Abstract
Purpose
To reveal the ability of S-Map strain elastography to diagnose fibrosis in nonalcoholic fatty liver disease (NAFLD) and to compare its diagnostic ability with that of shear wave elastography (SWE).
Methods
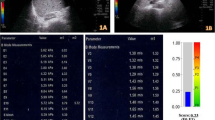
Participants were patients with NAFLD who were scheduled to undergo liver biopsy at our institution between 2015 and 2019. A GE Healthcare LOGIQ E9 ultrasound system was used. For S-Map, the right lobe of the liver was visualized in the section where the heartbeat was detected by right intercostal scanning, a 4 × 2-cm region of interest (ROI) was defined at 5 cm from the liver surface, and ROI strain images were acquired. Measurements were repeated six times, with the average taken as the S-Map value. Correlations of S-Map and SWE values with fibrosis stage determined by liver biopsy were analyzed using multiple comparisons. The diagnostic performance of S-Map for fibrosis staging was assessed using receiver operating characteristic curves.
Results
In total, 107 patients (65 men, 42 women; mean age 51 ± 14 years) were analyzed. The S-Map value by fibrosis stage was 34.4 ± 10.9 for F0, 32.9 ± 9.1 for F1, 29.5 ± 5.6 for F2, 26.7 ± 6.0 for F3, and 22.8 ± 4.19 for F4. By fibrosis stage, the SWE value was 1.27 ± 0.25 for F0, 1.39 ± 0.20 for F1, 1.59 ± 0.20 for F2, 1.64 ± 0.17 for F3, and 1.88 ± 0.19 for F4. The diagnostic performance of S-Map (measured by area under the curve) was 0.75 for F2, 0.80 for F3, and 0.85 for F4. The diagnostic performance of SWE (measured by area under the curve) was 0.88 for F2, 0.87 for F3, and 0.92 for F4.
Conclusion
S-Map strain elastography was inferior to SWE in terms of ability to diagnose fibrosis in NAFLD.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
National Institutes of Health Consensus Development Conference Statement. Management of hepatitis C 2002 (June 10–12, 2002). Gastroenterology. 2002;123:2082–99.
Castera L. Noninvasive methods to assess liver disease in patients with hepatitis B or C. Gastroenterology. 2012;142:1293–302.
Cadranel JF. Good clinical practice guidelines for fine needle aspiration biopsy of the liver: past, present and future. Gastroenterol Clin Biol. 2002;26:823–4.
Wakui N, Takayama R, Kanekawa T, et al. Usefulness of arrival time parametric imaging in evaluating the degree of liver disease progression in chronic hepatitis C infection. J Ultrasound Med. 2012;31:373–82.
Wakui N, Nagai H, Yoshimine N, et al. Flash imaging used in the post-vascular phase of contrast-enhanced ultrasonography is useful for assessing the progression in patients with hepatitis C virus-related liver disease. Ultrasound Med Biol. 2019;45:1654–62.
Mariappan YK, Giaser KJ, Ehman RL. Magnetic resonance elastography: a review. Clin Anat. 2010;23:497–511.
Glaser KJ, Manduca A, Ehman RL. Review of MR elastography applications and recent developments. J Magn Reson Imaging. 2012;36:757–74.
Igarashi H, Shigiyama F, Wakui N, et al. Whole hepatic lipid volume quantification and color mapping by multi-slice and multi-point magnetic resonance imaging. Hepatol Res. 2019;49:1374–85.
Wong VS, Hughes V, Trull A, et al. Serum hyaluronic acid is a useful marker of liver fibrosis in chronic hepatitis C virus infection. J Viral Hepat. 1998;5:187–92.
Rockey DC, Bissel DM. Noninvasive measures of liver fibrosis. Hepatology. 2006;43:113–20.
Yamada N, Sanada Y, Tashiro M, et al. Serum Mac-2 binding protein glycosylation isomer predicts grade F4 liver fibrosis in patients with biliary atresia. J Gastroenterol. 2017;52:245–52.
Wai CT, Greenson JK, Fontana RJ, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;8:518–26.
Vallet-Pichard A, Mallet V, Nalpas B, et al. FIB-4: an inexpensive and accurate marker of fibrosis in HCV infection. Comparison with liver biopsy and fibrotest. Hepatology. 2007;46:32–6.
Ferraioli G, Filice C, Castera L, et al. WFUMB guidelines and recommendations for clinical use of ultrasound elastography: Part 3: liver. Ultrasound Med Biol. 2015;41:1161–79.
Kudo M, Shiina T, Moriyasu F, et al. JSUM ultrasound elastography practice guidelines: liver. J Med Ultrason. 2013;40:325–57.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American association for the study of liver disease. Hepatology. 2018;67:328–57.
Abe T, Kuroda H, Fujiwara Y, et al. Accuracy of 2D shear wave elastography in the diagnosis of liver fibrosis in patients with chronic hepatitis C. J Clin Ultrasound. 2018;46:319–27.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Matteoni CA, Younossi ZM, Gramlich T, et al. Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology. 1999;116:1413–9.
Petta S, Muratore C, Craxi A. Non-alcoholic fatty liver disease pathogenesis: the present and the future. Dig Liver Dis. 2009;41:615–25.
Angulo P, Kleiner DE, Dam-Larsen S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes off patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149:389-97.e10.
Hagström H, Nasr P, Ekstedt M, et al. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J Hepatol. 2017;67:1265–73.
Estes C, Anstee QM, Arias-Loste MT, et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J Hepatol. 2018;69:896–904.
Yoneda M, Thomas E, Sclair SN, et al. Supersonic shear imaging and transient elastography with the XL probe accurately detect fibrosis in overweight or obese patients with chronic liver disease. Clin Gastroenterol Hepatol. 2015;13:1502-9.e5.
Gaia S, Carenzi S, Barilli AL, et al. Reliability of transient elastography for the detection of fibrosis in non-alcoholic fatty liver disease and chronic viral hepatitis. J Hepatol. 2011;54:64–71.
Petta S, Di Marco V, Cammà C, et al. Reliability of liver stiffness measurement in non-alcoholic fatty liver disease: the effects of body mass index. Aliment Pharmacol Ther. 2011;33:1350–60.
Wong VWS, Vergniol J, Wong GLH, et al. Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology. 2010;51:454–62.
Yoneda M, Yoneda M, Mawatari H, et al. Noninvasive assessment of liver fibrosis by measurement of stiffness in patients with nonalcoholic fatty liver disease (NAFLD). Dig Liver Dis. 2008;40:371–8.
Orlacchio A, Bolacchi F, Antonicoli M, et al. Liver elasticity in NASH patients evaluated with Real-time elastography (RTE). Ultrasound Med Biol. 2012;38:537–44.
Ochi H, Hirooka M, Koizumi Y, et al. Real-time tissue elastography for evaluation of hepatic fibrosis and portal hypertension in nonalcoholic fatty liver fibrosis. Hepatology. 2012;56:1271–8.
Cassinotto C, Boursier J, de Lédinghen V, et al. Liver stiffness in nonalcoholic fatty liver disease: a comparison of supersonic shear imaging, FibroScan, and ARFI with liver biopsy. Hepatology. 2016;63:1817–27.
Furlan A, Tublin ME, Yu L, et al. Comparison of 2D shear wave elastography, transient elastography, and MR elastography for the diagnosis of fibrosis in patients with nonalcoholic fatty liver disease. AJR Am J Roentgenol. 2020;214:W20–6.
Acknowledgements
We thank Shunichirou Tanigawa, Takuma Oguri, and Naohisa Kamiyama for providing advice on the S-Map strain elastography technique.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
YO, NW, HN, and TM declare that they have no conflicts of interest.
Ethical approval
All the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. This study was approved by the Ethics Committee of Toho University Omori Medical Center.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ogino, Y., Wakui, N., Nagai, H. et al. Comparison of strain elastography and shear wave elastography in diagnosis of fibrosis in nonalcoholic fatty liver disease. J Med Ultrasonics 50, 187–195 (2023). https://doi.org/10.1007/s10396-023-01293-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10396-023-01293-z