Abstract
Purpose
To investigate the protective effects of d-allose, a rare sugar, on pressure-induced ischemia during vitrectomy in the rabbit eye.
Methods
The rabbits underwent pars plana vitrectomy, and continuous intraocular irrigation at a perfusion pressure of 140 mmHg was performed for 45 min. Intraocular pressure was regulated by adjusting the height of a bottle of balanced saline solution containing d-allose. Morphometric studies were performed to study the effects of d-allose on the histological changes induced by ischemia in the rabbit retina. Electroretinograms (ERGs) were taken before and 1 and 7 days after vitrectomy. Nitroblue tetrazolium was used as an index of superoxide anion (O ·−2 ) generation. Data were analyzed by use of the unpaired Student’s t test.
Results
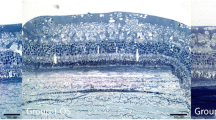
Seven days after ischemia, significant reductions in both number of ganglion cells and the thickness of the inner plexiform layer were observed. d-Allose significantly inhibited ischemic injury of the inner retina (P < 0.05). On postoperative day 7, amplitudes of ERG b-waves were significantly lower in the control group than in the d-allose group (P < 0.05). d-Allose suppressed the production of O2 −.
Conclusions
Intraocular irrigation with d-allose during vitrectomy may protect the retina against ischemia-induced damage.






Similar content being viewed by others
References
Bhuiyan SH, Itami Y, Yuhko R, Katayama T, Izumori K. d-Allose production from d-psicose using immobilized l-rhamnose isomerase. J Ferment Bioeng. 1998;85:540–2.
Murata A, Sekiya K, Watanabe Y, Yamaguchi F, Hatano N, Izumori K, et al. A novel inhibitory effect of d-allose on production of reactive oxygen species from neutrophils. J Biosci Bioeng. 2003;96:89–91.
Hirooka K, Miyamoto O, Jinming P, Du Y, Itano T, Baba T, et al. Neuroprotective effect of d-allose against retinal ischemia–reperfusion injury. Invest Ophthalmol Vis Sci. 2006;47:1653–7.
Seki M, Togashi H, Ando N. Optic nerve atrophy after vitrectomy for diabetic retinopathy: its systemic and local risk factors. Nippon Ganka Gakkai Zasshi. 2006;110:462–7.
Jacobson MD. Reactive oxygen species and programmed cell death. Trends Biochem Sci. 1996;21:83–6.
Kroemer G, Petit P, Zamzami N, Vayssière JL, Mignotte B. The biochemistry of programmed cell death. FASEB J. 1995;9:1277–87.
Bonne C, Muller A, Villain M. Free radicals in retinal ischemia. Gen Pharmacol. 1998;30:275–80.
Tamai K, Toumoto E, Majima A. Local hypothermia protects the retina from ischemic injury in vitrectomy. Br J Ophthalmol. 1997;81:789–94.
Traustason S, Eysteinsson T, Agnarsson BA, Stefánsson E. GABA agonists fail to protect the retina from ischemia–reperfusion injury. Exp Eye Res. 2009;88:361–6.
Digregorio KA, Cilento EV, Lantz RC. Measurement of superoxide release from single pulmonary alveolar macrophages. Am J Physiol. 1987;252:C677–83.
Ullrey DB, Kalckar HM. Search for cellular phosphorylation products of d-allose. Proc Natl Acad Sci USA. 1991;88:1504–5.
Coleman K, Fitzgerald D, Eustace, Bouchier-Hayes D, et al. Electroretinography, retinal ischemia and carotid artery disease. Eur J Vasc Surg. 1990;4:569–73.
Zager E, Ames A. Reduction of cellular energy requirements: screening for agents that may protect against CNS ischemia. J Neurosurg. 1988;69:568–79.
Choi DW. Glutamate neurotoxicity and diseases of the nervous system. Neuron. 1988;1:623–34.
Louzada-Junior P, Dias JJ, Santos WF, Lachat JJ, Bradford HF, Coutinho-Netto J. Glutamate release in experimental ischaemia of the retina: an approach using microdialysis. J Neurochem. 1992;59:358–63.
Adachi K, Kashii S, Masai H, Ueda M, Morizane C, Kaneda, et al. Mechanism of the pathogenesis of glutamate neurotoxicity in retinal ischemia. Graefes Arch Clin Exp Ophthalmol. 1998;236:766–74.
Auclair C, Voisin E. Nitroblue tetrazolium reduction. In: Greenwald RA, editor. Handbook of methods for oxygen radicals research. Boca Raton: CRC Press; 1988. p. 123–32.
Altman FP. Tetrazolium salts and formazans. Prog Histochem Cytochem. 1976;9:1–56.
Zhang H, Agardh E, Agardh CD. Nitro blue tetrazolium staining: a morphological demonstration of superoxide in the rat retina. Graefes Arch Ophthalmol. 1993;231:178–83.
Gonzalez-Flecha B, Boveris A. Mitochondrial sites of hydrogen peroxide production in reperfused rat kidney cortex. Biochim Biophys Acta. 1995;1243:361–6.
Hockenbery DM, Oltvai Z, Yin XM, Milliman CL, Korsmeyer SJ. Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell. 1993;75:241–51.
Kohner EM, Patel V, Rassam SM. Role of blood flow and impaired autoregulation in the pathogenesis of diabetic retinopathy. Diabetes. 1995;44:603–6.
Arnold AC. Pathogenesis of nonarteritic anterior ischemic optic neuropathy. J Neuroophthalmol. 2003;23:157–63.
Ikushima M, Tano Y, Ikeda T, Sato Y. Hyper-infusion pressure for diabetic membrane dissection. Jpn J Ophthalmol. 1990;34:393–400.
Ambati J, Chalam KV, Chawla DK, D’Angio CT, Guillet EG, Rose SJ, et al. Elevated γ-aminobutyric acid, glutamate, and vascular endothelial growth factor levels in the vitreous of patients with proliferative diabetic retinopathy. Arch Ophthalmol. 1997;11:1161–6.
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (20592078).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Mizote, M., Hirooka, K., Fukuda, K. et al. d-Allose as ischemic retina injury inhibitor during rabbit vitrectomy. Jpn J Ophthalmol 55, 294–300 (2011). https://doi.org/10.1007/s10384-011-0012-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-011-0012-8