Abstract
Purpose
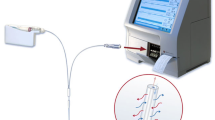
Intestinal ischemia is a major cause of anastomotic leak and death and remains a clinical challenge as the physician relies on several nonspecific signs, biologic markers, and radiologic studies to make the diagnosis. This study used rapid sampling online microdialysis to evaluate the biochemical changes occurring in a segment of human bowel during and after resection, and assessed for the feasibility and reproducibility of this technique in monitoring intestinal ischemia.
Methods
A custom made, rapid sampling online microdialysis analyzer was used to monitor the changes in the bowel wall of specimens being resected intraoperatively. Two patients were recruited for the pilot study to optimize the analyzer and seven patients undergoing colonic resections were recruited for the data collection and analysis.
Results
The concentration of glucose in the extracellular bowel wall fluid decreased transiently after division of individual feeding arteries followed by a rebound increase in the concentration back to baseline concentrations. After completion of resection, glucose concentrations continued to decrease while lactate concentrations increased constantly.
Conclusion
Rapid sampling microdialysis was feasible in the clinical environment. These results suggest that tissue responds to ischemic insult by mobilizing glucose stores which later decrease again, whereas lactate concentrations constantly increased.




Similar content being viewed by others
References
Bruce J, Krukowski ZH, Al-Khairy G, et al. Systematic review of the definition and measurement of anastomotic leak after gastrointestinal surgery. Br J Surg 2001;88:1157–68.
Krohg-Sorensen K, Line PD, Haaland T, et al. Intraoperative prediction of ischaemic injury of the bowel: a comparison of laser Doppler flowmetry and tissue oximetry to histological analysis. Eur J Vasc Surg 1992;6:518–24.
Lynch TG, Hobson RW, Kerr JC, et al. Doppler ultrasound, laser Doppler, and perfusion fluorometry in bowel ischemia. Arch Surg 1988;123:483–6.
Cerny V, Cvachovec K. Gastric tonometry and intramucosal pH—theoretical principles and clinical application. Physiol Res 2000;49:289–97.
Carter MS, Fantini GA, Sammartano RJ, et al. Qualitative and quantitative fluorescein fluorescence in determining intestinal viability. Am J Surg 1984;147:117–23.
Bradbury MS, Kavanagh PV, Bechtold RE, et al. Mesenteric venous thrombosis: diagnosis and noninvasive imaging. Radiographics 2002;22:527–41.
Murray MJ, Gonze MD, Nowak LR, et al. Serum D(-)-lactate levels as an aid to diagnosing acute intestinal ischemia. Am J Surg 1994;167:575–8.
Ungerstedt U, Pycock C. Functional correlates of dopamine neurotransmission. Bull Schweiz Akad Med Wiss 1974;30:44–55.
Benveniste H, Hansen AJ, Ottosen NS. Determination of brain interstitial concentrations by microdialysis. J Neurochem 1989;52:1741–50.
Battezzati A, Bertoli S. Methods of measuring metabolism during surgery in humans: focus on the liver-brain relationship. Curr Opin Clin Nutr Metab Care 2004;7:523–30.
Kennergren C, Mantovani V, Lonnroth P, et al. Extracellular amino acids as markers of myocardial ischemia during cardioplegic heart arrest. Cardiology 1999;91:31–40.
Hagstrom-Toft E, Enoksson S, Moberg E, et al. Absolute concentrations of glycerol and lactate in human skeletal muscle, adipose tissue, and blood. Am J Physiol 1997;273:E584–92.
Tenhunen JJ, Kosunen H, Juvonen P, et al. Intestinal mucosal microdialysis: histamine release in splanchnic ischemia/reperfusion injury in piglets. Inflamm Res 1996;45(Suppl 1):S52–3.
Oldner A, Goiny M, Ungerstedt U, et al. Splanchnic homeostasis during endotoxin challenge in the pig as assessed by microdialysis and tonometry. Shock 1996;6:188–93.
Sommer T, Larsen JF. Intraperitoneal and intraluminal microdialysis in the detection of experimental regional intestinal ischaemia. Br J Surg 2004;91:855–61.
Jansson K, Ungerstedt J, Jonsson T, et al. Human intraperitoneal microdialysis: increased lactate/pyruvate ratio suggests early visceral ischaemia. A pilot study. Scand J Gastroenterol 2003;38:1007–11.
Jones D, Parkin M, Langemann H, et al. On-line monitoring in neurointensive care- enzyme-based electrochemical assay for simultaneous, continuous monitoring of glucose and lactate from critical care patients. J Electroanal Chem 2002;538:243–52.
Bhatia R, Hashemi P, Razzaq A, et al. Application of rapid-sampling, online microdialysis to the monitoring of brain metabolism during aneurysm surgery. Neurosurgery 2006;58:ONS–313–21.
Hopwood SE, Parkin MC, Bezzina EL, et al. Transient changes in cortical glucose and lactate levels associated with peri-infarct depolarisations, studied with rapid-sampling microdialysis. J Cereb Blood Flow Metab 2005;25:391–401.
Parkin M, Hopwood S, Jones DA, et al. Dynamic changes in brain glucose and lactate in pericontusional areas of the human cerebral cortex, monitored with rapid sampling on-line microdialysis: relationship with depolarisation-like events. J Cereb Blood Flow Metab 2005;25:402–13.
Boutelle MG, Fillenz M. Clinical microdialysis: the role of on-line measurement and quantitative microdialysis. Acta Neurochir (Suppl) 1996;67:13–20.
Alemany CA, Oh W, Stonestreet BS. Effects of nitric oxide synthesis inhibition on mesenteric perfusion in young pigs. Am J Physiol 1997;272:G612–6.
Myers SI, Turnage RH, Hernandez R, et al. Autoregulation of renal and splanchnic blood flow following infra-renal aortic clamping is mediated by nitric oxide and vasodilator prostanoids. J Cardiovasc Surg 1996;37:97–103.
Jacobson ED, Pawlik WW. Adenosine regulation of mesenteric vasodilation. Gastroenterology 1994;107:1168–80.
Granger HJ, Norris CP. Intrinsic regulation of intestinal oxygenation in the anesthetized dog. Am J Physiol 1980;238:H836–43.
Granger DN, Granger HJ. Systems analysis of intestinal hemodynamics and oxygenation. Am J Physiol 1983;245:G786–96.
Bulkley GB, Kvietys PR, Parks DA, et al. Relationship of blood flow and oxygen consumption to ischemic injury in the canine small intestine. Gastroenterology 1985;89:852–7.
Tenhunen JJ, Kosunen H, Alhava E, et al. Intestinal luminal microdialysis: a new approach to assess gut mucosal ischemia. Anesthesiology 1999;91:1807–15.
Shepherd AP. Intestinal O2 consumption and 86Rb extraction during arterial hypoxia. Am J Physiol 1978;234:E248–51.
James JH, Luchette FA, McCarter FD, et al. Lactate is an unreliable indicator of tissue hypoxia in injury or sepsis. Lancet 1999;354:505–8.
Parkin M, Hopwood S, Strong A, et al. Resolving dynamic changes in brain metabolism using biosensors and on-line microdialysis. Trends Anal Chem 2003;22:487–97.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by Covidien.
An erratum to this article can be found at http://dx.doi.org/10.1007/s10350-008-9433-y
About this article
Cite this article
Deeba, S., Corcoles, E.P., Hanna, B.G. et al. Use of Rapid Sampling Microdialysis for Intraoperative Monitoring of Bowel Ischemia. Dis Colon Rectum 51, 1408–1413 (2008). https://doi.org/10.1007/s10350-008-9375-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10350-008-9375-4