-0_2
Kulkarni MB, Goel S (2020a) Microfluidic devices for synthesizing nanomaterials—a review. Nano Express 1(1):1–30. https://doi.org/10.1088/2632-959X/abcca6
Kulkarni MB, Goel S (2020b) Advances in continuous-flow based microfluidic PCR devices—a review. Eng Res Express. https://doi.org/10.1088/2631-8695/abd287
Kulkarni MB, Enaganti PK, Amreen K, Goel S (2020a) Integrated temperature controlling platform to synthesize ZnO nanoparticles and its deposition on Al-foil for biosensing. IEEE Sens J. https://doi.org/10.1109/JSEN.2021.3053642
Kulkarni MB, Yashas PKE, Amreen K, Goel S (2020b) Internet of Things enabled portable thermal management system with microfluidic platform to synthesize MnO2 nanoparticles for electrochemical sensing. Nanotechnology 31(42):1–8. https://doi.org/10.1088/1361-6528/ab9ed8
Kulkarni MB, Enaganti KV, Khairunnisa P, Jayabalan A (2021a) Smartphone enabled miniaturized temperature controller platform to synthesize nio/cuo nanoparticles for electrochemical sensing and nanomicelles for ocular drug delivery applications. Biomed Microdevices. https://doi.org/10.1007/s10544-021-00567-y
Kulkarni MB, Goyal S, Dhar A, Sriram D, Goel S (2021b) Miniaturized and IoT enabled continuous-flow based microfluidic PCR device for DNA amplification. IEEE Trans Nanobiosci 1241:1–8. https://doi.org/10.1109/tnb.2021.3092292
Law JWF, Mutalib NSA, Chan KG, Lee LH (2014) Rapid methods for the detection of foodborne bacterial pathogens: principles, applications, advantages and limitations. Front Microbiol 5:1–20. https://doi.org/10.3389/fmicb.2014.00770
Li Y, Zhang C, Xing D (2011) Fast identification of foodborne pathogenic viruses using continuous-flow reverse transcription-PCR with fluorescence detection. Microfluid Nanofluid 10(2):367–380. https://doi.org/10.1007/s10404-010-0675-3
Ma SY, Chiang YC, Hsu CH, Chen JJ, Hsu CC, Chao AC, Lin YS (2019) Peanut detection using droplet microfluidic polymerase chain reaction device. J Sens. https://doi.org/10.1155/2019/4712084
Manage DP, Morrissey YC, Stickel AJ, Lauzon J, Atrazhev A, Acker JP, Pilarski LM (2011) On-chip PCR amplification of genomic and viral templates in unprocessed whole blood. Microfluid Nanofluid 10(3):697–702. https://doi.org/10.1007/s10404-010-0702-4
Mavraki E, Moschou D, Kokkoris G, Vourdas N, Chatzandroulis S, Tserepi A (2011) A continuous flow μPCR device with integrated microheaters on a flexible polyimide substrate. Procedia Eng 25:1245–1248. https://doi.org/10.1016/j.proeng.2011.12.307
Mullis K, Faloona F, Scharf S, Saiki R, Horn G, Erlich H (1992) Specific enzymatic amplification of DNA in vitro: the polymerase chain reaction. 1986. Biotechnology 24(1):17–27
Nagai H, Fuchiwaki Y (2015) Portable microfluidic system for rapid genetic testing. Electron Commun Jpn 98(12):1–6. https://doi.org/10.1002/ecj.11753
Natarajan G, Humenik JN (2006) 3D ceramic microfluidic device manufacturing. J Phys Conf Ser 34(1):533–539. https://doi.org/10.1088/1742-6596/34/1/088
Northrup MA, Gonzalez C, Lehew S, Hills R (1995a) Development of a PCR-microreactor. Micro Total Anal Syst. https://doi.org/10.1007/978-94-011-0161-5_13
Northrup MA, Gonzalez C, Hadley D, Hills RF, Landre P, Lehew S, Saiki R, Sninsky JJ, Watson R, Watson R (1995b) MEMS-based miniature DNA analysis system. In: Eurosensors IX, the 14th international conference on solid state sensors, actuators, vol 1, pp 764–767
Ohno KI, Tachikawa K, Manz A (2008) Microfluidics: applications for analytical purposes in chemistry and biochemistry. Electrophoresis 29(22):4443–4453. https://doi.org/10.1002/elps.200800121
Pal A, Kulkarni MB, Gupta H, Ponnalagu RN, Dubey SK, Goel S (2021) Portable and autonomous device for real-time colorimetric detection: validation for phosphorous and nitrite detection. Sens Actuators A Phys 330:112896. https://doi.org/10.1016/j.sna.2021.112896
Pol R, Céspedes F, Gabriel D, Baeza M (2017) Microfluidic lab-on-a-chip platforms for environmental monitoring. TrAC Trends Anal Chem 95:62–68. https://doi.org/10.1016/j.trac.2017.08.001
Puneeth SB, Kulkarni MB, Goel S (2021) Microfluidic viscometers for biochemical and biomedical applications: a review. Eng Res Express 3:1–29. https://doi.org/10.1088/2631-8695/abfd47
Qiu X, Il Shu J, Baysal O, Wu J, Qian S, Ge S, Li K, Ye X, Xia N, Yu D (2019) Real-time capillary convective PCR based on horizontal thermal convection. Microfluid Nanofluid 23(3):1–8. https://doi.org/10.1007/s10404-019-2207-0
Ren K, Chen Y, Wu H (2014) New materials for microfluidics in biology. Curr Opin Biotechnol 25:78–85. https://doi.org/10.1016/j.copbio.2013.09.004
Riesch C, Reichel EK, Keplinger F, Jakoby B (2008) Characterizing vibrating cantilevers for liquid viscosity and density sensing. J Sens 2008:1–9. https://doi.org/10.1155/2008/697062
Snodgrass R, Gardner A, Jiang Li, Cheng Fu, Cesarman E, Erickson D (2016) KS-Detect–validation of solar thermal PCR for the diagnosis of Kaposi’s sarcoma using pseudo-biopsy samples. PLoS ONE 11(1):e0147636. https://doi.org/10.1371/journal.pone.0147636
Srikanth S, Dudala S, Raut S, Dubey SK, Ishii I, Javed A, Goel S (2020) Optimization and characterization of direct UV laser writing system for microscale applications. J Micromech Microeng. https://doi.org/10.1088/1361-6439/ab92ea
Tachibana H, Saito M, Shibuya S, Tsuji K, Miyagawa N, Yamanaka K, Tamiya E (2015) On-chip quantitative detection of pathogen genes by autonomous microfluidic PCR platform. Biosens Bioelectron 74:725–730. https://doi.org/10.1016/j.bios.2015.07.009
Tiwari SK, Bhat S, Mahato KK (2020) Design and fabrication of low-cost microfluidic channel for biomedical application. Sci Rep 10(1):1–14. https://doi.org/10.1038/s41598-020-65995-x
Zhang Y, Jiang HR (2016) A review on continuous-flow microfluidic PCR in droplets: advances, challenges and future. Anal Chim Acta 914:7–16. https://doi.org/10.1016/j.aca.2016.02.006
Acknowledgements
The authors are thankful to the Centre for Human Disease Research (CHDR), BITS-Pilani, Hyderabad Campus for providing financial support during this work. Madhusudan B Kulkarni is thankful to Tata Consultancy Services (TCS) for providing scholarship during this project. The authors are thankful to Mr. Srashti Gopal Goyal, Department of Pharmacy, BITS-Pilani, Hyderabad campus, for providing the samples and helping in the experimental setup.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kulkarni, M.B., Goel, S. Miniaturized DNA amplification platform with soft-lithographically fabricated continuous-flow PCR microfluidic device on a portable temperature controller. Microfluid Nanofluid 25, 69 (2021). https://doi.org/10.1007/s10404-021-02473-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-021-02473-4