Abstract
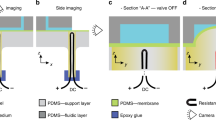
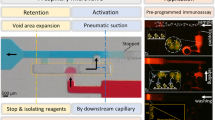
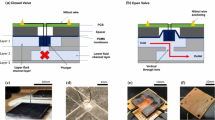
A multilayer capillary polymeric microfluidic device integrated with three normally closed electrowetting valves for timed fluidic delivery was developed. The microfluidic channel consisted two flexible layers of poly (ethylene terephthalate) bonded by a pressure-sensitive adhesive spacer tape. Channels were patterned in the spacer tape using laser ablation. Each valve contained two inkjet-printed silver electrodes in series. Capillary flow within the microchannel was stopped at the second electrode which was modified with a hydrophobic monolayer (valve closed). When a potential was applied across the electrodes, the hydrophobic monolayer became hydrophilic and allowed flow to continue (valve opened). The relationship between the actuation voltage, the actuation time, and the distance between two electrodes was performed using a microfluidic chip containing a single microchannel design. The results showed that a low voltage (4.5 V) was able to open the valve within 1 s when the distance between two electrodes was 1 mm. Increased voltages were needed to open the valves when the distance between two electrodes was increased. Additionally, the actuation time required to open the valve increased when voltage was decreased. A multichannel device was fabricated to demonstrate timed fluid delivery between three solutions. Our electrowetting valve system was fabricated using low-cost materials and techniques, can be actuated by a battery, and can be integrated into portable microfluidic devices suitable for point-of-care analysis in resource-limited settings.






Similar content being viewed by others
References
Ando B, Baglio S (2011) Instrumentation notes inkjet-printed sensors: a useful approach for low cost, rapid prototyping. IEEE Instrum Meas Mag 14:36–40
Dai W, Zhao Y (2008) An electrowetting model for rough surfaces under low voltage. J Adhes Sci Technol 22:217–229
Fukuda K, Sekine T, Kumaki D, Tokito S (2013) Profile control of inkjet printed silver electrodes and their application to organic transistors. ACS Appl Mater Interfaces 5:3916–3920
Gervais L, Hitzbleck M, Delamarche E (2011) Capillary-driven multiparametric microfluidic chips for one-step immunoassays. Biosens Bioelectron 27:64–70
Holmes D, Gawad S (2010) The application of microfluidics in biology. In: Hughes MP, Hoettges KF (eds) Microengineering in Biotechnology. Humana Press, pp 55–80
Jong WR, Kuo TH, Ho SW, Chiu HH, Peng SH (2007) Flows in rectangular microchannels driven by capillary force and gravity. Int Commun Heat Mass Transf 34:186–196
Kwon O, Kim H, Ko H, Lee J, Lee B, Jung C, Choi J, Shin K (2013) Fabrication and characterization of inkjet-printed carbon nanotube electrode patterns on paper. Carbon 58:116–127
Lee A (2013) The third decade of microfluidics. Lab Chip 13:1660–1661
Lee Y, Kim S, Lee K, Myung NV, Choa Y (2013) Inkjet printed transparent conductive films using water-dispersible single-walled carbon nanotubes treated by UV/ozone irradiation. Thin Solid Films 536:160–165
Liedert R, Amundsen LK, Hokkanen A, Maki M, Aittakorpi A, Pakanen M, Scherer JR, Mathies RA, Kurkinen M, Uusitalo S, Hakalahti L, Nevanen TK, Siitari H, Soderlund H (2012) Disposable roll-to-roll hot embossed electrophoresis chip for detection of antibiotic resistance gene mecA in bacteria. Lab Chip 12:333–339
Mérian T, He F, Yan H, Chu D, Talbert JN, Goddard JM, Nugen SR (2012) Development and surface characterization of an electrowetting valve for capillary-driven microfluidics. Colloids Surf A Physicochem Eng Asp 414:251–258
Noh J, Yeom D, Lim C, Cha H, Han J, Kim J, Park Y, Subramanian V, Cho G (2010) Scalability of roll-to-roll gravure-printed electrodes on plastic foils. IEEE Trans Electron Packaging Manuf 33:275–283
Novo P, Volpetti F, Chu V, Conde JP (2013) Control of sequential fluid delivery in a fully autonomous capillary microfluidic device. Lab Chip 13:641–645
Nugen SR, Asiello PJ, Connelly JT, Baeumner AJ (2009) PMMA biosensor for nucleic acids with integrated mixer and electrochemical detection. Biosens Bioelectron 24:2428–2433
Pennathur S (2008) Flow control in microfluidics: are the workhorse flows adequate? Lab Chip 8:383–387
Perelaer J, Schubert US (2013) Novel approaches for low temperature sintering of inkjet-printed inorganic nanoparticles for roll-to-roll (R2R) applications. J Mater Res 28:564–573
Pollack M, Shenderov A, Fair R (2002) Electrowetting-based actuation of droplets for integrated microfluidics. Lab Chip 2:96–101
Rivet C, Lee H, Hirsch A, Hamilton S, Lu H (2011) Microfluidics for medical diagnostics and biosensors. Chem Eng Sci 66:1490–1507
Saeki F, Baum J, Moon H, Yoon JY, Kim CJ, Garrell RL (2001) Electrowetting on dielectrics: reducing voltage requirements for microfluidics. Abstr Pap Am Chem Soc 222:8-PMSE
Satoh W, Yokomaku H, Hosono H, Ohnishi N, Suzuki H (2008) Electrowetting-based valve for the control of the capillary flow. J Appl Phys 103:034903
Schulte TH, Bardell RL, Weigl BH (2002) Microfluidic technologies in clinical diagnostics. Clin Chim Acta 321:1–10
Srinivasan V, Pamula VK, Fair RB (2004) An integrated digital microfluidic lab-on-a-chip for clinical diagnostics on human physiological fluids. Lab Chip 4:310–315
Stone HA, Stroock AD, Ajdari A (2004) Engineering flows in small devices: microfluidics toward a lab-on-a-chip. Annu Rev Fluid Mech 36:381–411
Swickrath MJ, Shenoy S, Mann Jr JA, Belcher J, Kovar R, Wnek GE (2008) The design and fabrication of autonomous polymer-based surface tension-confined microfluidic platforms. Microfluid Nanofluid 4:601–611
Swickrath MJ, Burns SD, Wnek GE (2009) Modulating passive micromixing in 2-D microfluidic devices via discontinuities in surface energy. Sens Actuators B Chem 140:656–662
Vig AL, Makela T, Majander P, Lambertini V, Ahopelto J, Kristensen A (2011) Roll-to-roll fabricated lab-on-a-chip devices. J Micromech Microeng 21:035006
Walker SB, Lewis JA (2012) Reactive silver inks for patterning high-conductivity features at mild temperatures. J Am Chem Soc 134:1419–1421
Yager P, Edwards T, Fu E, Helton K, Nelson K, Tam MR, Weigl BH (2006) Microfluidic diagnostic technologies for global public health. Nature 442:412–418
Yager P, Domingo GJ, Gerdes J (2008) Point-of-care diagnostics for global health. Annu Rev Biomed Eng 10:107–144
Ziaie B, Baldi A, Lei M, Gu Y, Siegel R (2004) Hard and soft micromachining for BioMEMS: review of techniques and examples of applications in microfluidics and drug delivery. Adv Drug Deliv Rev 56:145–172
Acknowledgments
This project was supported in part by the UMass Amherst Center for Hierarchical Manufacturing, a nanoscience shared facility funded by the National Science Foundation under NSF Grant #CMMI-1025020. The authors thank undergraduate researcher Elsa Zhao for her assistance with silver ink synthesis. The authors would also like to thank S. Brett Walker and Professor Jennifer A. Lewis for assistance with waveform of reactive silver ink.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MPEG 34012 kb)
Rights and permissions
About this article
Cite this article
He, F., Nugen, S.R. Automating fluid delivery in a capillary microfluidic device using low-voltage electrowetting valves. Microfluid Nanofluid 16, 879–886 (2014). https://doi.org/10.1007/s10404-013-1317-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-013-1317-3