Abstract
Purpose
The suprachoroidal space is preferred for many reasons for implantation of retinal prostheses or drug delivery systems. This study aimed to investigate the safety and feasibility of suprachoroidal fibrin glue (FG), one of the adhesives used to fix the implanted material.
Methods
Human retinal pigment epithelium cells (hRPE) were cultured with FG, assayed with 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) assay, and studied by transmission electron microscopy. A total of 48 rabbits were randomly divided into three groups to undergo surgery: group 1, 0.1 ml suprachoroidal FG; group 2, 0.1 ml suprachoroidal normal saline (control); group 3, 0.1 ml intravitreal FG. Slit-lamp examination, indirect ophthalmoscopy, fundus photography, fundus fluorescein angiography (FFA), indocyanine green angiography (IA), electroretinography (ERG), and histopathological examination were performed at various time points till 90 days after injection.
Results
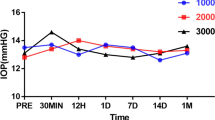
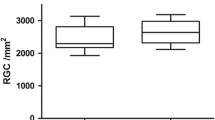
No apoptosis changes of electron microscopy were observed in the hRPE cultured with FG, and the difference in the MTT test results was not statistically significant when compared with the control group (P > 0.2). One day after suprachoroidal injection, slight choroidal hemorrhage occurred. No severe intraocular reactions were observed in any of the three groups, and FFA and IA showed no edema or leakage around blood vessels. There was no significant difference in latency or amplitude of the ERG wave among the three groups (P > 0.1). Histopathological examinations showed the suprachoroidal FG induced localized inflammation (from acute inflammation to chronic phagocytosis) at the early stage and finally turned into fibrosis tissue.
Conclusions
FG is feasible for suprachoroidal use, and no obvious toxic reaction was observed in either in vitro or in vivo studies.
Similar content being viewed by others
References
Zhou JA, Woo SJ, Park SI, et al. A suprachoroidal electrical retinal stimulator design for long-term animal experiments and in vivo assessment of its feasibility and biocompatibility in rabbits. J Biomed Biotechnol 2008;2008:547428.
Sakaguchi H, Fujikado T, Fang X, et al. Transretinal electrical stimulation with a suprachoroidal multichannel electrode in rabbit eyes. Jpn J Ophthalmol 2004;48:256–261.
Tokuda T, Asano R, Sugitani S, et al. In vivo stimulation on rabbit retina using CMOS LSI-based multi-chip flexible stimulator for retinal prosthesis. 2007 Conf Proc IEEE Eng Med Biol Soc; 2007. p. 5791–5794.
Okawa Y, Fujikado T, Miyoshi T, et al. Optical imaging to evaluate retinal activation by electrical currents using suprachoroidal-transretinal stimulation. Invest Ophthalmol Vis Sci 2007;48:4777–4784.
Nakauchi K, Fujikado T, Kanda H, et al. Threshold suprachoroidal-transretinal stimulation current resulting in retinal damage in rabbits. J Neural Eng 2007;4:S50–57.
Einmahl S, Savoldelli M, D’Hermies F, Tabatabay C, Gurny R, Behar-Cohen F. Evaluation of a novel biomaterial in the suprachoroidal space of the rabbit eye. Invest Ophthalmol Vis Sci 2002;43:1533–1539.
Olsen TW, Feng X, Wabner K, et al. Cannulation of the suprachoroidal space: a novel drug delivery methodology to the posterior segment. Am J Ophthalmol 2006;142:777–787.
Detweiler MB, Detweiler JG, Fenton J. Sutureless and reduced suture anastomosis of hollow vessels with fibrin glue: a review. J Invest Surg 1999;12:245–262.
Herget GW, Kassa M, Riede UN, Lu Y, Brethner L, Hasse J. Experimental use of an albumin-glutaraldehyde tissue adhesive for sealing pulmonary parenchyma and bronchial anastomoses. Eur J Cardiothorac Surg 2001;19:4–9.
Kram HB, Reuben BI, Fleming AW, Shoemaker WC. Use of fibrin glue in hepatic trauma. J Trauma 1988;28:1195–1201.
Steinkogler FJ, Moser E. Canaliculo-cystostomy using the fibrin glue technic. Fortschr Ophthalmol 1989;86:76–77.
Chan SM, Boisjoly H. Advances in the use of adhesives in ophthalmology. Curr Opin Ophthalmol 2004;15:305–310.
Zhou P, Zhao MW, Li XX, Yu WZ, Bian ZM. siRNA targeting mammalian target of rapamycin (mTOR) attenuates experimental proliferative vitreoretinopathy. Curr Eye Res 2007;32:973–984.
Eibl KH, Banas B, Schoenfeld CL, et al. Alkylphosphocholines inhibit proliferation of human retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 2003;44:3556–3561.
Tao Y, Li XX, Jiang YR, Bai XB, Wu BD, Dong JQ. Diffusion of macromolecule through retina after experimental branch retinal vein occlusion and estimate of intraretinal barrier. Curr Drug Metab 2007;8:151–156.
Tilanus MA, Deutman AF. Full-thickness macular holes treated with vitrectomy and tissue glue. Int Ophthalmol 1994–1995;18:355–358.
Alston SM, Solen KA, Broderick AH, Sukavaneshvar S, Mohammad SF. New method to prepare autologous fibrin glue on demand. Transl Res 2007;149:187–195.
Alston SM, Solen KA, Sukavaneshvar S, Mohammad SF. In vivo efficacy of a new autologous fibrin sealant. J Surg Res 2008;146:143–148.
Siedentop KH, Harris DM, Sanchez B. Autologous fibrin tissue adhesive: factors influencing bonding power. Laryngoscope 1988;98:731–733.
Machemer R. Proliferative vitreoretinopathy (PVR): a personal account of its pathogenesis and treatment. Proctor lecture. Invest Ophthalmol Vis Sci 1988;29:1771–1783.
Taban M, Ventura AA, Sharma S, Kaiser PK. Dynamic evaluation of sutureless vitrectomy wounds: an optical coherence tomography and histopathology study. Ophthalmology 2008;115:2221–2228.
Cong L, Sun D, Zhang Z, Jiao W, Rizzolo LJ, Peng S. A novel rabbit model for studying RPE transplantation. Invest Ophthalmol Vis Sci 2008;49:4115–4125.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.H. and Y.T. contributed equally to the work presented here and should therefore be regarded as equivalent first authors.
About this article
Cite this article
Hou, J., Tao, Y., Jiang, Yr. et al. In vivo and in vitro study of suprachoroidal fibrin glue. Jpn J Ophthalmol 53, 640–647 (2009). https://doi.org/10.1007/s10384-009-0725-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-009-0725-0