Abstract
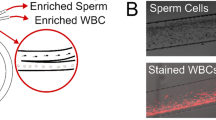
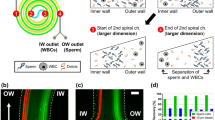
Sperm preparation is a necessary step in all methods of assisted reproductive technology. Here, we demonstrate the utility of a microfluidic spiral channel device for preparation of sperm for intrauterine insemination (IUI) directly from semen samples. After optimization of operating conditions, we use data from 130 washed semen samples to show that the microfluidic chip can retain 89% of all sperm cells and 90% of motile sperm cells, while removing 40% of the fluid volume and 82% of white blood cells. Then, we show that, when combined with a centrifugal wash, the microfluidic chip can be used in a protocol for sperm preparation. This protocol is capable of selecting 40–70% of motile sperm in 15 min. Compared to the clinical gold standard, density gradient centrifugation, this offers a higher sperm recovery, a gentler approach, and reduced reagent costs.






Similar content being viewed by others
References
Agarwal A, Gupta S, Sharma R (2016) Andrological evaluation of male infertility. Springer, Cham, pp 109–112
Ainsworth C, Nixon B, Aitken RJ (2005) Development of a novel electrophoretic system for the isolation of human spermatozoa. Hum Reprod 20:2261–2270
Aitken R et al (2014) Potential importance of transition metals in the induction of DNA damage by sperm preparation media. Hum Reprod 29:2136–2147
Asghar W et al (2014) Selection of functional human sperm with higher DNA integrity and fewer reactive oxygen species. Adv Healthc Mater 3:1671–1679
Behr B, Demirci U (2016) Microfluidic sperm sorting device for selection of functional human sperm for IUI application. Fertil Steril 105:e17–e18
Berendsen JT, Eijkel JC, Wetzels AM, Segerink LI (2019) Separation of spermatozoa from erythrocytes using their tumbling mechanism in a pinch flow fractionation device. Microsyst Nanoeng 5:24
Boivin J, Bunting L, Collins JA, Nygren KG (2007) International estimates of infertility prevalence and treatment-seeking: potential need and demand for infertility medical care. Hum Reprod 22:1506–1512
Cho BS et al (2003) Passively driven integrated microfluidic system for separation of motile sperm. Anal Chem 75:1671–1675
Di Carlo D, Irimia D, Tompkins RG, Toner M (2007) Continuous inertial focusing, ordering, and separation of particles in microchannels. Proc Natl Acad Sci 104:18892–18897
Huang H-Y et al (2014) Motile human sperm sorting by an integrated microfluidic system. J Nanomed Nanotechnol 5:1
Hur SC, Choi S-E, Kwon S, Carlo DD (2011) Inertial focusing of non-spherical microparticles. Appl Phys Lett 99:044101
Jafek A et al (2019) PD34-07 a novel microfluidic device to improve sperm searching in microdissection TESE samples. J Urol 201:e647–e648
Jenkins T et al (2017) Rapid microfluidic sperm isolation from microtese samples in men with non-obstructive azoospermia. Fertil Steril 108:e244
Kuntaegowdanahalli SS, Bhagat AAS, Kumar G, Papautsky I (2009) Inertial microfluidics for continuous particle separation in spiral microchannels. Lab Chip 9:2973–2980
Liu N, Petchakup C, Tay HM, Li KHH, Hou HW (2019) Applications of microfluidic systems in biology and medicine. Springer, Singapore, pp 99–150
Malvezzi H, Sharma R, Agarwal A, Abuzenadah AM, Abu-Elmagd M (2014) Sperm quality after density gradient centrifugation with three commercially available media: a controlled trial. Reprod Biol Endocrinol 12:121
Masaeli M et al (2012) Continuous inertial focusing and separation of particles by shape. Phys Rev X 2:031017
Muratori M et al (2016) Variation of DNA fragmentation levels during density gradient sperm selection for assisted reproduction techniques: a possible new male predictive parameter of pregnancy? Medicine (Baltimore) 95(20):e3624. https://doi.org/10.1097/MD.0000000000003624
Norris JV et al (2009) Acoustic differential extraction for forensic analysis of sexual assault evidence. Anal Chem 81:6089–6095
Nosrati R et al (2014) Rapid selection of sperm with high DNA integrity. Lab Chip 14:1142–1150
Ombelet W, Dhont N, Thijssen A, Bosmans E, Kruger T (2014) Semen quality and prediction of IUI success in male subfertility: a systematic review. Reprod Biomed Online 28:300–309
Rosales-Cruzaley E, Cota-Elizondo P, Sánchez D, Lapizco-Encinas BH (2013) Sperm cells manipulation employing dielectrophoresis. Bioprocess Biosyst Eng 36:1353–1362
Ruiter-Ligeti J, Agbo C, Dahan M (2015) The impact of semen processing on sperm quality and pregnancy rates with intrauterine inseminations. Fertil Steril 104:e246
Samuel R et al (2018) Microfluidic—based sperm sorting and analysis for treatment of male infertility. Transl Androl Urol 7:S336
Samuel R et al (2020) Microfluidic system for rapid isolation of sperm from microdissection TESE specimens. Urology 140:70–76. https://doi.org/10.1016/j.urology.2019.12.053
Son J et al (2015) Non-motile sperm cell separation using a spiral channel. Anal Methods 7:8041–8047
Son J, Samuel R, Gale BK, Carrell DT, Hotaling JM (2017) Separation of sperm cells from samples containing high concentrations of white blood cells using a spiral channel. Biomicrofluidics 11:054106
Son J, Jafek AR, Carrell DT, Hotaling JM, Gale BK (2019) Sperm-like-particle (SLP) behavior in curved microfluidic channels. Microfluid Nanofluid 23:4
Wu L, Guan G, Hou HW, Bhagat AAS, Han J (2012) Separation of leukocytes from blood using spiral channel with trapezoid cross-section. Anal Chem 84:9324–9331
Wu J-K et al (2017) High-throughput flowing upstream sperm sorting in a retarding flow field for human semen analysis. Analyst 142:938–944
Xiang N, Ni Z (2015) High-throughput blood cell focusing and plasma isolation using spiral inertial microfluidic devices. Biomed Microdevice 17:110
Yalti S, Gürbüz B, Sezer H, Celik S (2004) Effects of semen characteristics on IUI combined with mild ovarian stimulation. Arch Androl 50:239–246
Zhou J, Papautsky I (2013) Fundamentals of inertial focusing in microchannels. Lab Chip 13:1121–1132
Acknowledgements
AJ acknowledges funding by the Graduate Research Fellowship, National Science Foundation Grant 1747505. Research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health under Award Number R44HD095355. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
AJ: collected and analyzed experimental data and wrote the main manuscript text; HF: designed and fabricated microfluidic chips; DB: assisted in sample acquisition and experimentation; BG and RS: directed the research; KA and TJ: provided medical supervision. All authors have read the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
BG, RS, KA, and TJ have partial ownership of Nanonc, Inc.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jafek, A., Feng, H., Broberg, D. et al. Optimization of Dean flow microfluidic chip for sperm preparation for intrauterine insemination. Microfluid Nanofluid 24, 60 (2020). https://doi.org/10.1007/s10404-020-02366-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02366-y