Abstract
Purpose
To examine the 16-week outcomes of switching to brolucizumab in eyes with neovascular age-related macular degeneration (nAMD) refractory to aflibercept.
Study design
Retrospective observational study.
Methods
Data of eyes with nAMD who switched to brolucizumab because of resistance to aflibercept were collected. The best-corrected visual acuity (BCVA; in logarithm of the minimum angle of resolution), central retinal thickness (CRT), central choroidal thickness (CCT), and exudative status on optical coherence tomography were analyzed.
Results
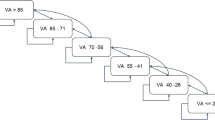
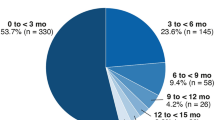
A total of 48 eyes of 48 patients were reviewed. At 4 to 7 weeks after switching, BCVA changed from 0.26 ± 0.19 to 0.25 ± 0.21 (not significant; P = 0.95), but CRT significantly decreased from 298.9 ± 108.4 µm to 241.9 ± 92.5 µm (P < 0.001) and CCT from 182.6 ± 89.3 µm to 169.7 ± 82.6 µm (P < 0.001). Of the 23 eyes refractory to monthly aflibercept injections, 12 (52.2%) achieved a dry macula, and 8 (34.8%) reduced exudative changes at 1 month. At 16 weeks, 31 eyes (64.6%) achieved the treatment interval ≥ 8 weeks. Two patients (4.2%) dropped out, 7 eyes (14.6%) developed intraocular inflammation (IOI), and 8 eyes (16.7%) switched back to aflibercept because of the failure to extend the treatment interval ≥ 8 weeks.
Conclusion
Switching to brolucizumab in eyes refractory to aflibercept conferred favorable outcomes in controlling exudative changes. However, IOI and the regulation of the treatment interval to at least 8 weeks during the maintenance phase disrupted the continuation of brolucizumab treatment.



Similar content being viewed by others
References
Traine PG, Pfister IB, Zandi S, Spindler J, Garweg JG. Long-term outcome of intravitreal aflibercept treatment for neovascular age-related macular degeneration using a “treat-and-extend” regimen. Ophthalmol Retina. 2019;3:393–9.
Tsunekawa Y, Kataoka K, Asai K, Ito Y, Terasaki H. Four-year outcome of aflibercept administration using a treat-and-extend regimen in eyes with recurrent neovascular age-related macular degeneration. Jpn J Ophthalmol. 2021;65:69–76.
Dugel PU, Koh A, Ogura Y, Jaffe GJ, Schmidt-Erfurth U, Brown DM, et al. HAWK and HARRIER: phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127:72–84.
Dugel PU, Singh RP, Koh A, Ogura Y, Weissgerber G, Gedif K, et al. HAWK and HARRIER: ninety-six-week outcomes from the phase 3 trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2021;128:89–99.
Augsburger M, Sarra GM, Imesch P. Treat and extend versus pro re nata regimens of ranibizumab and aflibercept in neovascular age-related macular degeneration: a comparative study. Graefes Arch Clin Exp Ophthalmol. 2019;257:1889–95.
DeCroos FC, Reed D, Adam MK, Salz D, Gupta OP, Ho AC, et al. Treat-and-extend therapy using aflibercept for neovascular age-related macular degeneration: a prospective clinical trial. Am J Ophthalmol. 2017;180:142–50.
Ohji M, Takahashi K, Okada AA, Kobayashi M, Matsuda Y, Terano Y, et al. Efficacy and safety of intravitreal aflibercept treat-and-extend regimens in exudative age-related macular degeneration: 52- and 96-week findings from ALTAIR : a randomized controlled trial. Adv Ther. 2020;37:1173–87.
Kataoka K, Horiguchi E, Kawano K, Ushida H, Nakano Y, Ito Y, et al. Three cases of brolucizumab-associated retinal vasculitis treated with systemic and local steroid therapy. Jpn J Ophthalmol. 2021;65:199–207.
Enriquez AB, Baumal CR, Crane AM, Witkin AJ, Lally DR, Liang MC, et al. Early experience with brolucizumab treatment of neovascular age-related macular degeneration. JAMA Ophthalmol. 2021;139:441–8.
Bulirsch LM, Sassmannshausen M, Nadal J, Liegl R, Thiele S, Holz FG. Short-term real-world outcomes following intravitreal brolucizumab for neovascular AMD: SHIFT study. Br J Ophthalmol. 2021. https://doi.org/10.1136/bjophthalmol-2020-318672.
Matsumoto H, Hoshino J, Mukai R, Nakamura K, Akiyama H. Short-term outcomes of intravitreal brolucizumab for treatment-naive neovascular age-related macular degeneration with type 1 choroidal neovascularization including polypoidal choroidal vasculopathy. Sci Rep. 2021;11:6759.
Yamamoto A, Okada AA, Kano M, Koizumi H, Saito M, Maruko I, et al. One-year results of intravitreal aflibercept for polypoidal choroidal vasculopathy. Ophthalmology. 2015;122:1866–72.
Lee WK, Iida T, Ogura Y, Chen SJ, Wong TY, Mitchell P, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy in the PLANET study: a randomized clinical trial. JAMA Ophthalmol. 2018;136:786–93.
Yonekawa Y, Andreoli C, Miller JB, Loewenstein JI, Sobrin L, Eliott D, et al. Conversion to aflibercept for chronic refractory or recurrent neovascular age-related macular degeneration. Am J Ophthalmol. 2013;156:29–35.
Haug SJ, Hien DL, Uludag G, Ngoc TTT, Lajevardi S, Halim MS, et al. Retinal arterial occlusive vasculitis following intravitreal brolucizumab administration. Am J Ophthalmol Case Rep. 2020;18:100680.
Kondapalli SSA. Retinal vasculitis after administration of brolucizumab resulting in severe loss of visual acuity. JAMA Ophthalmol. 2020;138:1103–4.
Maruko I, Okada AA, Iida T, Hasegawa T, Izumi T, Kawai M, et al. Brolucizumab-related intraocular inflammation in Japanese patients with age-related macular degeneration: a short-term multicenter study. Graefes Arch Clin Exp Ophthalmol. 2021;259:2857–9.
Baumal CR, Spaide RF, Vajzovic L, Freund KB, Walter SD, John V, et al. Retinal vasculitis and intraocular inflammation after intravitreal injection of brolucizumab. Ophthalmology. 2020;127:1345–59.
Witkin AJ, Hahn P, Murray TG, Arevalo JF, Blinder KJ, Choudhry N, et al. Occlusive retinal vasculitis following intravitreal brolucizumab. J Vitreoretin Dis. 2020;4:69–279.
Gharbiya M, Cruciani F, Mariotti C, Grandinetti F, Marenco M, Cacace V. Choroidal thickness changes after intravitreal antivascular endothelial growth factor therapy for age-related macular degeneration: ranibizumab versus aflibercept. J Ocul Pharmacol Ther. 2015;31:357–62.
Koizumi H, Kano M, Yamamoto A, Saito M, Maruko I, Sekiryu T, et al. Subfoveal choroidal thickness during aflibercept therapy for neovascular age-related macular degeneration: twelve-month results. Ophthalmology. 2016;123:617–24.
Kim JH, Lee TG, Chang YS, Kim CG, Cho SW. Short-term choroidal thickness changes in patients treated with either ranibizumab or aflibercept: a comparative study. Br J Ophthalmol. 2016;100:1634–9.
Mazaraki K, Fassnacht-Riederle H, Blum R, Becker M, Michels S. Change in choroidal thickness after intravitreal aflibercept in pretreated and treatment-naive eyes for neovascular age-related macular degeneration. Br J Ophthalmol. 2015;99:1341–4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
H. Ota, None; J. Takeuchi, Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (Novartis, Santen, Bayer); Y. Nakano, None; E. Horiguchi, None; Y. Taki, None; Y. Ito, Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (Bayer, Canon, ZEISS, Kowa, Novartis, Pfizer, Santen); H. Terasaki, Grant (ZEISS, Kowa, Otsuka, Senju, Santen, Alcon, Sanofi, Novartis, ROHTO, Bayer, Wakamoto, HOYA, Johnson&Johnson), Consulting fees (Bayer), Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (Otsuka, Kowa, Santen, Senju, Sanofi, Alcon, ROHTO, Novartis, Wakamoto, Bayer), Support for attending meetings and/or travel (Bayer); K. M. Nishiguchi, Grant (Takara Bio, Takeda, JCR Pharma, Alcon, Bayer, Novartis, Santen), Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (Alcon, Bayer, Novartis, Santen, Chugai, Kowa, Senju, Otsuka, Wakamoto), Patents planned, issued or pending (A gene therapy using genome editing with a single AAV vector); K. Kataoka, Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (Novartis, Senju, Santen, Bayer, Boehringer Ingelheim, Bayer).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Keiko Kataoka
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Ota, H., Takeuchi, J., Nakano, Y. et al. Switching from aflibercept to brolucizumab for the treatment of refractory neovascular age-related macular degeneration. Jpn J Ophthalmol 66, 278–284 (2022). https://doi.org/10.1007/s10384-022-00908-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-022-00908-1